Introduction
Varicella-zoster virus (VZV) infection is a self-limited childhood disease1,2). However, it sometimes can be a life-threatening condition, especially in immunocompromised patients such as bone marrow transplant recipients, who are receiving corticosteroids therapy or chemotherapy and patients with human immune deficiency virus infection1,2,3,4,5,6,7,8). Although, it is very rare, VZV infection can be also lead to severe illness in immunocompetent persons2,3,6,7).
We report a case of primary VZV infection with very rare complications of acute respiratory distress syndrome (ARDS), acute hepatitis, disseminated intravascular coagulation (DIC), and encephalopathy in an 8-year-old boy undergoing chemotherapy for acute lymphoblastic leukemia (ALL).
Case report
An 8-year-old boy was admitted to The Catholic University of Korea, Seoul St. Mary's Hospital after experiencing one day of severe abdominal pain. He was diagnosed with ALL (L3) 4 years ago. After first relapse, the patient received a bone marrow transplant 3 years ago, but second relapse occurred unfortunately. It had been three weeks since his last chemotherapy, which included a high dose of methotraxate. He was no vaccinated against chickenpox following the bone marrow transplant.
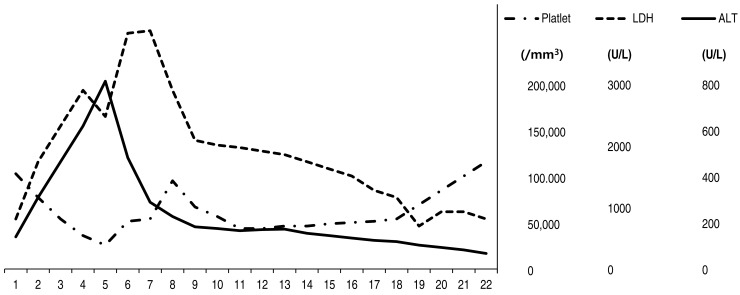
His body temperature was normal. He showed hepatosplenomegaly and tenderness in the periumbilical area. A complete blood count showed hemoglobin of 12.1 g/dL, white blood cells (WBCs) count of 3,490/mm3 with segmented neutrophils of 80.0%, and platelet count of 135,000/mm3. Liver function tests showed slightly increased levels of aspartate aminotransferase (AST, 78 U/L) and alanine aminotransferase (ALT, 135 U/L) (Fig. 1). Chest, abdomen x-rays and abdomen and pelvis computed tomography scans appeared normal.
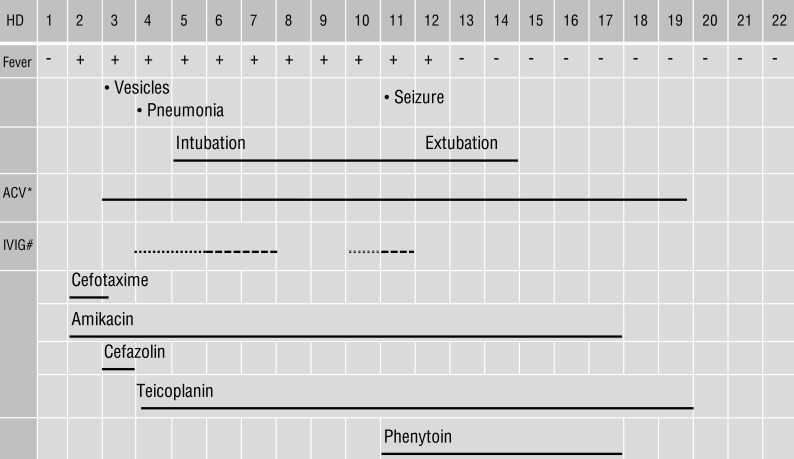
The next day, the patient was running a fever of up to 39.0℃, so empirical intravenous antibiotics were started. His abdominal pain remained constant. On his third day, multiple erythematous vesicles formed on his trunk. Consequently, 1,500 mg/m2 of acyclovir divided into three doses per day was administered.
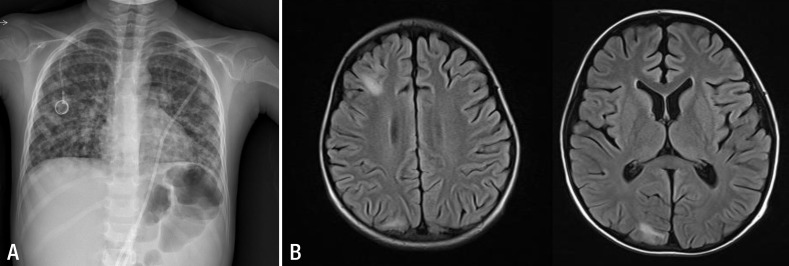
On hospital day 5, the vesicles developed across the whole body and he complained of dyspnea. A chest x-ray (Fig. 2A) showed numerous small nodules in the entire both lungs, which lead to a diagnosis of varicella pneumonia. Laboratory tests revealed WBCs of 5,090/mm3, platelet count of 32,000/mm3, AST of 931 U/L, ALT of 788 U/L, lactate dehydrogenase (LDH) of 3,196 U/L (Fig. 1). DIC profile was also positive. We continued administration of intravenous acyclovir, changed the intravenous antibiotics, and added intravenous immunoglobulin (IVIG, 500 mg/kg/day), single donor platelets, fresh frozen plasma, and antithrombin III. The patient was transferred to the intensive care unit (ICU).
The next evening, the patient began to show drowsy mental state and dyspnea was aggravated. A chest x-ray showed total haziness and ARDS. He required intubation and mechanical respiratory support. We increased the dose of IVIG to 1 g/kg/day.
On hospital day 7, a chest x-ray showed marked improvements in the lung, but cardiomegaly was noted. Platelet count was 69,000/mm3, and AST and ALT were 224 U/L and 280 U/L (Fig. 1). The patient was successfully recovered from DIC. We used IVIG for a total of four days, however fever persisted and the vesicular skin lesions showed no change. On echocardiography, cardiac function was normal.
On hospital day 9, the patient's blood pressure (BP) began to increase up to systolic pressure of 150 mm/Hg and diastrolic pressure of 125 mm/Hg. We continuously administered an intravenous labetarol, however no definite effect was observed. Using by real-time polymerase chain reaction (PCR), VZV was detected in patient serum that had had been sampled at the onset of skin lesions. We decided to administer IVIG for two more days.
On the morning of hospital day 11, the patient experienced a generalized tonic-clonic seizure that lasted 30 minutes which we thought was a sign of hypertensive crisis or VZV involvement in central nerve system. We added intravenous nitroprusside and conducted cerebrospinal fluid (CSF) tapping. The results were normal, and VZV antibody and PCR in CSF were negative. We increased the antihypertensives dose and added intravenous phenytoin. No more seizures were occurred thereafter.
By hospital day 13 and the ninth day after intubation, the patient's pulmonary condition was stable. He was successfully taken off the ventilator. He no longer had a fever, and all skin lesions reached the scarring stage.
On hospital day 17, the patient was transferred from the ICU to the general ward. His vital signs were stable. All laboratory data showed improvements. Lymphocyte subset showed CD3 of 87.0% (normal range [NR], 61%-85%), CD4 of 14.8% (NR, 23%-58%), CD8 of 70.6% (NR, 19%-48%), CD19 of 9.6% (NR, 7%-23%), and CD56 of 3.0%. (NR, 6%-29%). These results suggested that the patient's cellular immunity was not yet recovered. Due to the patient's history of seizure, we performed brain magnetic resonance imaging (MRI) and electroencephalogram (EEG). The brain MRI (Fig. 2B) showed multiple patchy high-signal-intensity lesions in the subcortical regions of the right frontal, parietal and occipital lobes as a results of VZV-induced encephalitis. The EEG looked normal, so phenytoin was stopped.
We ceased administration of intravenous antibiotics and acyclovir on hospital day 19. The patient complained of no other symptoms. He was discharged on hospital day as he had been restored to a healthy appearance and most laboratory data showed improvement (Fig. 1). The clinical course of patient was as follows (Fig. 3). One month later, chemotherapy was resumed. And the follow-up brain MRI was done three months later, multiple high-signal-intensity lesions due to VZV-induced encephalitis were disappeared.
Discussion
Most healthy children with varicella infection have mild illness. However, dissemination of the primary infection can occur, particularly in immunocompromised children1,2,3,4,5,6,7). The lungs, liver, brain and heart are usually involved in progressive varicella1). The mortality rate of disseminated varicella infection is extremely high in immunocompromised children because their clinical course is fulminant and atypical4,5).
Acyclovir is a highly potent inhibitor of VZV and early treatment with acyclovir has been shown to prevent dissemination and progression of disease9). However, acyclovir alone may not be able to fully fight disseminated VZV infection in immunocompromised patients4,5), because immunocompromised children, who have lower T-cell responses, cannot control VZV, resulting in both prolonged infection and reactivation9). Although, it is very rare, VZV resistance to acyclovir has been reported in these patients9).
In this case, in spite of administration of acyclovir, VZV infection rapidly progressed to ARDS. We added IVIG with mechanical respiratory support. The recommended dosage for sepsis is 400 mg/kg/day administered as intravenous infusion for five days10). However, a dosage of 500 mg/kg/day was not sufficient. After increasing the dosage to 1 g/kg/day, the patient had no fever or new skin lesions. IVIG provides antibodies that neutralize virus invasion and suppress viremia, acting synergistically with acyclovir4). Lu et al.4) used IVIG with acyclovir in an immunocompromised child with disseminated varicella that had resulted in multiorgan failure. Various adverse effects of IVIG like acute hypersensitivity, thromboembolism, renal failure, hypotension, aseptic meningitis and fluid overload are widely known11). However, most adverse effects associated with its administration are immediate, mild, and transient12). And severe late adverse effects like renal toxicity and thromboembolic events were closely related with rate of infusion and patient's underlying risk factors, not with dose12). We therefore suggest that clinicians should consider IVIG with acyclovir to treat patients with disseminated VZV infection.
The benefits of corticosteroid treatment in varicella pneumonia remain controversial. There have been no reports of corticosteroid treatment in immunocompromised children. We did not chose corticosteroid because, it has the potential to lower the host's resistance to infection and to suppress the host's immune system10).
In rare case, cidofovir was used in hepatic failure due to VZV for antiviral activity against acyclovir and foscarnet-resistant VZV-strains13). The efficacy of alternative viral drugs alone or in combination with standard antiviral substances needs individual decision-making for situations13). Varicella-zsoter immunoglobulin was also used some in solid-organ transplant recipients14).
In conclusion, disseminated varicella infection may cause high morbidity and mortality in immunocompromised children. However, early administration of acyclovir and high-dose of immunoglobulin may result in a positive outcome, as it did in this case.



 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


