Article Contents
| Clin Exp Pediatr > Volume 65(2); 2022 |
|
Abstract
Background
There is evidence of a relationship between prenatal excess androgen exposure and central nervous developmental problems and attention-deficit/hyperactivity disorder (ADHD) in the offspring of mothers with polycystic ovary syndrome (PCOS).
Purpose
Here we aimed to use a meta-analysis to investigate whether the offspring of mothers with PCOS are at an increased chance of developing ADHD.
Methods
Three main English databases were searched for articles published through December 2020. The NewcastleOttawa Scale was used to assess study quality. Study heterogeneity was determined using I2 statistics and publication bias was assessed using Begg and Egger tests. The results are presented as odds ratio (OR) and relative ratio (RR) estimates with 95% confidence intervals (CIs) using a random-effects model.
Results
Six articles (3 cohort and 3 case-control studies; 401,413 total ADHD cases) met the study criteria. Maternal PCOS was associated with an increased risk of ADHD in the offspring based on OR and RR (OR, 1.42; 95% CI, 1.27–1.57) and (RR, 1.43; 95% CI, 1.35–1.51), respectively. There was no heterogeneity among the included articles based on OR (I2=0.0%, P=0.588) and RR (I2=0.0%, P=0.878).
Attention-deficit/hyperactivity disorder (ADHD) is a childhood mood disorders though some symptoms often continue to adulthood [1]. It is characterized by pervasive symptoms of attention-deficit/hyperactivity according to Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) [2]. ADHD categorized in 3 types inattentive, hyperactive-impulsive, or both inattention and hyperactive-impulsive symptoms [3]. The prevalence of ADHD is varied in worldwide. According to the meta-analysis overall prevalence of ADHD was 7.2% [4]. The etiology of ADHD is not fully understood but many environmental, genetic, and environment-gene interactions factors can lead to the development of ADHD [5]. In terms of frequency, ADHD is more common in boys than girls [6]. These observations suggest that early intrauterine exposure of the fetus to a high level of androgen and sex hormone are probably associated with hyperactivity by affecting brain development [7].
One of the sources of excess androgen exposure of mother is polycystic ovary syndrome (PCOS). Other common finding in women with PCOS are chorionic anovulation, subinfertility, and metabolic syndrome [8]. According to a prospective cohort study, the risk of failing the Ages and Stages Questionnaire among children who born to women reported PCOS treatment was more than the offspring of women who reported receiving no treatment for PCOS [9]. The association between maternal PCOS with ADHD, autism spectrum disorders (ASDs), and ADHD comorbid with ASD have been reported in some studies may suggest evidence on offspring behavioral and developmental disorders [10,11]. The evidence of meta-analyses emphasizes that the odds of ASD in the offspring women with PCOS have increased 1.66 times [12] but the association between polycystic ovary syndrome and the risk of ADHD among their offspring has not been systematically reviewed. Therefore, we conducted the first review and meta-analysis to assess whether women with PCOS have increased risk of having an offspring with ADHD.
The aim of this review was to determine whether a mother with PCOS has increased chance of ADHD in her offspring. The review is reported based on the PRISMA checklist [13].
The English electronic databases included the Web of Science, PubMed, and Scopus were systematically searched for studies published to December 2020 with restricted English language using the following search strategies in accordance with the MeSH browser keywords and free-text words: [((((("polycystic ovary syndrome"[Title/Abstract]) OR ("Stein-Leventhal Syndrome"[Title/Abstract])) OR ("Sclerocystic Ovary"[Title/Abstract])) OR ("Ovarian Syndrome"[Title/Abstract])) OR ("Polycystic Ovary Syndrome 1"[Title/Abstract]) AND ((((("Attention Deficit Hyperactivity Disorders"[Title/Abstract]) OR ("Hyperkinetic Syndrome"[Title/Abstract])) OR (ADDH*[Title/Abstract])) OR (ADDH[Title/Abstract])) OR ("Minimal Brain Dysfunction" [Title/Abstract])) OR ("Attention Deficit Disorders"[Title/Abstract]) AND "Case-control study "[Title/Abstract]) AND "Cohort study" [Title/Abstract])]. In addition, the list reference of selected articles was checked and contacted the authors if required to obtain full texts of study.
All case-control and cohort (both prospective and retrospective) studies that provided data on the rate of ADDH among the offspring of women with PCOS were included the review.
The PCOS criteria in mother are defined by Rotterdam, National Institute of Health and the accepted clinical criteria International Classification of Diseases (ICD)-8, ICD-9/ICD-10, and self-reported. Rotterdam criteria are defined by 2 of the following 3 features: (1) oligo or anovulation, (2) clinical and/or biochemical signs of hyperandrogenism, or (3) polycystic ovaries [14]. The ADHD criteria are defined by the accepted clinical criteria DSM-IV, ICD-9, and ICD-10. ADHD criteria are defined 5 or more symptoms of inattention and/or ≥5 symptoms of hyperactivity/impulsivity must have persisted for ≥6 months to a degree that is inconsistent with the developmental level and negatively impacts social and academic/occupational activities [15].
All experimental in vivo/in vitro study, cross-sectional, review, and case report/series studies were excluded.
Two authors (EJ and AM) independently searched, and screened duplicates, the titles, and abstracts the studies. The information of full texts extracted using a structured form included author name, year, design, sample size, estimate, adjustment, maternal age rang (year)/mean, ADHD criteria, and study quality. Disagreements of the extractors were resolved by discussion with a third person.
The Newcastle-Ottawa Scale (NOS) for cohort and case-control studies have been used to the evaluation of quality in individual studies [16]. The NOS includes 8 items. Each item gets a score when it will be appropriate criteria. Therefore, a maximum score considered high quality.
STATA 16 (StataCorp LP., College Station, TX, USA) was used for analyses. The heterogeneity of the studies was assessed using I2. The I2 equal or lower than 50% considered adequate heterogeneity. In this study, random effect model was applied due to true effect size. We presented results using odds ratio (OR) and relative ratio (RR) estimates with its 95% confidence intervals (CI) using a random-effects model. Publication bias was assessed using the funnel plot and Egger and Begg tests [17].
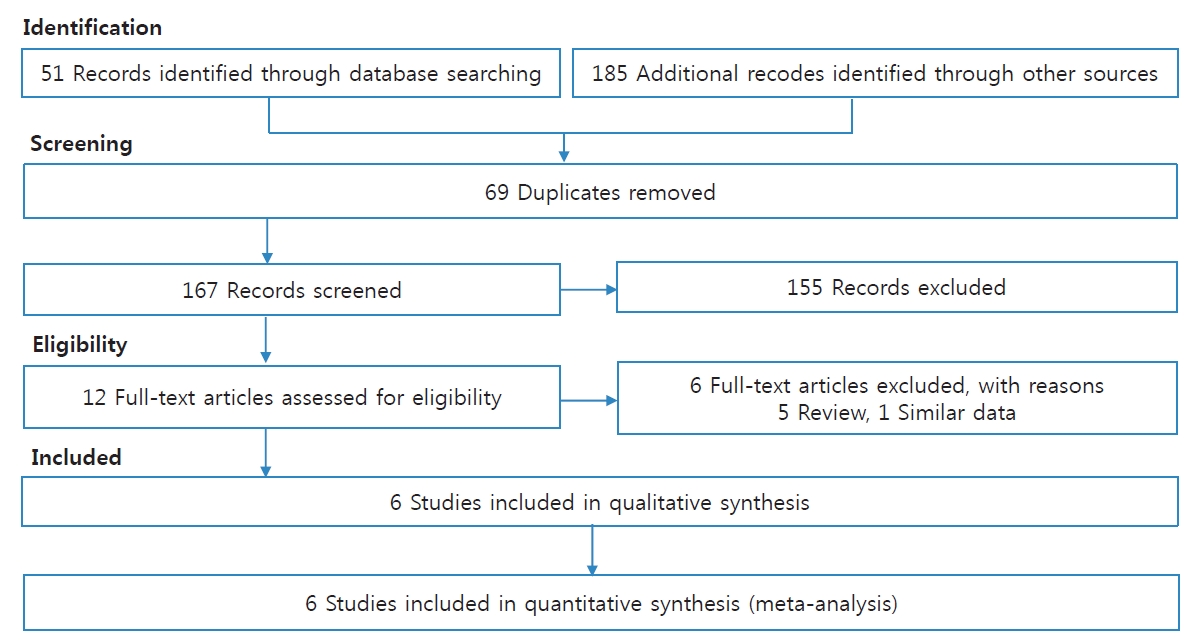
We identified 236 articles by search in 3 major databases and other sources. Among these, 69 duplicate articles and 155 unrelated articles were removed. Then, 12 articles were remained for review of the full paper. After review, 6 articles were excluded due to 5 review articles and 1 article with similar data to another article. In total, 6 articles (3 cohort studies and 3 case-control studies) were included in the present meta-analysis [10,11,18-21]. Fig. 1 presents the process of the included articles.
Preterm labor, maternal and paternal age, birth order, household income, mother's country of birth, small for gestational age, and preeclampsia were the confounding variables of the association between PCOS and the risk of ADHD among offspring.
Fig. 2 presents the association between PCOS and the risk of ADHD among the included studies. The pool estimates of the present meta-analysis reported that PCO in maternal was associated with the risk of ADHD among offspring based on OR and RR (OR, 1.42; 95% CI, 1.27–1.57) and (RR, 1.43; 95% CI, 1.35–1.51), respectively. There was not heterogeneity among included articles based on OR (I2=0.0%, P=0.588) and RR (I2=0.0%, P=0.878).
The publication bias did not find among studies based on Begg and Egger tests. The P values for Egger and Begg tests were P=0.133 and P=0.188, respectively.
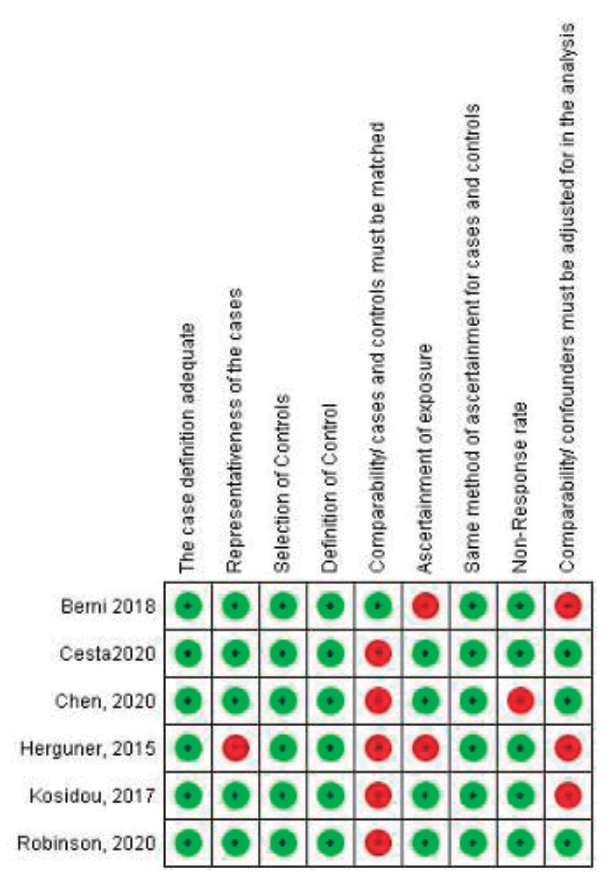
The quality of the studies based on the NOS Scale was presented in Table 1. The quality of the studies in the present meta-analysis was categorized into 5 studies with high quality and 1 study with low quality. Fig. 3 shows review authors' judgments about each risk of bias item for each included study.
To our knowledge, this is the first systematic review to assess the risk of ADHD in the children of women with PCOS compared to control group. Overall, based on the available evidence, the meta-analysis showed that the PCOS in maternal was associated with increasing the risk of ADHD among her offspring. There was between-study homogeneity in the present meta-analysis.
However, in a meta-analysis of 10 studies has been reported evidence that chance of having ASD significantly increased in offspring of women with PCOS [12]. Although, brain structure of ADHD and ASD have distinct neural features in gray and white matter structures, the majority of ASD symptoms commonly cooccur with ADHD symptoms but the minority of individuals with ADHD demonstrates ASD symptoms [22].
PCOS may expose the developing fetus to excess androgens because women with PCOS increase circulating androgen levels during pregnancy and have more placental androgen capacity. Brain development is influenced significantly by exposure to androgens during early gestation [23]. Prenatal androgen exposure may increase the risk of ASD and ADHD by affecting dendritic morphology, nerve density, abnormal synapse function, and morphology. Also, PCOS in mother is associated with more circulating androgens and inflammatory cytokines, which may affect fetal brain development [24].
Robinson et al. [21] showed that one-third of women with PCOS had hirsutism. The hirsutism in maternal was association with ADHD among offspring, conduct disorder, peer relationship problems, prosocial problems, and emotional symptoms.
Some reasonable mechanism underlying the potential association between maternal PCOS and ADHD. Poor pregnancy outcome has been reported in women with PCOS such as abortion, preeclampsia, gestational diabetes mellitus (GDM), small/large for gestational age infants, cesarean delivery, and preterm delivery [25]. It is known that the risk of ADHD increases among offspring whose mothers had obesity, preeclampsia and GDM [26-28]. Obesity, infertility, and the use of fertility treatment more experienced by women with PCOS might be influenced offspring [29,30]. Women with PCOS are known to have a higher risk of developing depression and adult ADHD. This risk, in turn, might cause an increase in psychiatric morbidity in their children [18].
Our study has some limitations. Although, most of the studies in present meta-analysis were adjusted for some risk factors of ADHD. However, 2 studies reported crude form for ADHD. In addition, there was limit studies about the association between of PCOS and the risk of ADHD that has been done so far. Therefore, these limitations may increase the possibility of bias that should be determined when interpreting these findings. However, our study with 401,413 participants showed that PCOS is a risk factor for ADHD among offspring.
Our study showed that maternal PCOS is a risk factor for ADHD. Therefore, Screening for ADHD among children of these women should be considered as part of the comprehensive clinical care for women with PCOS.
Acknowledgments
This study was funded by Hamadan University of Medical Sciences with code 9911218343.
We thank from Hamadan University of Medical Sciences for their support.
Fig. 2.
Forest plot of the association between polycystic ovary syndrome and the risk of attention-deficit/hyperactivity disorder. OR, odds ratio; RR, relative ratio.

Table 1.
Characteristics of the included studies
| Study | Design | Sample size | Estimate | Adjustment | PCOS criteria | Mean maternal age rang (yr) | ADHD criteria | Quality |
|---|---|---|---|---|---|---|---|---|
| Kosidou et al. [10], 2017 | Case-control | 58,912 | Odds ratio | Adjusted | ICD-8/ICD-9/ICD-10 | 27.6 | ICD-9/ICD-10 | High |
| Robinson et al. [21], 2020 | Cohort | 1,915 | Risk ratio | Adjusted | Self-report | 31.3 | DSM-IV | High |
| Chen et al. [20], 2020 | Cohort | 105,409 | Hazard ratio | Adjusted | ICD-9/ICD-10 | No data | ICD-10 | High |
| Cesta et al. [19], 2020 | Cohort | 218,111 | Hazard ratio | Adjusted | Rotterdam/ICD-9/ICD-10 | 28 | ICD-9/ICD-10 | High |
| Berni et al. [18], 2018 | Case-control | 16,986 | Odds ratio | Crude | ICD-10 | 27 | ICD-10 | High |
| Hergüner et al. [11], 2015 | Case-control | 80 | Odds ratio | Crude | Rotterdam | 18,035 | DSM-IV | Low |
References
1. Ginsberg Y, Quintero J, Anand E, Casillas M, Upadhyaya HP. Underdiagnosis of attention-deficit/hyperactivity disorder in adult patients: a review of the literature. Prim Care Companion CNS Disord 2014;16:PCC.13r01600.


2. Zayats T, Neale BM. Recent advances in understanding of attention deficit hyperactivity disorder (ADHD): how genetics are shaping our conceptualization of this disorder [version 2; peer review: 3 approved]. F1000Res 2019;8:F1000 Faculty Rev-2060.

3. McFadden D, Westhafer JG, Pasanen EG, Carlson CL, Tucker DM. Physiological evidence of hypermasculinization in boys with the inattentive type of attention-deficit/hyperactivity disorder (ADHD). Clin Neurosci Res 2005;5:233-45.

4. Thomas R, Sanders S, Doust J, Beller E, Glasziou P. Prevalence of attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. Pediatrics 2015;135:e994-1001.


5. Sciberras E, Mulraney M, Silva D, Coghill D. Prenatal risk factors and the etiology of ADHD—review of existing evidence. Curr Psych Rep 2017;19:1.

6. Mowlem F, Agnew-Blais J, Taylor E, Asherson P. Do different factors influence whether girls versus boys meet ADHD diagnostic criteria? Sex differences among children with high ADHD symptoms. Psych Res 2019;272:765-73.

7. Ball G, Malpas CB, Genc S, Efron D, Sciberras E, Anderson V, et al. Multimodal structural neuroimaging markers of brain development and ADHD symptoms. Am J Psychiatry 2019;176:57-66.


8. Mohammad MB, Seghinsara AM. Polycystic ovary syndrome (PCOS), diagnostic criteria, and AMH. Asian Pac J Cancer Prev 2017;18:17-21.


9. Bell GA, Sundaram R, Mumford SL, Park H, Mills J, Bell EM, et al. Maternal polycystic ovarian syndrome and early offspring development. Hum Reprod 2018;33:1307-15.



10. Kosidou K, Dalman C, Widman L, Arver S, Lee BK, Magnusson C, et al. Maternal polycystic ovary syndrome and risk for attention-deficit/hyperactivity disorder in the offspring. Biol Psychiatry 2017;82:651-9.


11. Hergüner S, Harmancı H, Toy H. Attention deficit-hyperactivity disorder symptoms in women with polycystic ovary syndrome. Int J Psychiatry Med 2015;50:317-25.


12. Katsigianni M, Karageorgiou V, Lambrinoudaki I, Siristatidis C. Maternal polycystic ovarian syndrome in autism spectrum disorder: a systematic review and meta-analysis. Mol Psychiatry 2019;24:1787-97.


13. Peters JP, Hooft L, Grolman W, Stegeman I. Reporting quality of systematic reviews and meta-analyses of otorhinolaryngologic articles based on the PRISMA statement. PLoS One 2015;10:e0136540.



14. Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 2004;81:19-25.

15. Bell AS. A critical review of ADHD diagnostic criteria: what to address in the DSM-V. J Atten Disord 2011;15:3-10.


16. Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses [Internet]. Ottawa (ON): Ottawa Hospital Research Institute; 2021 [cited 2020 Nov 20]. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
17. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629-34.



18. Berni TR, Morgan CL, Berni ER, Rees DA. Polycystic ovary syndrome is associated with adverse mental health and neurodevelopmental outcomes. J Clin Endocrinol Metab 2018;103:2116-25.


19. Cesta CE, Öberg AS, Ibrahimson A, Yusuf I, Larsson H, Almqvist C, et al. Maternal polycystic ovary syndrome and risk of neuropsychiatric disorders in offspring: prenatal androgen exposure or genetic confounding? Psychol Med 2020;50:616-24.


20. Chen X, Kong L, Piltonen TT, Gissler M, Lavebratt C. Association of polycystic ovary syndrome or anovulatory infertility with offspring psychiatric and mild neurodevelopmental disorders: a Finnish populationbased cohort study. Hum Reprod 2020;35:2336-47.



21. Robinson SL, Ghassabian A, Sundaram R, Trinh MH, Bell EM, Mendola P, et al. The associations of maternal polycystic ovary syndrome and hirsutism with behavioral problems in offspring. Fertil Steril 2020;113:435-43.



22. Antshel KM, Zhang-James Y, Wagner KE, Ledesma A, Faraone SV. An update on the comorbidity of ADHD and ASD: a focus on clinical management. Expert Rev Neurother 2016;16:279-93.


23. Maliqueo M, Lara HE, Sánchez F, Echiburú B, Crisosto N, Sir-Petermann T. Placental steroidogenesis in pregnant women with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol 2013;166:151-5.


24. Maliqueo M, Sundström Poromaa I, Vanky E, Fornes R, Benrick A, Åkerud H, et al. Placental STAT3 signaling is activated in women with polycystic ovary syndrome. Hum Reprod 2015;30:692-700.


25. Kjerulff LE, Sanchez-Ramos L, Duffy D. Pregnancy outcomes in women with polycystic ovary syndrome: a meta-analysis. Am J Obstet Gynecol 2011;204:558.e1-6.


26. Xiang AH, Wang X, Martinez MP, Getahun D, Page KA, Buchanan TA, et al. Maternal gestational diabetes mellitus, type 1 diabetes, and type 2 diabetes during pregnancy and risk of ADHD in offspring. Diabetes Care 2018;41:2502-8.


27. Maher GM, Dalman C, O’Keeffe GW, Kearney PM, McCarthy FP, Kenny LC, et al. Association between preeclampsia and attention-deficit hyperactivity disorder: a population-based and sibling-matched cohort study. Acta Psychiatr Scand 2020;142:275-83.


28. Jenabi E, Bashirian S, Khazaei S, Basiri Z. The maternal prepregnancy body mass index and the risk of attention deficit hyperactivity disorder among children and adolescents: a systematic review and meta-analysis. Korean J Pediatr 2019;62:374-9.








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


