Article Contents
| Clin Exp Pediatr > Volume 66(12); 2023 |
|
Abstract
Nonalcoholic fatty liver disease (NAFLD), a spectrum of liver diseases characterized by excessive fat accumulation, is the leading cause of chronic liver disease. The global prevalence of NAFLD is increasing in both adults and children. In Korea, the prevalence of pediatric NAFLD increased from 8.2% in 2009 to 12.1% in 2018 according to a national surveillance study. For early screening of pediatric NAFLD, laboratory tests including aspartate aminotransferase and alanine aminotransferase; biomarkers including hepatic steatosis index, triglyceride glucose index, and fibrosis-4 index; and imaging studies including ultrasonography and magnetic resonance imaging are required. Insulin resistance plays a major role in the pathogenesis of NAFLD, which promotes insulin resistance. Thus, the association between NAFLD and insulin resistance, diabetes mellitus, and metabolic syndrome has been reported in many studies. This review addresses issues related to the epidemiology and investigation of NAFLD as well as the association between NAFLD and insulin resistance and metabolic syndrome with focus on pediatric NAFLD.
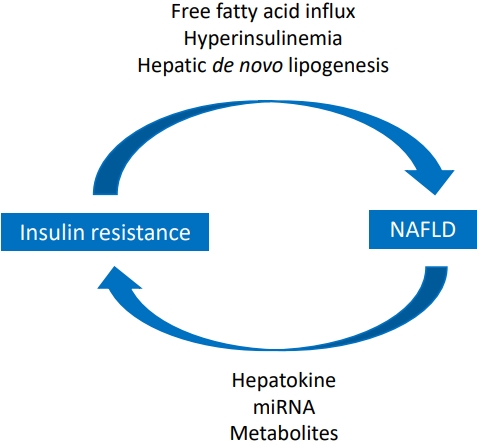
Graphical abstract. Relationship between insulin resistance and nonalcoholic fatty liver disease (NAFLD).
Nonalcoholic fatty liver disease (NAFLD) is an entire spectrum of liver disease induced by excessive fat accumulation in the liver, including simple steatosis, nonalcoholic steatohepatitis, and advanced fibrosis and cirrhosis in patients without known causes, including significant alcohol consumption [1,2]. NAFLD, one of the major causes of end-stage liver disease, is associated with cardiovascular risk factors, including insulin resistance and dyslipidemia [3]. Thus, NAFLD is related to all-cause mortality, including liver-related death, cardiovascular disease, and diabetes mellitus [4]. Moreover, NAFLD is the leading cause of chronic liver disease, and its prevalence is increasing globally [5].
Since Ludwig et al. [6] defined nonalcoholic steatohepatitis in 1980 as liver tissue injury similar to alcoholic hepatitis among patients who did not drink alcohol, there have been numerous investigations into NAFLD. In particular, many recent studies considered NAFLD a multisystem disease focusing on the association between NAFLD and insulin resistance [4,7-9]. Furthermore, renaming NAFLD as metabolic-associated fatty liver disease (MAFLD) has been suggested based on the evidence of no safe limitation for alcohol use in cases of fatty liver [10].
However, there are limited publications in the current literature regarding the relationship between NAFLD and insulin resistance in children. This review discussed the epidemiology and investigation of NAFLD and the association between NAFLD and insulin resistance with a primary focus on pediatric NAFLD.
A meta-analysis reported that the global prevalence of NALFD increased from 21.9% in 1991 to 37.3% in 2019, while the adverse trend was more prominent in South America and Europe [11]. In the United States, the prevalence of NAFLD was 24% in a meta-analysis [12]. In Korea, the prevalence of NAFLD was 31% among men and 22% among women [13]. During the coronavirus disease 2019 (COVID-19) outbreak, the proportion of newly diagnosed NAFLD cases was 25% among adults in a health checkup program in a Japanese study [14]. A Canadian study reported that the prevalence of MAFLD was 55.3% among participants with post-acute COVID-19 syndrome [15].
Among children and adolescents, the global prevalence of NAFLD increased from 19.3 million in 1990 to 29.5 million in 2017, being higher in developed than developing regions [16]. In another global study, the prevalence of NAFLD was 7.6% among all youth and 34.2% among youth with obesity [17]. In the US, the NAFLD prevalence among all youth increased from 3.9% in 1988–1994 to 10.7% in 2007–2010 [18]. In China, the NAFLD prevalence was 9.0% in 2009 among all youth [19]. In Korea, the prevalence of pediatric NAFLD, defined as an alanine aminotransferase (ALT) elevation without hepatitis B virus or hepatitis C virus infection, increased from 8.2% in 2009 to 12.1% in 2018 in a national surveillance study [1]. Moreover, the adverse trend was more prominent among youth, even young children, with a normal body mass index (BMI). In another Korean populationbased study, the prevalence of NAFLD was 14.7% in boys and 6.5% in girls [20]. In a study of the Korean health checkup program performed in Seoul, the prevalence of NAFLD diagnosed on ultrasonography was 11.2% among youth [21]. Further studies are required to clarify changes in the prevalence of pediatric NAFLD during the COVID-19 outbreak.
Early screening for NAFLD is important for preventing severe complications because NAFLD is usually asymptomatic and reversible in the early stages with proper management [21]. Although liver biopsy is the gold standard for the diagnosis of NAFLD, it is very invasive [3,22]. Moreover, its results cannot represent the entire liver because the sample is limited to a small part of the liver. Thus, laboratory tests and imaging studies are required for the early detection of pediatric NAFLD [20,21,23].
ALT level has acceptable sensitivity for detecting NAFLD, although it is not correlated with the histologic grade of NAFLD [3,24,25]. Moreover, some children with NAFLD have normal ALT and ALT levels, which can be elevated in other liver diseases [3,26]. Meanwhile, aspartate aminotransferase (AST) level is correlated with hepatic fibrosis progression, although it has not been suggested as an independent screening tool for NAFLD in children [3,24,25].
The North American Society of Pediatric Gastroenterology, Hepatology, and Nutrition guidelines recommend ALT as a screening test due to its minimal invasiveness and adequate sensitivity [3]. The guidelines suggest NAFLD screening for obese children aged 9–11 years and overweight children with risk factors such as central obesity, insulin resistance, dyslipidemia, sleep apnea, and/or family history of NAFLD. Further evaluations for NAFLD are suggested in children with ALT levels higher than the upper limit value for longer than 3 months, suggested as 26 U/L for boys and 22 U/L for girls based on the National Health and Nutrition Examination Survey [27]. A Canadian study suggested the upper limit of ALT as 30 mg/dL in children 1–12 years old and 24 mg/dL in those aged 13–19 years [28]. In Korea, the 95th percentile value for ALT level was suggested as 24.1 U/L for boys and 17.7 U/L for girls in a population-based study [29]. Another Korean study suggested an upper limit of ALT of 39 U/L for boys and 31 U/L for girls and those of AST as 39 U/L for boys and 41 U/L for girls aged 2–7 years and 34 U/L for girls aged 8–14 years [30].
To overcome the limitations of liver biopsy and laboratory tests, various parameters such as the hepatic steatosis index (HSI) and fatty liver index, derived from AST, ALT, and/or platelets, have been suggested to predict hepatic steatosis and fibrosis in adults [31,32]. In addition, the triglyceride glucose (TyG) index, a parameter derived as the product of fasting triglyceride and glucose levels, has been suggested to be a robust predictive marker for NAFLD based on the relationship between NAFLD and dyslipidemia and IR [33]. However, investigations on the usefulness of these parameters in children and adolescents are limited.
A pediatric study showed a positive association between HSI and the presence of hepatic steatosis [34]. A population-based study reported that modified TyG indices, which combined the TyG index and obesity-related parameters, were superior to the TyG index for predicting pediatric NAFLD [20]. A cohort study reported that HSI was superior to the TyG index for NAFLD predictions in children [35]. Song et al. [21] suggested the AST-to-platelet ratio index (APRI), fibrosis-4 index, and HSI as predictive markers of pediatric NAFLD. In addition, this study reported that the modified APRI, which combined APRI and obesityrelated parameters, such as BMI and waist circumference, were superior to other parameters. For predicting hepatic fibrosis, the area under the curve of receiver operating characteristic curve values was 0.741 for APRI, 0.71 for NAFLD fibrosis score, and 0.546 for pediatric NAFLD fibrosis index in a retrospective study [36]. Formulas of biomarkers for pediatric NAFLD and fibrosis are shown in Table 1.
Ultrasonography is an easily accessible imaging modality without radiation exposure that can exclude other hepatic pathologies and assess hepatic fatty infiltration grade based on degree of echogenicity [3,23]. Hepatic steatosis shows high echogenicity because more echoes return to the ultrasound transducer due to disturbed propagation and attenuation of the sound wave by lipid droplets in hepatocytes [37]. The normal liver shows a homogenous echo texture similar to that of the renal cortex. A mildly fatty liver shows diffuse increased echogenicity versus the renal cortex, while a moderately fatty liver shows obscured vessel walls due to increased liver echogenicity. In a severely fatty liver, the diaphragm is not visible owing to acoustic attenuation. However, ultrasound is physician- and device-dependent and cannot be used for a quantitative analysis. Moreover, its accuracy is limited due to its low sensitivity and specificity [3].
Based on evidence of increased attenuation of the ultrasound beam in the fatty liver, the controlled attenuation parameter (CAP) is suggested as an imaging modality for the quantification of fat deposition in the liver [23,38]. The CAP estimates the decreased amplitude of ultrasound as it is amplified through the liver tissue [37]. In addition, the liver stiffness measurement (LSM) represents a measure of the shear wave velocity generated by a push pulse as it passes [39]. CAP and LSM can be obtained by a FibroScan (Echosens, Paris, France), an ultrasound-based vibration-controlled transient elastography device, to estimate liver stiffness [40]. In a pediatric study, 95th percentile values of CAP and LSM were 214.53 dB/m and 4.78 kPa, respectively [40]. However, the clinical application of CAP is limited due to significant measurement failure and an unavailability of anatomic imaging [23,38]. The LSM can be altered by several factors such as venous pressure, inflammation, amyloid deposition, and cholestasis [39].
To overcome the limitations of CAP, attenuation imaging (ATI) has recently been suggested to quantify liver fat using realtime ultrasound. ATI estimates the degree of ultrasound beam attenuation with depth using a rudimentary echo decay profile after removing the systemic effects of ultrasound [41]. Operators can easily measure ATI values during grayscale ultrasound by automatically removing vessels and artifacts. ATI is highly correlated with magnetic resonance imaging (MRI) proton density fat fraction (PDFF) and shows outstanding performance for the detection of fatty liver in adults [42,43]. In a pediatric study, ATI was useful for distinguishing fatty from normal liver and moderate to severe from mild fatty liver [23]. In this study, the median ATI values for normal, mild fatty, and moderate to severe fatty liver were 0.54, 0.63, and 0.73 dB/cm/MHz, respectively. The cutoff ATI value was 0.69 dB/cm/MHz for differentiating moderate to severe fatty liver from mild fatty liver.
MRI measures PDFF, the ratio of proton density in fatty liver and that in the whole liver, based on chemical shift imaging [41]. Magnetic resonance signals are attenuated or combined by the resonance frequency of fat and water molecules and different phases. Signals of fat and water are combined and the total signal intensity is higher on the in-phase echo time, while water and fat signals cancel each other out and the total signal intensity is reduced in the out-of-phase echo time. Thus, the normal liver does not show a difference in signal intensities between the inphase and out-of-phase images, whereas the signal intensity is reduced on the out-of-phase image in hepatic steatosis. Tang et al. [44] reported that MRI-PDFF was significantly correlated with histologic steatosis grade, while the area under the receiver operating characteristic curve was 0.989 for distinguishing participants with healthy liver from those with hepatic steatosis among patients with NAFLD, including children.
Magnetic resonance elastography (MRE) can be used to quantify hepatic fibrosis and cirrhosis. MRE estimates liver stiffness and fibrosis and distinguishes steatohepatitis from simple steatosis [37,38]. Yin et al. [45] reported that the sensitivity and specificity of MRE for the detection of hepatic fibrosis were 98% and 99%, respectively, using a cutoff value of 2.93 kPa. A prospective study reported that the mean liver stiffness was 2.45 kPa among children and adolescents, higher than that in adults (2.1 kPa) [46]. In this study, the 95th percentile of normal liver stiffness was 3.11–3.28 kPa in children and 2.70–3.35 kPa in adolescents. Another prospective study reported that the mean liver stiffness was 2.1 kPa among children with normal liver, while the 95th percentile of normal liver stiffness was 2.8 kPa [47].
Insulin resistance plays a major role in the pathogenesis of NAFLD, and ectopic fat accumulation plays a key role in insulin resistance [9,48]. Insulin downregulates gluconeogenesis by promoting glycogen synthesis in the liver and muscle and inhibiting lipolysis in adipose tissue [4,48,49]. Fat accumulation due to excess caloric intake and decreased physical activity induces adipocyte hypertrophy and dysfunction as well as insulin resistance, thereby increasing lipolysis and free fatty acid levels (Fig. 1) [48,50]. Free fatty acids taken up by the cell via fatty acid transport protein increases long-chain acyl-CoA and diacylglycerol, which induces insulin resistance by inhibiting insulin receptor function via activating protein kinase C in the liver and skeletal muscles (Fig. 2) [48]. In the skeletal muscle, decreased glycogen synthesis due to insulin resistance induces hyperglycemia and postprandial hyperinsulinemia, which induce hepatic de novo lipogenesis, hypertriglyceridemia, increased very low-density lipoprotein (VLDL) production, and decreased plasma high-density lipoprotein (HDL) cholesterol [51]. In the liver, hyperinsulinemia promotes the activation of sterol regulatory element-binding protein 1 (SREBP1) and hyperglycemia and activates carbohydrate response element-binding protein (ChREBP) [51,52]. SREBP1 and ChREBP promote de novo lipogenesis, lipid production from carbohydrate in the liver. Although de novo lipogenesis is not the main source of lipids, it can be a main factor for lipid supply in people with obesity and hyperinsulinemia [4]. Moreover, hyperinsulinemia promotes lipid accumulation by inactivating Forkhead box protein A2, a transcription factor promoting lipid oxidation in the liver [52]. The paradox in the pathogenesis of NAFLD wherein insulin fails to suppress glucose production while continuing lipogenesis in the liver can be explained by selective insulin resistance, which indicates different actions of insulin between glycogen synthesis and lipogenesis [51,53].
In summary, insulin resistance promotes lipolysis in adipose tissue, which increases the influx of free fatty acids into the skeletal muscle and liver [48,50,51]. In the muscle cells, insulin resistance inhibits glycogen synthesis, which increases glucose influx into the liver. Finally, hyperinsulinemia and increased de novo lipogenesis accelerate lipid accumulation in the liver [4].
Hepatocytes secrete various hepatokines, protein signals involved in lipid metabolism and insulin sensitivity [54]. In adipose and skeletal muscles, many hepatokines influence the pathways involved in lipolysis, lipogenesis, inflammation, and fatty acid oxidation. Some hepatokines independently affect insulin secretion from the pancreas and glucose uptake by the peripheral tissues. Hepatokine secretion is altered in fatty livers, which affects fatty acid metabolism and induces insulin resistance and inflammation in other tissues. For example, Fetuin A retinol binding protein 4 suppress insulin signaling by inducing phosphorylation of cJun-N-terminal-kinase and insulin receptor substrate 1 [54,55]. In addition, Fetuin A circulating selenoprotein P reduces insulin sensitivity by inhibiting insulin receptors [54,56].
Micro RNAs (miRNAs) are noncoding RNAs that inhibit the translation and induce mRNA degradation in lysosomes by binding to the 3′ untranslated region of messenger RNAs [57]. Specific miRNAs control metabolic functions, including insulin sensitivity in the liver, skeletal muscle, and adipose tissue and insulin secretion in the pancreas [54,58]. In NAFLD, an increase in neutral sphingomyelinase 2 activity and ceramide accumulation promotes miRNA secretion, thus altering lipid transport and insulin signaling [59,60].
Liver-derived major metabolite classes including lipoproteins and acyl-carnitines transduce specific metabolic signals, which are altered in NAFLD [54]. VLDL secretion, which transports triglycerides and cholesterol to the peripheral tissues, is promoted in NAFLD, which induces insulin resistance by decreasing glucose transporter 4 insertion in the membrane and cholesterol accumulation in the skeletal muscle [61,62]. Moreover, increased VLDL secretion is associated with inflammation such as tumor necrosis factor-alpha production, which further accelerates hepatic VLDL secretion and insulin resistance [63-65]. In fatty liver, plasma acylcarnitines are increased and accumulate in the muscle, which promotes insulin resistance in the skeletal muscle due to mitochondrial stress and reactive oxygen production [66-68].
NAFLD is associated with a higher prevalence of diabetes and cardiovascular diseases, and diabetes is a risk factor for the development of liver fibrosis and cirrhosis [69-71]. The association of NAFLD with metabolic syndrome and insulin resistance has been reported in many studies (Table 2). In a global meta-analysis, the prevalence of NAFLD among patients with type 2 diabetes was 55.5%, more than 2-fold higher than that in the general population [72]. Another meta-analysis reported that NAFLD was related to a 2-fold incidence in metabolic syndrome and type 2 diabetes [73]. D'Adamo et al. [74] reported that youth with obesity and fatty liver had impaired insulin action in the liver and muscle. A pediatric study reported that NAFLD was associated with a two-fold prevalence of prediabetes or diabetes [69]. Nadeau et al. [75] reported that nearly half of all youth with type 2 diabetes showed abnormal ALT levels. In a population-based study from Korea, the prevalence of prediabetes was 15.6% among youth with NAFLD versus 7.8% among those without NAFLD [1]. In addition, the prevalence of NAFLD was 15.6% among youth with prediabetes versus 9.1% among those without prediabetes. The odds ratio of prediabetes for NAFLD was 1.85. Children with metabolic syndrome had a 5 times higher odds ratio of having NAFLD versus those without metabolic syndrome even after the adjustment for age, sex, race, ethnicity, BMI, and hyperinsulinemia in a cross-sectional study [76]. In another study, the odds ratio of NAFLD for metabolic syndrome was 2.65 among children with obesity [77].
Based on the association between NAFLD, insulin resistance, and metabolic syndrome, a new concept, MAFLD, has been suggested [10]. Furthermore, the definition of pediatric MAFLD was suggested by an international expert consensus statement as hepatic steatosis with one of the following: (1) excess adiposity, including overweight and obesity and/or abdominal obesity; (2) prediabetes or type 2 diabetes; and (3) 2 or more metabolic risk factors such as abnormal triglycerides, HDL cholesterol, blood pressure, and triglyceride-to-HDL cholesterol ratio [78].
This review discussed the epidemiology of pediatric NAFLD and the association between insulin resistance and NAFLD. Although the prevalence of NAFLD is increasing globally, with an increased prevalence of obesity and obesity-related comorbidities, investigations on proper screening methods and the non-invasive diagnosis and treatment of NAFLD are limited in children and adolescents. Moreover, the prevalence of obesity has increased globally during the COVID-19 outbreak, but investigations of the prevalence of pediatric NAFLD and the association between COVID-19 and NAFLD are limited. Further studies investigating non-invasive diagnostic methods of NAFLD as well as the effect of COVID-19 on NAFLD, focusing on the relationship between NAFLD and insulin resistance, are required for the management of future risks of advanced liver diseases and cardiovascular diseases. In addition, investigations into the clinical application of MAFLD in children and adolescents are required to confirm the validity of renaming NAFLD as MAFLD.
Fig. 1.
Insulin resistance in adipocytes. Fat accumulation due to excess caloric intake and decreased physical activity induces hypertrophy and dysfunction of adipocytes, which induces adipocyte hypoxia and an unfolded protein response. This response is followed by the inflammatory pathway and cytokine production, which interfere with the insulin signaling pathway. Finally, insulin resistance increases lipolysis and free fatty acids.
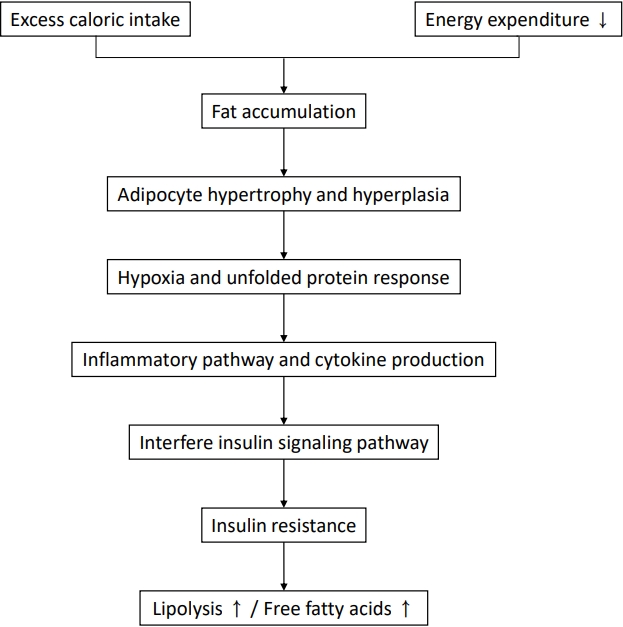
Fig. 2.
Insulin resistance in skeletal muscles and liver. In skeletal muscle and liver, influx of FFA increases LC-CoA and DAG, which induces insulin resistance by inhibiting the function of insulin receptors via activation of PKC. Insulin resistance induces hyperglycemia via hyperinsulinemia and decreases glycogen synthesis. De novo lipogenesis is induced by ChREBP activation and SREBP-1c in the liver. In addition, hyperinsulinemia promotes lipid accumulation by inactivating Forkhead box protein A2, a transcription factor that promotes lipid oxidation in the liver. FFA, free fatty acids; LC-CoA, long-chain acyl-CoA; DAG, diacylglycerol; PKC, protein kinase C; ChREBP, carbohydrate response element-binding protein; SREBP- 1c, sterol regulatory element-binding protein 1c; Foxa2, Forkhead box protein A2.

Table 1.
NAFLD, nonalcoholic fatty liver disease; TyG, triglycerides and glucose index; TG, triglyceride; BMI, body mass index; SDS, standard deviation score; WC, waist circumference; WHtR, waist-to-height ratio; WHR, waist-to-hip ratio; AST, aspartate aminotransferase; APRI, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gamma-glutamyl transferase.
Table 2.
Description of studies examining the association between NAFLD, diabetes mellitus, and metabolic syndrome in children and adolescents
| Study | Country | Ethnicity | Design | Sample size | Age | Main findings |
|---|---|---|---|---|---|---|
| Nobili et al. [69] | Italy | Caucasian | Cohort | 99 Children with NAFLD and 118 children without NAFLD | Children with NAFLD: 11.7±3.1 children without NAFLD: 11.3 ±2.5 | Prevalence of prediabetes or diabetes was 20.6% in children with NAFLD and 11% in children without NAFLD |
| Nadeau et al. [75] | US | Hispanic, Caucasian, AfricanAmerican, Asian | Retrospective | 115 Children with type 2 diabetes | 15.4±0.4 | The prevalence of elevated alanine amino transferase level was 48% among children with type 2 diabetes. |
| Song et al. [1] | Korea | Asian | Retrospective, cross-sectional | 6,327 Children and adolescents | Boys: 14.33±0.05 | The odds ratio of prediabetes for NAFLD was 1.85. |
| Girls: 14.30±0.06 | ||||||
| Yang et al. [36] | Korea | Asian | Retrospective cross-sectional | 152 Children and adolescents | 12.9±2.8 | The prevalence of type 2 diabetes was 33% among children with NAFLD and 10.3% among those without NAFLD. |
| Schwimmer et al. [76] | US | Hispanic, Caucasian, AfricanAmerican, Asian | Case-control | 150 Children with NAFLD and 150 children without NAFLD | Children with NAFLD: 12.5±2.6 children without NAFLD: 12.8 ±2.9 | The prevalence of impaired fasting glucose was 13% among children with NAFLD and 2% among those without NAFLD. |
| Gupta et al. [77] | US | Hispanic, Caucasian, AfricanAmerican, Asian | Retrospective | 700 obese children | 11.9±3.5 | The odds ratio of NAFLD for metabolic syndrome was 2.65. |
| ElKoofy et al. [79] | Egypt | Asian | Retrospective | 33 Children with overweight or obesity | 8.4±3 | The prevalence of metabolic syndrome was 80% among children with NAFLD and 44.4% among those without NAFLD. |
| Tominaga et al. [80] | Japan | Asian | Case-control, cross-sectional | 846 Children | 6–15 | Odds ratio for NAFLD increased with increase in metabolic syndrome components. |
References
1. Song K, Park G, Lee HS, Lee M, Lee HI, Ahn J, et al. Trends in prediabetes and non-alcoholic fatty liver disease associated with abdominal obesity among Korean children and adolescents: based on the Korea National Health and Nutrition Examination Survey between 2009 and 2018. Biomedicines 2022;10:584.



2. Yang HR. Noninvasive diagnosis of pediatric nonalcoholic fatty liver disease. Korean J Pediatr 2013;56:45-51.



3. Vos MB, Abrams SH, Barlow SE, Caprio S, Daniels SR, Kohli R, et al. NASPGHAN clinical practice guideline for the diagnosis and treatment of nonalcoholic fatty liver disease in children: recommendations from the expert committee on NAFLD (ECON) and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN). J Pediatr Gastroenterol Nutr 2017;64:319-34.



4. Alam S, Mustafa G, Alam M, Ahmad N. Insulin resistance in development and progression of nonalcoholic fatty liver disease. World J Gastrointest Pathophysiol 2016;7:211-7.



5. Younossi Z, Anstee QM, Marietti M, Hardy T, Henry L, Eslam M, et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol 2018;15:11-20.



6. Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc 1980;55:434-8.


8. Tanase DM, Gosav EM, Costea CF, Ciocoiu M, Lacatusu CM, Maranduca MA, et al. The intricate relationship between type 2 diabetes mellitus (T2DM), insulin resistance (IR), and nonalcoholic fatty liver disease (NAFLD). J Diabetes Res 2020;2020:3920196.




9. Marušić M, Paić M, Knobloch M, Liberati Pršo AM. NAFLD, insulin resistance, and diabetes mellitus type 2. Can J Gastroenterol Hepatol 2021;2021:6613827.


10. Fouad Y, Waked I, Bollipo S, Gomaa A, Ajlouni Y, Attia D. What's in a name? Renaming 'NAFLD' to 'MAFLD'. Liver Int 2020;40:1254-61.



11. Le MH, Yeo YH, Li X, Li J, Zou B, Wu Y, et al. 2019 global NAFLD prevalence: a systematic review and meta-analysis. Clin Gastroenterol Hepatol 2021;20:2809-17.

12. Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016;64:73-84.


13. Kang SY, Kim YJ, Park HS. Trends in the prevalence of non-alcoholic fatty liver disease and its future predictions in Korean men, 1998-2035. J Clin Med 2020;9:2626.



14. Fujii H, Nakamura N, Fukumoto S, Kimura T, Nakano A, Nadatani Y, et al. Lifestyle changes during the coronavirus disease 2019 pandemic impact metabolic dysfunction-associated fatty liver disease. Liver Int 2022;42:995-1004.



15. Milic J, Barbieri S, Gozzi L, Brigo A, Beghé B, Verduri A, et al. Metabolicassociated fatty liver disease is highly prevalent in the postacute COVID syndrome. Open Forum Infect Dis 2022;9:ofac003.




16. Zhang X, Wu M, Liu Z, Yuan H, Wu X, Shi T, et al. Increasing prevalence of NAFLD/NASH among children, adolescents and young adults from 1990 to 2017: a population-based observational study. BMJ Open 2021;11:e042843.



17. Anderson EL, Howe LD, Jones HE, Higgins JP, Lawlor DA, Fraser A. The prevalence of non-alcoholic fatty liver disease in children and adolescents: a systematic review and meta-analysis. PLoS One 2015;10:e0140908.



18. Welsh JA, Karpen S, Vos MB. Increasing prevalence of nonalcoholic fatty liver disease among United States adolescents, 1988-1994 to 2007-2010. J Pediatr 2013;162:496-500.e1.



19. Song P, Yu J, Wang M, Chang X, Wang J, An L. Prevalence and correlates of suspected nonalcoholic fatty liver disease in Chinese children. Int J Environ Res Public Health 2017;14:465.



20. Song K, Park G, Lee HS, Lee M, Lee HI, Choi HS, et al. Comparison of the triglyceride glucose index and modified triglyceride glucose indices to predict nonalcoholic fatty liver disease in youths. J Pediatr 2022;242:79-85.e1.


21. Song K, Lee HW, Choi HS, Park G, Lee HS, Kim SJ, et al. Comparison of the modified TyG indices and other parameters to predict non-alcoholic fatty liver disease in Youth. Biology (Basel) 2022;11:685.



22. Shah J, Okubote T, Alkhouri N. Overview of updated practice guidelines for pediatric nonalcoholic fatty liver disease. Gastroenterol Hepatol (N Y) 2018;14:407-14.


23. Song K, Son NH, Chang DR, Chae HW, Shin HJ. Feasibility of ultrasound attenuation imaging for assessing pediatric hepatic steatosis. Biology 2022;11:1087.



24. Loaeza-del-Castillo A, Paz-Pineda F, Oviedo-Cárdenas E, Sánchez-Avila F, Vargas-Vorácková F. AST to platelet ratio index (APRI) for the noninvasive evaluation of liver fibrosis. Ann Hepatol 2008;7:350-7.


25. Vajro P, Lenta S, Socha P, Dhawan A, McKiernan P, Baumann U, et al. Diagnosis of nonalcoholic fatty liver disease in children and adolescents: position paper of the ESPGHAN Hepatology Committee. J Pediatr Gastroenterol Nutr 2012;54:700-13.


26. Molleston JP, Schwimmer JB, Yates KP, Murray KF, Cummings OW, Lavine JE, et al. Histological abnormalities in children with nonalcoholic fatty liver disease and normal or mildly elevated alanine aminotransferase levels. J Pediatr 2014;164:707-13.e3.



27. Schwimmer JB, Dunn W, Norman GJ, Pardee PE, Middleton MS, Kerkar N, et al. SAFETY study: alanine aminotransferase cutoff values are set too high for reliable detection of pediatric chronic liver disease. Gastroenterology 2010;138:1357-64; 1364.e1-2.



28. Schwimmer JB, Newton KP, Awai HI, Choi LJ, Garcia MA, Ellis LL, et al. Paediatric gastroenterology evaluation of overweight and obese children referred from primary care for suspected non-alcoholic fatty liver disease. Aliment Pharmacol Ther 2013;38:1267-77.



29. Kang Y, Park S, Kim S, Koh H. Normal serum alanine aminotransferase and non-alcoholic fatty liver disease among Korean adolescents: a cross-sectional study using data from KNHANES 2010-2015. BMC Pediatr 2018;18:215.




30. Cho SM, Lee SG, Kim HS, Kim JH. Establishing pediatric reference intervals for 13 biochemical analytes derived from normal subjects in a pediatric endocrinology clinic in Korea. Clin Biochem 2014;47:268-71.


31. Lee JH, Kim D, Kim HJ, Lee CH, Yang JI, Kim W, et al. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis 2010;42:503-8.


32. Long MT, Gandhi S, Loomba R. Advances in non-invasive biomarkers for the diagnosis and monitoring of non-alcoholic fatty liver disease. Metabolism 2020;111S:154259.



33. Kim HS, Cho YK, Kim EH, Lee MJ, Jung CH, Park JY, et al. Triglyceride glucose-waist circumference is superior to the homeostasis model assessment of insulin resistance in identifying nonalcoholic fatty liver disease in healthy subjects. J Clin Med 2021;11:41.



34. Ferraioli G, Calcaterra V, Lissandrin R, Guazzotti M, Maiocchi L, Tinelli C, et al. Noninvasive assessment of liver steatosis in children: the clinical value of controlled attenuation parameter. BMC Gastroenterol 2017;17:61.




35. Shi M, Liu P, Li J, Su Y, Zhou X, Wu C, et al. The performance of noninvasive indexes of adults in identification of nonalcoholic fatty liver disease in children. J Diabetes 2021;13:744-53.



36. Yang A, Jung N, Kim S, Lee JE. Association between non-invasive diagnostic methods of liver fibrosis and type 2 diabetes in pediatric patients with nonalcoholic fatty liver disease. Front Pediatr 2022;10:825141.



37. Di Martino M, Koryukova K, Bezzi M, Catalano C. Imaging features of non-alcoholic fatty liver disease in children and adolescents. Children (Basel) 2017;4:73.



38. Tamaki N, Ajmera V, Loomba R. Non-invasive methods for imaging hepatic steatosis and their clinical importance in NAFLD. Nat Rev Endocrinol 2022;18:55-66.




39. Oeda S, Tanaka K, Oshima A, Matsumoto Y, Sueoka E, Takahashi H. Diagnostic accuracy of FibroScan and factors affecting measurements. Diagnostics (Basel) 2020;10:940.



40. Zeng J, Zhang X, Sun C, Pan Q, Lu WY, Chen Q, et al. Feasibility study and reference values of FibroScan 502 with M probe in healthy preschool children aged 5years. BMC Pediatr 2019;19:129.




41. Jang JK, Choi SH, Lee JS, Kim SY, Lee SS, Kim KW. Accuracy of the ultrasound attenuation coefficient for the evaluation of hepatic steatosis: a systematic review and meta-analysis of prospective studies. Ultrasonography 2022;41:83-92.




42. Jeon SK, Lee JM, Joo I, Yoon JH, Lee DH, Lee JY, et al. Prospective evaluation of hepatic steatosis using ultrasound attenuation imaging in patients with chronic liver disease with magnetic resonance imaging proton density fat fraction as the reference standard. Ultrasound Med Biol 2019;45:1407-16.


43. Ferraioli G, Maiocchi L, Savietto G, Tinelli C, Nichetti M, Rondanelli M, et al. Performance of the attenuation imaging technology in the detection of liver steatosis. J Ultrasound Med 2021;40:1325-32.




44. Tang A, Tan J, Sun M, Hamilton G, Bydder M, Wolfson T, et al. Nonalcoholic fatty liver disease: MR imaging of liver proton density fat fraction to assess hepatic steatosis. Radiology 2013;267:422-31.



45. Yin M, Talwalkar JA, Glaser KJ, Manduca A, Grimm RC, Rossman PJ, et al. Assessment of hepatic fibrosis with magnetic resonance elastography. Clin Gastroenterol Hepatol 2007;5:1207-13.e2.



46. Sawh MC, Newton KP, Goyal NP, Angeles JE, Harlow K, Bross C, et al. Normal range for MR elastography measured liver stiffness in children without liver disease. J Magn Reson Imaging 2020;51:919-27.




47. Trout AT, Anupindi SA, Gee MS, Khanna G, Xanthakos SA, Serai SD, et al. Normal liver stiffness measured with MR elastography in children. Radiology 2020;297:663-9.



48. Snel M, Jonker JT, Schoones J, Lamb H, de Roos A, Pijl H, et al. Ectopic fat and insulin resistance: pathophysiology and effect of diet and lifestyle interventions. Int J Endocrinol 2012;2012:983814.




49. Nogueira-de-Almeida CA. Metabolic syndrome definition in adolescents should incorporate insulin resistance. Ann Pediatr Endocrinol Metab 2020;25:287-8.




50. Polyzos SA, Kountouras J, Mantzoros CS. Obesity and nonalcoholic fatty liver disease: From pathophysiology to therapeutics. Metabolism 2019;92:82-97.


51. Samuel VT, Shulman GI. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. J Clin Invest 2016;126:12-22.



52. Kohjima M, Higuchi N, Kato M, Kotoh K, Yoshimoto T, Fujino T, et al. SREBP-1c, regulated by the insulin and AMPK signaling pathways, plays a role in nonalcoholic fatty liver disease. Int J Mol Med 2008;21:507-11.


53. Vatner DF, Majumdar SK, Kumashiro N, Petersen MC, Rahimi Y, Gattu AK, et al. Insulin-independent regulation of hepatic triglyceride synthesis by fatty acids. Proc Natl Acad Sci U S A 2015;112:1143-8.



54. Watt MJ, Miotto PM, De Nardo W, Montgomery MK. The liver as an endocrine organ-linking NAFLD and insulin resistance. Endocr Rev 2019;40:1367-93.



55. Ou HY, Wu HT, Hung HC, Yang YC, Wu JS, Chang CJ. Endoplasmic reticulum stress induces the expression of fetuin-A to develop insulin resistance. Endocrinology 2012;153:2974-84.



56. Misu H, Takamura T, Takayama H, Hayashi H, Matsuzawa-Nagata N, Kurita S, et al. A liver-derived secretory protein, selenoprotein P, causes insulin resistance. Cell Metab 2010;12:483-95.


57. Guo H, Ingolia NT, Weissman JS, Bartel DP. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 2010;466:835-40.




58. Hashimoto N, Tanaka T. Role of miRNAs in the pathogenesis and susceptibility of diabetes mellitus. J Hum Genet 2017;62:141-50.



59. Dávalos A, Goedeke L, Smibert P, Ramírez CM, Warrier NP, Andreo U, et al. miR-33a/b contribute to the regulation of fatty acid metabolism and insulin signaling. Proc Natl Acad Sci U S A 2011;108:9232-7.



60. Apostolopoulou M, Gordillo R, Koliaki C, Gancheva S, Jelenik T, De Filippo E, et al. Specific hepatic sphingolipids relate to insulin resistance, oxidative stress, and inflammation in nonalcoholic steatohepatitis. Diabetes Care 2018;41:1235-43.



61. Barrientos G, Sánchez-Aguilera P, Jaimovich E, Hidalgo C, Llanos P. Membrane cholesterol in skeletal muscle: a novel player in excitationcontraction coupling and insulin resistance. J Diabetes Res 2017;2017:3941898.




62. Roden M, Price TB, Perseghin G, Petersen KF, Rothman DL, Cline GW, et al. Mechanism of free fatty acid-induced insulin resistance in humans. J Clin Invest 1996;97:2859-65.



63. Song Y, Zhao M, Cheng X, Shen J, Khound R, Zhang K, et al. CREBH mediates metabolic inflammation to hepatic VLDL overproduction and hyperlipoproteinemia. J Mol Med (Berl) 2017;95:839-49.




64. Akash MSH, Rehman K, Liaqat A. Tumor necrosis factor-alpha: role in development of insulin resistance and pathogenesis of type 2 diabetes mellitus. J Cell Biochem 2018;119:105-10.



65. Kim J, Lee J. Role of obesity-induced inflammation in the development of insulin resistance and type 2 diabetes: history of the research and remaining questions. Ann Pediatr Endocrinol Metab 2021;26:1-13.




66. Aguer C, McCoin CS, Knotts TA, Thrush AB, Ono-Moore K, McPherson R, et al. Acylcarnitines: potential implications for skeletal muscle insulin resistance. Faseb J 2015;29:336-45.




67. Koves TR, Ussher JR, Noland RC, Slentz D, Mosedale M, Ilkayeva O, et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab 2008;7:45-56.


68. Kalhan SC, Guo L, Edmison J, Dasarathy S, McCullough AJ, Hanson RW, et al. Plasma metabolomic profile in nonalcoholic fatty liver disease. Metabolism 2011;60:404-13.



69. Nobili V, Mantovani A, Cianfarani S, Alisi A, Mosca A, Sartorelli MR, et al. Prevalence of prediabetes and diabetes in children and adolescents with biopsy-proven non-alcoholic fatty liver disease. J Hepatol 2019;71:80210.

70. Newton KP, Hou J, Crimmins NA, Lavine JE, Barlow SE, Xanthakos SA, et al. Prevalence of prediabetes and type 2 diabetes in children with nonalcoholic fatty liver disease. JAMA Pediatr 2016;170:e161971.



71. Castorani V, Polidori N, Giannini C, Blasetti A, Chiarelli F. Insulin resistance and type 2 diabetes in children. Ann Pediatr Endocrinol Metab 2020;25:217-26.




72. Younossi ZM, Golabi P, de Avila L, Paik JM, Srishord M, Fukui N, et al. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: a systematic review and meta-analysis. J Hepatol 2019;71:793-801.


73. Ballestri S, Zona S, Targher G, Romagnoli D, Baldelli E, Nascimbeni F, et al. Nonalcoholic fatty liver disease is associated with an almost twofold increased risk of incident type 2 diabetes and metabolic syndrome. Evidence from a systematic review and meta-analysis. J Gastroenterol Hepatol 2016;31:936-44.



74. D'Adamo E, Cali AM, Weiss R, Santoro N, Pierpont B, Northrup V, et al. Central role of fatty liver in the pathogenesis of insulin resistance in obese adolescents. Diabetes Care 2010;33:1817-22.




75. Nadeau KJ, Klingensmith G, Zeitler P. Type 2 diabetes in children is frequently associated with elevated alanine aminotransferase. J Pediatr Gastroenterol Nutr 2005;41:94-8.


76. Schwimmer JB, Pardee PE, Lavine JE, Blumkin AK, Cook S. Cardiovascular risk factors and the metabolic syndrome in pediatric nonalcoholic fatty liver disease. Circulation 2008;118:277-83.



77. Gupta R, Bhangoo A, Matthews NA, Anhalt H, Matta Y, Lamichhane B, et al. The prevalence of non-alcoholic fatty liver disease and metabolic syndrome in obese children. J Pediatr Endocrinol Metab 2011;24:907-11.


78. Eslam M, Alkhouri N, Vajro P, Baumann U, Weiss R, Socha P, et al. Defining paediatric metabolic (dysfunction)-associated fatty liver disease: an international expert consensus statement. Lancet Gastroenterol Hepatol 2021;6:864-73.


79. El-Koofy NM, Anwar GM, El-Raziky MS, El-Hennawy AM, El-Mougy FM, El-Karaksy HM, et al. The association of metabolic syndrome, insulin resistance and non-alcoholic fatty liver disease in overweight/obese children. Saudi J Gastroenterol 2012;18:44-9.








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


