Article Contents
| Clin Exp Pediatr > Volume 67(1); 2024 |
|
Abstract
Background
Purpose
Methods
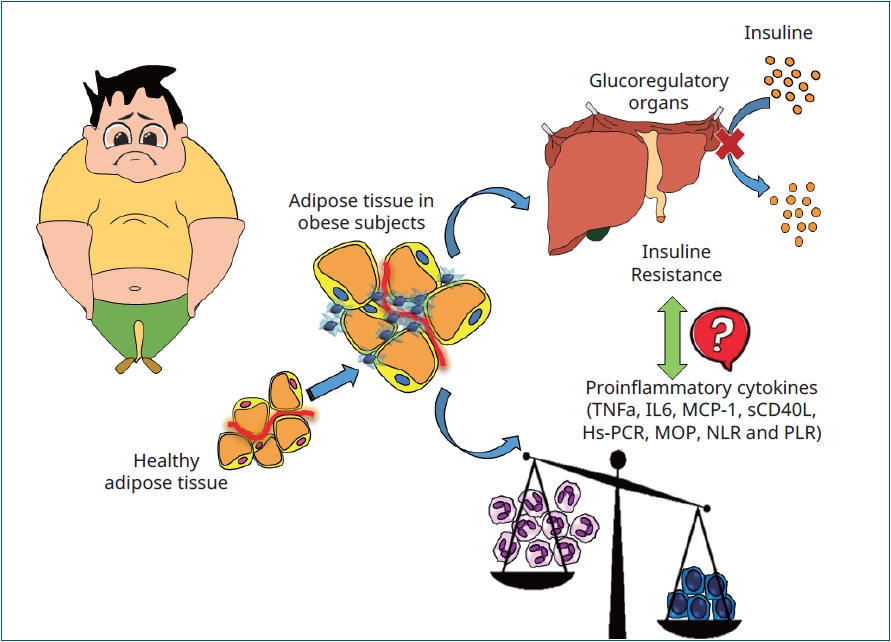
Results
Footnotes
Fig. 1.
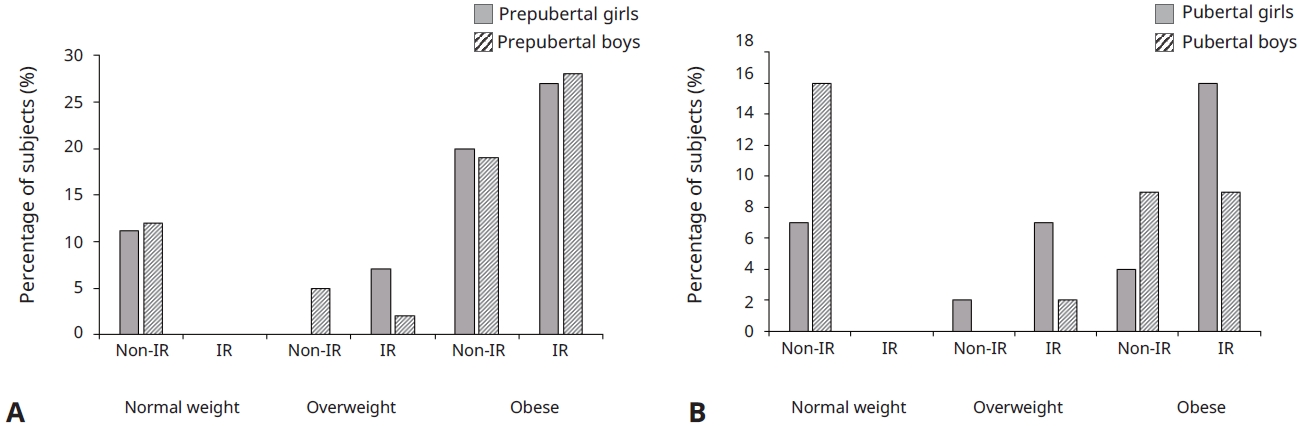
Table 1.
| Variable | Overweight (n=10) | Obesity (n=58) | Normal weight (n=20) | P value | β |
|---|---|---|---|---|---|
| Sex, male:female | 3:7 | 28:30 | 12:8 | 0.209 | 0.60 |
| Age (yr) | 11 (9–12) | 10 (9–12) | 12 (9–13) | 0.080 | 0.89 |
| BMI (kg/m2) | 25.38±2.90 | 29.85±9.07c) | 18.45±2.32c) | 0.001 | 0.90 |
| BMI z score | 1.79±0.34a,b) | 2.57±0.58b,c) | -0.14±0.78a,c) | 0.001 | 0.99 |
| Height z score | 1.17±0.63a) | 1.46±1.07c) | -0.59±0.67a,c) | 0.001 | 0.49 |
| WBCs (109/L) | 7.541±1.821 | 7.7818±1.709 | 7.394±2.259 | 0.745 | 0.80 |
| Neutrophil (109/L) | 4.059±1.388 | 4.062±1.219 | 3.685±1.594 | 0.524 | 0.70 |
| Lymphocyte (109/L) | 3.040±0.764 | 2.832±0.711 | 3.095±0.974 | 0.406 | 0.66 |
| Platelets (109/L) | 275.545±62.848 | 307.854±66.455 | 300.389±71.465 | 0.669 | 0.76 |
| FBG (mg/dL) | 74.17±12.61 | 76.61±11.60 | 77.44±8.23 | 0.733 | 0.79 |
| Insulin (μU/mL) | 20.66±13.64a) | 22.28±13.50c) | 7.15±1.06a,c) | 0.001 | 0.67 |
| HOMA | 4.32±2.98 | 4.26±2.75c) | 1.37±0.28c) | 0.001 | 0.95 |
| IR | 57% | 72% | - | - | - |
| TC (mg/dL) | 163±48 | 164±37 | 154±23 | 0.556 | 0.70 |
| HDL-C (mg/dL) | 40±10 | 39±8c) | 44±8c) | 0.050 | 0.70 |
| LDL-C (mg/dL) | 95±33 | 104±33 | 98±25 | 0.320 | 0.66 |
| Non-HDL-C (mg/dL) | 123±50 | 125±35 | 109±23 | 0.236 | 0.66 |
| Triglyceride (mg/dL) | 169±151 | 114±65c) | 72±18c) | 0.001 | 0.50 |
| ALT (U/L) | 14±4b) | 26±15b,c) | 14±7c) | 0.001 | 0.55 |
| AST (U/L) | 14±3 | 22±12c) | 14±8c) | 0.010 | 0.50 |
BMI, body mass index; WBC, white blood cell; FBG, fasting blood glucose; HOMA, homeostasis model assessment; IR, insulin resistance; TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; non-HDL-C, non–high-density lipoprotein cholesterol; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Values are presented as mean±standard deviation or median (interquartile range) unless otherwise indeciated.
Boldface indicates a statistically significant difference with P <0.05.
Table 2.
| Variable |
Boys |
P value | β |
Girls |
P value | β | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Overweight | Obesity | Normal weight | Overweight | Obesity | Normal weight | |||||
| No. | 3 | 28 | 12 | - | - | 7 | 30 | 8 | - | - |
| NLR | 1.74±0.50 | 1.54±0.47 | 1.15±0.48 | 0.053 | 0.54 | 1.34±0.53 | 1.50±0.87 | 1.46±0.67 | 0.881 | 0.89 |
| PLR | 114.42±33.59 | 118.28±31.25 | 97.21±44.21 | 0.258 | 0.62 | 88.55±22.33 | 111.42±43.56 | 121.29±34.26 | 0.327 | 0.61 |
| ESR (mm/hr) | 7±4 | 13±8c) | 4±3c) | 0.010 | 0.50 | 18±5 | 13±7 | 12±8 | 0.168 | 0.55 |
| MCP-1 (pg/mL) | 117.67±45.35 | 162.57±48.72c) | 93.18±12.42c) | 0.003 | 0.76 | 165.00±73.88 | 170.53±51.19c) | 96.28±8.65c) | 0.001 | 0.34 |
| TNF-α (pg/mL) | 19.23±8.62 | 17.95±6.00c) | 12.09±1.18c) | 0.017 | 0.40 | 17.90±6.72 | 17.90±6.73c) | 13.44±1.26c) | 0.012 | 0.14 |
| IL-6 (pg/mL) | 0.54±0.18b) | 2.34±2.43b,c) | 0.65±0.13c) | 0.028 | 0.46 | 1.98±2.70 | 2.30±2.53c) | 0.61±0.13c) | 0.008 | 0.10 |
| MPO (μU/mL) | 1.12±0.25 | 1.18±0.14 | 1.12±0.12 | 0.483 | 0.71 | 1.14±0.08 | 1.19±0.15 | 1.27±0.19 | 0.292 | 0.65 |
| hs-CRP (mg/L) | 4.27±4.82 | 3.09±2.82c) | 0.41±0.23c) | 0.013 | 0.49 | 2.50±2.25 | 2.67±2.35 | 0.55±0.24 | 0.076 | 0.56 |
| sCD40L (pg/mL) | 886.66±323.67 | 960.40±262.08c) | 110.91±15.62c) | 0.001 | 0.91 | 1066.43±408.82a) | 881.00±274.47c) | 108.28±17.24a,c) | 0.001 | 0.81 |
Values are presented as mean±standard deviation.
NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; ESR, erythrocyte sedimentation rate; MCP-1, monocyte chemoattractant protein-1; TNF-α: tumor necrosis factor-α; IL-6, interleukin 6; MPO, myeloperoxidase; hs-CRP, high-sensitivity C-reactive protein; sCD40L, soluble CD40 ligand.
Boldface indicates a statistically significant difference with P <0.05.
Table 3.
Values are presented as mean±standard deviation.
IR, insulin resistance; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; ESR, erythrocyte sedimentation rate; MCP-1, monocyte chemoattractant protein-1; TNF-α: tumor necrosis factor-α; IL-6, interleukin 6; MPO, myeloperoxidase; hs-CRP, high-sensitivity C-reactive protein; sCD40L, soluble CD40 ligand.
No significant intergroup differences were found, so a post hoc analysis was not necessary.
Table 4.
NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; ESR, erythrocyte sedimentation rate; MCP-1, monocyte chemoattractant protein-1; TNF-α, tumor necrosis factor-α; IL-6, interleukin-6; MPO, myeloperoxidase; hs-CRP, high-sensitivity C-reactive protein; sCD40L, soluble CD40 ligand.
Pearson correlation coefficient was applied.
Boldface indicates a statistically significant difference with P <0.05.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


