Article Contents
| Korean J Pediatr > Volume 59(9); 2016 |
|
Abstract
The menarcheal age of Korean women has been rapidly decreasing for the last 50 years, and the average menarcheal age of women born in the 1990s is approaching 12.6 years. In addition, interest in early puberty has been increasing recently owing to the rapid increase in precocious puberty. Generally, out of concern for short stature and early menarche, idiopathic central precocious puberty in female adolescents is treated with gonadotropin-releasing hormone analogs. Studies to date have described the association between early menarche and psychosocial problems such as delinquency and risky sexual behavior, as well as physical health problems such as obesity, diabetes, cardiovascular diseases, and breast cancer throughout the lifespan of women. However, the pathophysiological mechanism underlying this association has not been clarified thus far. In this article, we review and discuss the existing literature to describe the current understanding of the effects of early menarche on the physical and psychosocial health of adolescent girls and adult women.
The menarcheal age of Korean women has been rapidly decreasing for the last 50 years, and the average menarcheal age of women born in the 1990s is approaching 12.6 years1). In addition, interest in early puberty has been increasing recently in Korea owing to the rapid increase in precocious puberty2). As central precocious puberty leads to early menarche, treatment is necessary to stop or prevent regular cyclical menstruation. Moreover, gonadotropin-releasing hormone analog treatment is used to promote increase in height because early skeletal maturation in this condition diminishes the final adult height3). Women who experience very early puberty, for example, precocious puberty or menarche earlier than the average age, are known to be at a greater risk of psychosocial problems4) such as delinquency and risky sexual behavior, as well as physical health problems5,6,7,8) such as obesity, diabetes, cardiovascular disease, and breast cancer. In addition, studies related to these topics have been actively conducted in Korean adolescents and adult women. The author reviewed these studies and hopes that this review article provides the opportunity to comprehensively examine physical and psychosocial health problems that may occur in women's lifetimes due to early puberty. In addition, based on recent studies, the author proposes feasible alternatives that can be helpful for the prevention of problems in adolescents who have experienced early puberty.
The use of cigarettes, alcohol, and marijuana is more prevalent in adolescents who experience early puberty than in those who experience either average- or late-age puberty9), in spite of the slight differences in national and community characteristics10). Internalizing problems such as depression, anxiety, bulimia, and excessive psychosomatic symptoms, as well as externalizing symptoms such as substance abuse, smoking, bullying, and truancy, are also more prevalent in adolescent girls who experience early puberty11). However, in case of problems such as substance abuse, social isolation, sexual behavior, and mental distress in adolescents who experience early puberty or early menarche, the difference decreases over 4–5 years of the puberty period as adolescents who experience average- or late-age puberty quickly catch up4,12). However, substance abuse related to alcohol, smoking, or illegal drugs in girls up to 15 years of age was more common among adolescent girls who had their menarche before 12 years of age than those who had it after 12 years of age. The difference narrowed at the age of 17 years13). Delinquency and physical aggression in adolescent girls who experienced early puberty reportedly increased at around 11 years of age when menarche occurred and delinquency continued, while physical aggression decreased during puberty. As adolescents at this age have a close association with the deviant behavior of their best friends, they might be vulnerable to negative peer influences14). Therefore, it is necessary to prevent and intensively manage girls in elementary school, that is, 2–3 years before menarche, which is the most sensitive time when adolescent girls are expected to have an early menarche and puberty are exposed to risky behavior. Currently, smoking prevention educational programs funded by the Korean Ministry of Health and Welfare are implemented for first- to third-grade elementary school students, emphasizing the harmfulness of second-hand smoking15). Fourth- to sixth-grade students are educated about the direct physical damage caused by smoking. Specific educational programs on alcohol or substance abuse are still targeted at middle school level and above. Step-by-step educational programs are needed in accordance with the developmental stage of puberty. Students with early puberty are thought to require education about the direct physical damage caused by smoking from early elementary school. Parallel development of educational materials that can be easily understood is also important. Educational programs during elementary school to prevent alcohol and substance abuse are also believed to be required for students with early puberty.
A recent meta-analysis showed that more girls who experienced early puberty engaged in sexual behavior such as sexual intercourse, noncoital sexual behavior (petting, kissing, caressing, and oral sex), and risky sexual behavior (unwanted pregnancy, contracting sexually transmitted infections/human immunodeficiency virus, noncondom/contraception use, and drug/alcohol use during sex) compared with boys16). A similar result was found in a study of Korean adolescents17), which used the eighth Korean Youth Risk Behavior Web-based survey (Table 1). The prevalences of intimate sexual relationships (kissing or petting), sexual intercourse, unprotected intercourse, and pregnancy were 1.54 times (95% confidence interval [CI], 1.28–1.87), 2.35 times (95% CI, 1.65–3.36), 1.92 times (95% CI, 1.06–3.46), and 5.72 times higher (95% CI, 2.31–14.15), respectively, in adolescents who had a very early menarche (before or during grade 4) than in those who had menarche after the fifth grade in elementary school. Surprisingly, the prevalences of intimate homosexual sexual relationships and intercourse were also 3.53 times (95% CI, 2.22–5.59) and 7.70 times higher (95% CI, 4.04–14.66), respectively. In addition, the risk of being the victim and the assailant in sexual assaults was 2.89 times (95% CI, 1.98–4.22) and 13.55 times higher (95% CI, 6.61–27.78), respectively. These phenomena seem to be the extreme cross-section of "peer socialization," which is a characteristic of adolescents18). Girls whose sexual maturity has begun tend to seek a group or friends who have a similar degree of sexual maturity and influence each other's behaviors; thus, they are exposed to the risks of sexual and risky behaviors. Therefore, to protect adolescents who are sexually mature but unprepared to face the corresponding experience, a social safety network tailored to the sexual development stages of children and adolescents should be established.
Early menarche can be used as an indicator of early sexual maturation. High blood estradiol levels and low sex hormone-binding globulin levels during puberty are known to increase adiposity19). Therefore, interest in this subject has increased, as adolescents and young women who experience early menarche are reported to have a higher body mass index (BMI)20).
A study of 8,080 women aged 15 years or older that was performed using the Canadian Community Health Survey reported that the mean BMI decreased by approximately 0.5 kg/m2 as the menarcheal age was delayed by 1 year5). A study based on the fourth Korea National Health and Nutrition Examination Survey (2007–2009) reported that the odds ratio of obesity in women who had menarche before 12 years of age was 1.845 times higher (95% CI, 1.441–2.361) than in those who had menarche at age 12 years or older1).
Freedman et al.21) reported that the association between early menarcheal age and adult obesity might have been slightly overestimated and may be more closely associated with childhood obesity. A study of 794 young Finns reported that a higher childhood BMI was associated with early menarche and might contribute to higher adult BMI and increased cardiovascular risk22). Pierce et al.23,24) claimed that the association between early menarcheal age and adult obesity is difficult to explain when considering BMI during ages between 4 and 6 years, and early menarcheal age is associated with a higher BMI between the age of 7 and 15 years. Although the clinically significant period is not clear, obesity in adolescents appears to be strongly associated with early menarche and chronic metabolic diseases such as diabetes. The results from a series of studies reported a strong association between the age at early menarche and the development of diabetes. However, the analysis of specific factors in some studies indicated that adult BMI or adiposity was reportedly the most influential factor6,25), whereas others indicate that the history of early menarche itself rather than the current BMI or adiposity was a more influential factor26,27).
A similar result was found in a study using data from the fourth Korea National Health and Nutrition Examination Survey (2007–2009)28). The prevalence of type 2 diabetes among Korean middle-aged women (20–50 years of age) was 3.61 times higher (95% CI, 1.90–6.88) in women who had menarche before 12 years of age than in those who had it later. Central obesity (waist circumference ≥85 cm), obesity (BMI >25 kg/m2), and insulin resistance were 1.83 times (95% CI, 1.38–2.44), 2.02 times (95% CI, 1.55–2.64), and 1.80 times higher (95% CI, 1.33–2.45), respectively, in women who had menarche before 12 years of age. In a study by Baek et al.29), the prevalences of dysglycemia, prediabetes, and diabetes were 1.85 times (95% CI, 1.28–2.66), 1.80 times (95% CI, 1.24–2.61), and 2.43 times higher (95% CI, 1.04–5.69), respectively, in middle-aged Korean women who had menarche before 13 years of age than in women who had menarche at the mean age (Fig. 1).
The Kanghwa cohort study investigated the risk of coronary heart disease mortality in 3,257 postmenopausal women older than the age of 55 years from 1985 to 2005. It reported that the risk of coronary heart disease mortality in those who had menarche after 17 years of age was 51% lower than that in those who had menarche between the age of 10 and 16 years30). Although it suggests an association between early menarcheal age and the risk of coronary heart disease, the age range was too broad.
Among 19,462 people who participated in a cohort study of Californian Seventh-day Adventist women from 1976 to 1998, total mortality and the incidences of ischemic heart disease and stroke increased in women who had menarche before the age of 11 years. A delay in menarche by 1 year reduced the total mortality by 4.5%, ischemic heart disease mortality by 6.0%, and stroke mortality by 8.6%31). The association with cardiometabolic risk factors such as dyslipidemdia, hypertension, high BMI and waist circumference, and diabetes mellitus, or mortality from ischemic heart disease was not sufficiently explained in the study.
A population-based prospective study of 15,807 UK-based women from 1993 to 1997 investigated the association between menarcheal age, and cardiovascular disease events and mortality based on follow-up results until 200732). They reported that the risk of cardiovascular disease events and mortality increased in women older than 40 years who had an early menarche. Early menarche is associated with metabolic syndrome components such as higher BMI, waist circumference, blood pressure, glycated hemoglobin, and a worse lipid profile affected by adult adiposity. When menarche occurred between 8 and 11 years of age, the risk of developing hypertension was 1.13 times (95% CI, 1.02–1.24), cardiovascular disease incidents were 1.17 times (95% CI, 1.07–1.27), coronary heart disease incidents were 1.23 times (95% CI, 1.06–1.43), cardiovascular disease mortality was 1.28 times (95% CI, 1.02-1.62), and all-cause mortality was 1.22 times higher (95% CI, 1.07–1.39) than that when menarche occurred after 12 years old.
Recently, the largest-scale study so far on menarcheal age and the risk of developing coronary heart and other vascular diseases in 1.2 million women was published7). In that study, the risk of coronary heart disease incidents was lowest for a menarcheal age of 13 years, and gradually increased for lower or higher menarcheal age, resulting in a U-shaped relationship. The risk was highest in the groups that had menarche at ≤10 and ≥17 years of age, with relative risks of 1.27 (95% CI, 1.22–1.31; P<0.0001) and 1.23 (95% CI, 1.16–1.30; P<0.0001), respectively (Fig. 2). The association between the risk of cerebrovascular disease incidents and menarcheal age, though weak, was similar to the result found for coronary heart disease (Fig. 3). Compared with the group with a menarcheal age of 13 years, the risk was highest in the groups with a menarcheal age of ≤10 and ≥17 years, with a relative risk of 1.16 (95% CI, 1.09–1.24) and 1.13 (95% CI, 1.03–1.24), respectively. Although one study suggested that late menarche may be associated with endothelial dysfunction33), this study could not clearly explain the increased risk of coronary heart disease in women who had a late menarcheal age. The subjects in the study were divided into groups of lean, overweight, and obese women; current, past, and never smokers; and low, middle, and high socioeconomic status. A U-shaped relationship was found for all subgroups. Thus, this study demonstrated that there is limited association between the increased risk of coronary heart disease in women with early menarche, and BMI, smoking, and socioeconomic status. Early menarcheal age was reportedly associated with hypertension and diabetes mellitus32). These risk factors may be important for the association of early menarche with cardiovascular and cerebrovascular diseases.
The association between early menarche and incidence of breast cancer has been reported extensively. This proves the hypothesis that earlier menarche results in a longer accumulated period of estrogen and progesterone exposure levels, lengthening the duration of breast mitotic activity and increasing the likelihood of tumor occurrence. An international case-control study found that breast cancer risk decreased by 20% when menarche was delayed by 2 years8). Germline mutations of BRCA1 are highly associated with the development of breast cancer, and breast cancer risk among BRCA1 carriers in women who had menarche at the age of 14–15 years was 54% lower than that in women who had menarche before the age of 11 years. This result suggests that early menarche is a strong risk factor of breast cancer34). However, in the International BRCA1/2 Carrier Cohort Study, wherein 1,601 BRCA1/2 carriers were divided into the early menarche group with a menarcheal age of ≤11 years, the normal menarche group with a menarcheal age of 12–14 years, and the delayed menarche group with a menarcheal age of ≥15 years, no difference was found between the BRCA1/2 carrier groups. In addition, no significant differences in the lifetime duration of breast mitotic activity, menstrual cycle, and breast cancer risk were found in cases of decreased breast mitotic activity such as full-term pregnancy after menarche and in oral contraceptive users, in contradiction with previous results35). Therefore, the influence of early menarche is thought to be relatively small in the context of family history of breast cancer.
A meta-analysis of 117 epidemiological studies, including more than 110,000 people, showed that the breast cancer incidence risk increased by 5% when menarche was 1 year early. While breast cancer did not develop at an earlier age when menarche occurred at an earlier age, the correlation between breast cancer incidence and menarcheal age was high. The delay in the menopausal age becomes a risk factor of breast cancer, as it also lengthens a woman's reproductive period; the risk was increased by 2.9% when menopause was delayed by 1 year. Therefore, early menarche was reportedly a stronger breast cancer risk factor than delayed menopausal age. This might be because early menarche has a much higher correlation than early menopause, with other breast cancer risk factors such as parity, age at first birth, height, BMI, and adiposity36).
The number of ovulatory cycles was also thought to play an important role in the pathogenesis of endometrial cancers such as breast cancer. The association between the development of endometrial cancer and reproductive characteristics such as early menarche, late menopause, and nulliparity has been studied. A meta-analysis of eight prospective studies with 4,553 endometrial cancer patients was conducted. The analysis revealed that the endometrial cancer risk in the group with the highest menarcheal age was 32% lower than that in the group with the lowest menarcheal age and confirmed that the cancer risk decreased by 4% when menarche was delayed by 2 years37).
A study of 443,909 Korean women whose details were provided by the Korea Medical Insurance Corporation from 1993 to 1994 reported an association between the risk of 10 major cancers and the age at menarche after a 12-year follow-up. In the study, the breast cancer risk in women who had menarche before the age of 15 years was 1.52 times higher (95% CI, 1.36–1.70) than that in women who had menarche after the age of 17 years. When only postmenopausal women were analyzed, the breast cancer risk in women who had menarche before the age of 15 years was 1.76 times higher (95% CI, 1.44–2.14) than that in women who had menarche after the age of 17 years. Moreover, the risk in women who had menopause after 55 years of age was 1.80 times higher (95% CI, 1.31–2.49) than that in women who had menopause before 45 years of age. No association was found between the development of uterine cervix, ovary, or thyroid cancer and early menarche or late menopause38). This study could not support the conclusion that early menarcheal age is associated with an increase in cancer-related mortality. However, a study on early menarcheal age and mortality in British women revealed that cancer mortality was 1.25 times higher (95% CI, 1.03–1.51) in women who had menarche at the age of 8–11 years and decreased by 5% when menarche was delayed by 1 year32).
Recently, Lazar et al.39) reported the results of a long-term follow-up study on young adults with precocious puberty, the most severe form of early menarche. They found no difference in the risk of obesity, metabolic derangements, or cancer morbidities between gonadotropin-releasing hormone analog-treated, untreated, and control subjects. Because the control subjects were relatively younger and fewer in number, studies with a larger number of patients with longer follow-up are needed. Therefore, it is too early to conclude that gonadotropin-releasing hormone analog treatment for adolescent girls who experience central precocious puberty or early puberty can reduce the risk of obesity, metabolic derangements, or cancer morbidities.
Studies to date describe the association between early menarche and psychosocial problems, as well as physical health problems, over the entire lifespan of women, but they do not provide a clear pathological mechanism that can explain this association. Nevertheless, a comprehensive and easily understood psychosocial and sexual health education program should be developed to educate adolescents with early menarche. In addition, lifestyle changes would be critical for preventing obesity and metabolic syndromes, and regular screening for cancer prevention is important not only for adolescents but also for young women.
Notes
Conflicts of interest:
No potential conflict of interest relevant to this article was reported.
References
1. Ahn JH, Lim SW, Song BS, Seo J, Lee JA, Kim DH, et al. Age at menarche in the Korean female: secular trends and relationship to adulthood body mass index. Ann Pediatr Endocrinol Metab 2013;18:60–64.



2. Health insurance Review & Assessment Service. The early detection of precocious puberty is important [internet]. Seoul: Health insurance Review & Assessment Service, 2013;cited 2015 Sep 10. available from: http://www.hira.or.kr.
3. Kwon EB, Lee SJ, Cha M, Kim SY. Changes in the predicted adult height after gonadotropin-releasing hormone agonist treatment in girls with idiopathic true precocious puberty. Ann Pediatr Endocrinol Metab 2012;17:160–168.

4. Lien L, Haavet OR, Dalgard F. Do mental health and behavioural problems of early menarche persist into late adolescence? A three year follow-up study among adolescent girls in Oslo, Norway. Soc Sci Med 2010;71:529–533.


5. Harris MA, Prior JC, Koehoorn M. Age at menarche in the Canadian population: secular trends and relationship to adulthood BMI. J Adolesc Health 2008;43:548–554.


6. Lakshman R, Forouhi N, Luben R, Bingham S, Khaw K, Wareham N, et al. Association between age at menarche and risk of diabetes in adults: results from the EPIC-Norfolk cohort study. Diabetologia 2008;51:781–786.


7. Canoy D, Beral V, Balkwill A, Wright FL, Kroll ME, Reeves GK, et al. Age at menarche and risks of coronary heart and other vascular diseases in a large UK cohort. Circulation 2015;131:237–244.


8. Hsieh CC, Trichopoulos D, Katsouyanni K, Yuasa S. Age at menarche, age at menopause, height and obesity as risk factors for breast cancer: associations and interactions in an international case-control study. Int J Cancer 1990;46:796–800.


9. Cance JD, Ennett ST, Morgan-Lopez AA, Foshee VA, Talley AE. Perceived pubertal timing and recent substance use among adolescents: a longitudinal perspective. Addiction 2013;108:1845–1854.



10. Al-Sahab B, Ardern CI, Hamadeh MJ, Tamim H. Age at menarche and current substance use among Canadian adolescent girls: results of a cross-sectional study. BMC Public Health 2012;12:195



11. Kaltiala-Heino R, Marttunen M, Rantanen P, Rimpelä M. Early puberty is associated with mental health problems in middle adolescence. Soc Sci Med 2003;57:1055–1064.


12. Copeland W, Shanahan L, Miller S, Costello EJ, Angold A, Maughan B. Outcomes of early pubertal timing in young women: a prospective population-based study. Am J Psychiatry 2010;167:1218–1225.



13. Kaltiala-Heino R, Koivisto AM, Marttunen M, Fröjd S. Pubertal timing and substance use in middle adolescence: a 2-year follow-up study. J Youth Adolesc 2011;40:1288–1301.


14. Mrug S, Elliott MN, Davies S, Tortolero SR, Cuccaro P, Schuster MA. Early puberty, negative peer influence, and problem behaviors in adolescent girls. Pediatrics 2014;133:7–14.



15. Ministry of Education. Student Health Information Center [Internet]. Seoul: Ministry of Education, Student Health Information Center, c2010;cited 2015 Nov 15. Available from: http://www.schoolhealth.kr/.
16. Baams L, Dubas JS, Overbeek G, van Aken MA. Transitions in body and behavior: a meta-analytic study on the relationship between pubertal development and adolescent sexual behavior. J Adolesc Health 2015;56:586–598.


17. Cheong JI, Lee CH, Park JH, Ye BJ, Kwon KA, Lee YS, et al. The effect of early menarche on the sexual behaviors of Korean female adolescents. Ann Pediatr Endocrinol Metab 2015;20:130–135.



18. Stattin H, Kerr M, Skoog T. Early pubertal timing and girls' problem behavior: integrating two hypotheses. J Youth Adolesc 2011;40:1271–1287.


19. Tchernof A, Després JP. Sex steroid hormones, sex hormone-binding globulin, and obesity in men and women. Horm Metab Res 2000;32:526–536.


20. Laitinen J, Power C, Järvelin MR. Family social class, maternal body mass index, childhood body mass index, and age at menarche as predictors of adult obesity. Am J Clin Nutr 2001;74:287–294.


21. Freedman DS, Khan LK, Serdula MK, Dietz WH, Srinivasan SR, Berenson GS, et al. The relation of menarcheal age to obesity in childhood and adulthood: the Bogalusa heart study. BMC Pediatr 2003;3:3



22. Kivimäki M, Lawlor DA, Smith GD, Elovainio M, Jokela M, Keltikangas-Järvinen L, et al. Association of age at menarche with cardiovascular risk factors, vascular structure, and function in adulthood: the Cardiovascular Risk in Young Finns study. Am J Clin Nutr 2008;87:1876–1882.


23. Pierce MB, Leon DA. Age at menarche and adult BMI in the Aberdeen children of the 1950s cohort study. Am J Clin Nutr 2005;82:733–739.


24. Pierce MB, Kuh D, Hardy R. The role of BMI across the life course in the relationship between age at menarche and diabetes, in a British Birth Cohort. Diabet Med 2012;29:600–603.



25. Chen L, Zhang C, Yeung E, Ye A, Mumford SL, Wactawski-Wende J, et al. Age at menarche and metabolic markers for type 2 diabetes in premenopausal women: the BioCycle Study. J Clin Endocrinol Metab 2011;96:E1007–E1012.



26. Elks CE, Ong KK, Scott RA, van der Schouw YT, Brand JS, Wark PA, et al. Age at menarche and type 2 diabetes risk: the EPIC-InterAct study. Diabetes Care 2013;36:3526–3534.



27. Stöckl D, Döring A, Peters A, Thorand B, Heier M, Huth C, et al. Age at menarche is associated with prediabetes and diabetes in women (aged 32-81 years) from the general population: the KORA F4 Study. Diabetologia 2012;55:681–688.


28. Lim JS, Lee HS, Kim EY, Yi KH, Hwang JS. Early menarche increases the risk of Type 2 diabetes in young and middle-aged Korean women. Diabet Med 2015;32:521–525.


29. Baek TH, Lim NK, Kim MJ, Lee J, Ryu S, Chang Y, et al. Age at menarche and its association with dysglycemia in Korean middle-aged women. Menopause 2015;22:542–548.



30. Chang HS, Odongua N, Ohrr H, Sull JW, Nam CM. Reproductive risk factors for cardiovascular disease mortality among postmenopausal women in Korea: the Kangwha Cohort Study, 1985-2005. Menopause 2011;18:1205–1212.


31. Jacobsen BK, Oda K, Knutsen SF, Fraser GE. Age at menarche, total mortality and mortality from ischaemic heart disease and stroke: the Adventist Health Study, 1976-88. Int J Epidemiol 2009;38:245–252.



32. Lakshman R, Forouhi NG, Sharp SJ, Luben R, Bingham SA, Khaw KT, et al. Early age at menarche associated with cardiovascular disease and mortality. J Clin Endocrinol Metab 2009;94:4953–4960.


33. Schnabel RB, Biener MP, Wilde S, Sinning CR, Ojeda FM, Zeller T, et al. Sex differences in noninvasive vascular function in the community. J Hypertens 2013;31:1437–1446.


34. Kotsopoulos J, Lubinski J, Lynch HT, Neuhausen SL, Ghadirian P, Isaacs C, et al. Age at menarche and the risk of breast cancer in BRCA1 and BRCA2 mutation carriers. Cancer Causes Control 2005;16:667–674.


35. Chang-Claude J, Andrieu N, Rookus M, Brohet R, Antoniou AC, Peock S, et al. Age at menarche and menopause and breast cancer risk in the International BRCA1/2 Carrier Cohort Study. Cancer Epidemiol Biomarkers Prev 2007;16:740–746.


36. Collaborative Group on Hormonal Factors in Breast Cancer. Menarche, menopause, and breast cancer risk: individual participant meta-analysis, including 118 964 women with breast cancer from 117 epidemiological studies. Lancet Oncol 2012;13:1141–1151.



37. Gong TT, Wang YL, Ma XX. Age at menarche and endometrial cancer risk: a dose-response meta-analysis of prospective studies. Sci Rep 2015;5:14051



Fig. 1
Prevalence of diabetes, prediabetes, and dysglycemia according to age at menarche. Adapted from Baek et al. Menopause 2015;22:542-8, with the permission of Wolters Kluwer Health, Inc.29).
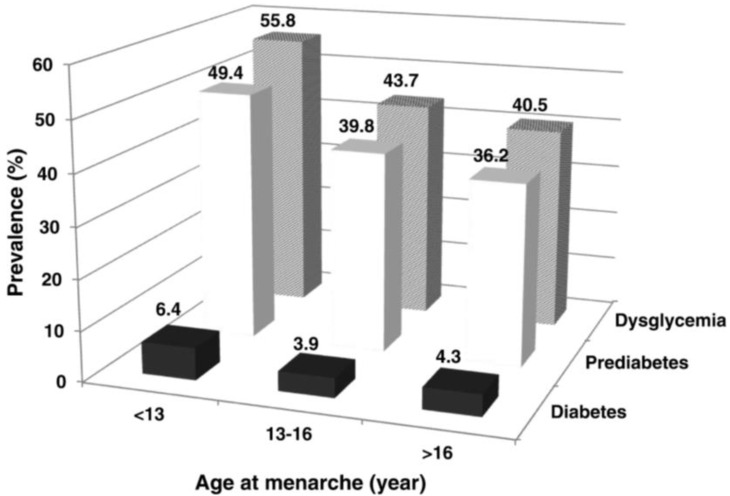
Fig. 2
Relative risk (RR) and 95% confidence interval (CI) of incident coronary heart disease (CHD) according to age at menarche. RRs (hazard ratios) are stratified by region of recruitment and adjusted for year of birth, body mass index, height, smoking, alcohol consumption, exercise, and socioeconomic status. The reference category is menarche at 13 years of age. The area of the square is inversely proportional to the variance of the log risk. ICD-10, International Classification of Diseases, 10th revision. Adapted from Canoy et al. Circulation 2015;131:237-44, with the permission of Wolters Kluwer Health, Inc.7).
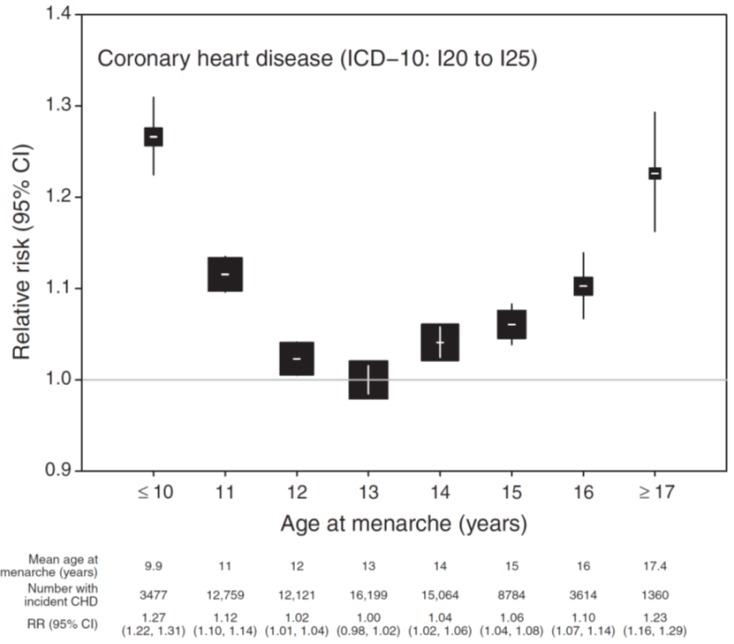
Fig. 3
Relative risk (RR) and 95% confidence interval (CI) of incident cerebrovascular disease (CeVD) according to age at menarche. RRs (hazard ratios) are stratified by region of recruitment and adjusted for year of birth, body mass index, height, smoking, alcohol consumption, exercise, and socioeconomic status. The reference category is menarche at 13 years of age. The area of the square is inversely proportional to the variance of the log risk. ICD-10, International Classification of Diseases, 10th revision. Adapted from Canoy et al. Circulation 2015;131:237-44, with the permission of Wolters Kluwer Health, Inc.7).
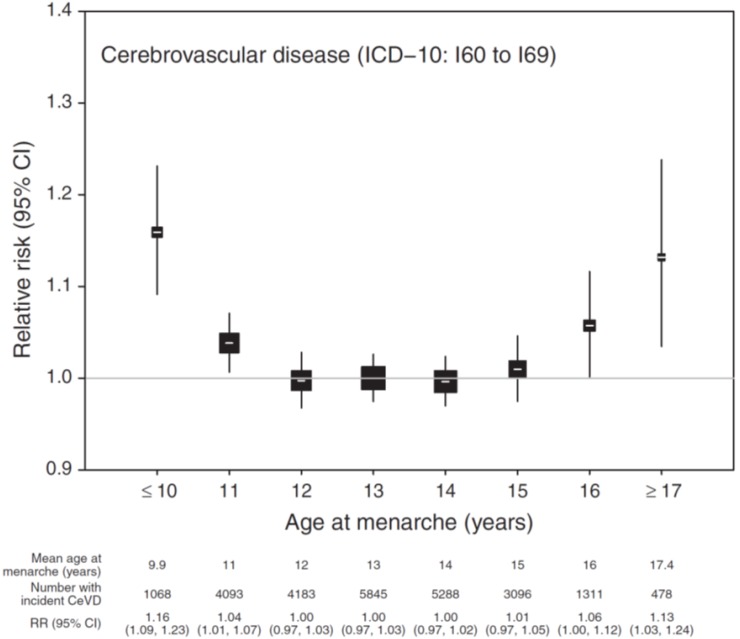
Table 1
Effects of early menarche on sexual behaviors from the Korean Youth Risk Behavior Web-based survey (2012)
Values are presented as number (%).
OR, odds ratio; CI, confidence interval.
*Adjusted for sociodemographic variables of paternal educational level, maternal educational level, living with parents, academic performance, and household income; comparison between the early and normal menarche groups.
Adapted from Cheong et al. Ann Pediatr Endocrinol Metab 2015;20:130-5, according to the open-access policy of The Korean Society of Pediatric Endocrinology17).
-
METRICS

-
- 36 Crossref
- 0 Scopus
- 13,275 View
- 330 Download



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation

