Article Contents
| Clin Exp Pediatr > Volume 66(11); 2023 |
|
Abstract
Background
Dehydration due to acute diarrhea is among the leading causes of mortality. However, advancements in management and technology do not help clinicians differentiate dehydration degrees. Ultrasound using the inferior vena cava to aorta (IVC/ Ao) ratio is a promising noninvasive technique to identify significant pediatric dehydration.
Puspose
Therefore, this systematic review and meta-analysis aimed to examine the diagnostic parameters of the IVC/Ao ratio for predicting clinically significant dehydration in pediatric patients.
Methods
We searched the MEDLINE, PubMed, Cochrane Library, ScienceDirect, and Google Scholar databases for studies of pediatric patients (≤18 years old) who presented with signs and symptoms of dehydration due to acute diarrhea, gastroenteritis, or vomiting. The inclusion criteria were cross-sectional, case-control, cohort, and randomized controlled trial study design and publication in any language. We then conducted a meta-analysis using the midas and metandi commands from Stata software.
Results
Five studies of 461 patients were included. The combined sensitivity was 86% (95% confidence interval [CI], 79%–91%), while the specificity was 73% (95% CI, 59%–84%). The area under the curve was 0.89 (95% CI, 0.86–0.91). The positive likelihood ratio (LR+) was 3.2 (95% CI, 2.1–5.1) with a 76% posttest probability, while the negative likelihood ratio (LR-) was 0.18 (95% CI, 0.12–0.28) with a 16% posttest probability. The combined negative predictive value was 0.83 (95% CI, 0.75–0.91), while the positive predictive value was 0.75 (95% CI, 0.68–0.82).
Graphical abstract
Dehydration is a physiological disturbance that affects salt and water loss, and the most common cause of pediatric dehydration is infectious diarrhea caused by viral or bacterial pathogens [1]. According to the World Health Organization (WHO), among the 1.7 billion diarrhea cases annually, around 525,000 children under 5 years of age are killed, making diarrhea the second most common cause of death in this population [2]. One study in Japan reported that the incidence of hospitalization in children <5 years of age due to rotavirus gastroenteritis is 13 per 1,000 person-years, while the cumulative incidence by 5 years of age is 6.6% [3]. In Indonesia, the burden of rotavirus diarrhea is around 60%, and children with rotavirus diarrhea are more likely to develop vomiting and dehydration [4].
Dehydration is a common concern for parents and clinicians because prompt and adequate rehydration therapy prevents mortality [5,6]. However, to prescribe sufficient rehydration therapy that does not under- or overtreat dehydration, the clinician must correctly determine its severity [7]. Under- or untreated dehydration results in acidosis, electrolyte disturbances, and end-organ damage, such as renal insufficiency, lethargy, and cardiovascular instability. Similarly, overtreatment of dehydration prolongs the length of stay and may result in unnecessary morbidity [7]. Despite advancements in knowledge and technology, classifying the degrees of dehydration in pediatric patients remains difficult [8].
Current methods for detecting significant dehydration, usually defined as a fluid deficit of ≥5% of the patient’s body weight, rely heavily on history taking, physical findings, complementary laboratory findings, and physician gestalt. Different guidelines vary significantly in determining pediatric dehydration criteria, cutoffs, and diagnosis [9]. One systematic review found that a combination of abnormal capillary refill time, abnormal skin turgor, and abnormal respiratory pattern are the most valuable signs for predicting 5% pediatric dehydration. The same systematic review also reported that history taking and laboratory tests are only moderately useful. However, these clinical signs possess poor sensitivity, limiting their utility [7]. A prolonged capillary refill time, touted as the best single sign to predict 5% pediatric dehydration [10], also suffers from low sensitivity [11].
Several clinical scoring systems, such as The Etiology, Risk Factors and Interactions of Enteric Infections and Malnutrition and the Consequences for Child Health and Development score [12], modified Vesikari score [13], Community Diarrhea score [14], Clinical Dehydration Scale [15], Gorelick scale [16], and WHO scale [17], produce conflicting results due to their development in a specific population using specific definitions, settings, methodologies, and diarrhea etiologies [18-20]. Moreover, laboratory panels such as electrolyte, bicarbonate, blood urea nitrogen, serum anion gap, and urine specific gravity have no straightforward clinical utility for diagnosing pediatric dehydration [7,21-24].
Point-of-care ultrasonography has garnered significant attention, and an article even calls it a diagnostic tool beyond a stethoscope [25]. Because of its dynamic nature, high portability, and shallow learning curve [25-27], ultrasonography is increasingly accepted and used in many fields [28]. The first study to assess pediatric dehydration using ultrasonography was conducted by Kosiak et al. [29], who introduced the inferior vena cava to the aorta (IVC/Ao) diameter index.
However, since its first publication in 2007, the IVC/Ao ratio has not been widely used in pediatric dehydration settings. One explanation is the insufficient evidence to support its use. Studies are sparse, single-center, and underpowered and use different definitions and IVC/Ao ratio cutoffs. Only one meta-analysis assessed the IVC/Ao ratio in pediatric dehydration, and it included only one study, citing that current evidence does not support the routine use of ultrasound to determine dehydration severity [30]. In our opinion, an analysis of one study is insufficient to make a definitive statement about IVC/Ao ratio use in pediatric dehydration.
Therefore, this systematic review and meta-analysis aimed to examine the diagnostic parameters of the IVC/Ao ratio for predicting clinically significant dehydration in pediatric patients.
The authors followed the PRISMA (preferred reporting items for systematic reviews and meta-analyses) of Diagnostic Test Accuracy guidelines [31]. The study protocol was registered with the International PROSPERO (Prospective Register of Systematic Reviews) database (no. CRD42022324734).
The study population included all pediatric patients (≤18 years old) who presented with signs and symptoms of dehydration due to acute diarrhea, gastroenteritis, or vomiting. The primary aim of this study was to examine the sensitivity, specificity, and posttest probability of the IVC/Ao diameter ratio (the index test) measured by ultrasonography for detecting significant dehydration. The percent weight change from presentation to discharge was the benchmark for identifying substantial dehydration [20]. We excluded studies that did not use the same reference standard or assess dehydration or the IVC/Ao diameter ratio. Hence, our research question could be formulated as “In pediatric patients who present with dehydration due to acute diarrhea, gastroenteritis, or vomiting, how accurate is the IVC/Ao diameter ratio for ruling in and ruling out significant dehydration versus the percent weight change from presentation to discharge?”
Any cross-sectional, case-control, cohort, or randomized controlled trial published in any language met the inclusion criteria. The grey literature, such as conference abstracts, theses, and dissertations, was also searched. Case reports, case series, reviews, and animal studies were excluded. Studies with fewer than 50 samples were excluded. The reference lists of reviews were manually searched to ensure that all relevant studies were included.
The literature search began and ended on 7th April 2022. We searched five academic databases: MEDLINE, Cochrane Library, PubMed, ScienceDirect, and Google Scholar. The keywords used were related to the diagnostic tool (“diagnostic imaging,” “ultrasonography,” “inferior vena cava,” “aorta,” and “inferior vena cava to aorta”), population (“pediatric” and “adolescent”), and condition (“diarrhea,” “vomiting,” “gastroenteritis,” and “dehydration”).
Supplementary Table 1 lists the Medical Subject Heading terms for each database. We purposefully did not include any keywords mentioning “sensitivity," “specificity,” or any diagnostic-related terms to avoid missing potential studies [32]. All records were entered into the Rayyan program, which manually screened them and automatically identified duplicates [33].
Using this program, the authors might choose to group pertinent studies together. Two authors (GSO and JW) performed the first search and imported all information into Rayyan software. The initial searches were cross-checked by a different author (MI). Three authors independently reviewed each study that was identified. Conflicts were settled through group discussions and professional judgments (MW). When studies from the same dataset had overlapping time points, we selected data that provided the most information.
Two authors (GSO and NKH) independently extracted the data, whereas the others (JW and MI) verified their accuracy. We gathered pertinent data, including study identity (author and publication year), study characteristics (location, study design, participant age, and study duration), and information regarding ultrasonography (machines used, sensitivity and specificity, and IVC/Ao ratio cutoff).
We used the revised Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool to assess the risk of bias. There were no formal cutoff scores in QUADAS-2, and the risk of bias was presented visually [32]. Four reviewers evaluated the scale independently (GSO, JW, MI, and NKH), and any disagreements were resolved internally and through an expert decision (MW) until consensus was reached. We contacted the associated authors by email in cases of missing or incomplete data.
Before proceeding with the meta-analysis, model diagnostics were checked using a graphical depiction of residual-based goodness of fit, bivariate normality, influence, and outlier detection analysis. Any outlier studies were double-checked using a sensitivity analysis. Bivariate box plots were used to describe the degree of interdependence. We calculated the individual and pooled sensitivity and specificity using the bivariate model of sensitivity and specificity [34]. A summary receiver operating characteristic was used to display and illustrate the trade-off between sensitivity and specificity using the hierarchical summary receiver operating characteristic model [35]. An area under the curve was measured, and a value of 0.9–1 indicated excellent, 0.8–0.9 indicated very good, 0.7–0.8 indicated good, 0.6–0.7 indicated sufficient, and 0.5–0.6 indicated poor diagnostic accuracy [36]. The I2 index was used to measure heterogeneity; a value of 0% implied no observable heterogeneity, while a value of >50% was considered high heterogeneity [37]. A linear regression test of funnel plot asymmetry was used to evaluate publication bias, and a slope coefficient indicated significant asymmetry at values of P<0.1.
We calculated the posttest probability using a Fagan plot based on Bayes’ theorem and the likelihood ratio scattergram. As the prevalence of significant dehydration in pediatric patients varied, we chose a pre-test probability of 50% [38-40]. An arbitrary cutoff point for the likelihood ratio to have marked changes was >+10 for positive likelihood ratios (LR+) and <-0.1 for negative likelihood ratios (LR-) [41]. Additionally, the predictive values and probability modifying plots are displayed, more informative positive results producing curves that incline toward the (0,1) location and more informative negative results producing curves that incline toward the (1,0) line [42]. The “midas” and “metandi” commands of Stata ver. 17.0 (StataCorp, College Station, TX, USA) were used to conduct the analysis [43].
A total of 11,546 articles were retrieved: of them, 279 were immediately removed and 11,267 unique articles were screened. A total of 444 articles were excluded after the title and abstract screening, and five were ultimately included in this meta-analysis (Supplementary Fig. 1).
Additional manual citation searches did not yield additional studies. The characteristics of each study are shown in Table 1, and the descriptive diagnostic test parameters are listed in Table 2. The notable exclusions are presented in Supplementary Table 2. All studies were prospective cohort studies conducted in the emergency department. Across all five studies, there were 461 patients aged 0.8–18 years. Four studies [8,44-46] applied the same cutoff for significant dehydration (>5%), while the other [47] did not specify a cutoff. All studies used the transverse plane to measure IVC and Ao diameters. El Amrousy et al. [44] had the highest enrollment with 200 patients (43.4%). The prevalence of significant dehydration ranged from 32.2%8) to 89.4% [8]. In terms of the risk of bias, patient selection suffers from the highest risk of bias, followed by the index test, flow, and timing. The reference standard had the lowest bias risk. All studies had a low risk of concern in terms of their applicability. Overall, only one study [44] had a low risk of bias, while the other studies had some risk of bias (Supplementary Fig. 2, Supplementary Table 3).
Supplementary Fig. 3 shows the model diagnostics, whereas no studies were outliers with a moderate goodness of fit. However, the bivariate normality assumption was not fulfilled, and three studies [8,44,47] seemed more influential than others with a Cook’s distance of >0.5. The bivariate box plot (Supplementary Fig. 4) showed skewness of the test performance measures toward a higher specificity with lower sensitivity, providing indirect evidence of some threshold variability.
The combined sensitivity was 86% (95% CI, 79%–91%), while the specificity was 73% (95% CI, 59%–84%). Supplementary Fig. 5 shows the combined summary receiver operating characteristic curve with an area under the curve of 0.89 (95% CI, 0.86–0.91), indicating that the IVC/Ao ratio has very good diagnostic accuracy for detecting significant dehydration in pediatric patients. Supplementary Fig. 6 presents the studyspecific sensitivity and specificity, indicating threshold variability as sensitivity increased and specificity decreased and vice versa. The study of Jauregui et al. [8] had the highest sensitivity at 97% (95% CI, 85%–100%), while the study of Kim et al. [46] had the highest specificity at 91% (95% CI, 81%–97%). The I2 value was 83% (95% CI, 63%–100%) (P=0.002), indicating significant substantial heterogeneity. Supplementary Fig. 7 displays Fagan’s nomogram showing that the LR+ was 3.2 (95% CI, 2.1–5.1) with a 76% posttest probability while the LR- was 0.18 (95% CI, 0.12–0.28) with a 16% posttest probability. The likelihood ratio scattergram placed the summary point of likelihood ratios in the right lower quadrant, indicating that the IVC/Ao ratio could not be used to exclude or confirm significant dehydration in pediatric patients (Supplementary Fig. 8). The probability modifying plot tended toward the (1,0) line, indicating a more informative negative result. The combined negative predictive value was 0.83 (95% CI, 0.68–0.82), while the positive predictive value was 0.75 (95% CI, 0.68–0.82) (Fig. 1).
The linear regression test for funnel plot asymmetry yielded a P value of 0.16, indicating insignificant asymmetry. Thus, there was a low chance of publication bias (Supplementary Fig. 9). A sensitivity analysis revealed no significantly altered results when individual studies were omitted (Supplementary Table 3). According to the meta-regression analysis, probability methodology and having a 5% cutoff for significant dehydration contributed to heterogeneity with values of P<0.01 and P=0.1, respectively (Supplementary Table 4).
The IVC/Ao diameter ratio achieved a good sensitivity of 86% and moderate specificity of 73% in this meta-analysis. While these numbers may seem encouraging, sensitivity and specificity alone do not provide a basis for informed decisions following positive and negative screening test results due to false positives and negatives, respectively. Therefore, sensitivity and specificity alone do not rule out significant dehydration in pediatric patients [48]. However, one methodological study recommended displaying a side-by-side forest plot of sensitivity and specificity to assess threshold variability. The forest plot indicates that sensitivity increases with decreasing specificity, indicating threshold variability. This result is also supported by a bivariate boxplot analysis [49]. A variability threshold is a type of variation in which an arbitrary choice of the threshold value may lead to overoptimistic measures of test performance [50]. Only one study [44] prespecified its IVC/Ao ratio cutoff, while the others conducted a series of tests to determine the cutoff, resulting in threshold variability.
The likelihood of a test result in patients with the disease divided by the chance of the test results in patients without the condition is known as LR, another diagnostic test indicator that can be examined. The more the LR+ is >1 or <1 for LR-, the more or less likely the disease, respectively [51]. Therefore, the IVC/Ao ratio has a slight increase and moderate decrease in the likelihood of detecting significant dehydration with an LR+ of 3.2 and LR- of 0.18, respectively [52]. However, prevalence affects all of the above parameters, affecting diagnostic test accuracy [53,54]. A posttest probability depends on prevalence, a clinically more useful diagnostic test accuracy parameter. In this meta-analysis, an IVC/Ao ratio below the suggested cutoff increased the probability of significant dehydration by 26% (from 50% to 76%), while an IVC/Ao ratio above the suggested cutoff decreased the probability by 34% (from 50% to 16%).
The likelihood scattergram indicated that the IVC/Ao ratio cannot be used to exclude or confirm significant dehydration, meaning that the IVC/Ao ratio may only be good in test research and not diagnostic research [55]. All the studies included in this meta-analysis were test research, which has limited applicability to clinical practice. Koeller et al. [56] performed diagnostic research in which they compared The Dehydration: Assessing Kids Accurately (DHAKA) score model with the DHAKA-US and found that ultrasonography has no added benefit. However, it is necessary to point out that they measured the Ao/IVC ratio and not the other way around. We believe that ultrasonography, especially the IVC/Ao ratio, belongs to the “add-on diagnostic test” category in the emergency department for detecting significant dehydration after history taking, clinical signs and symptoms, and basic laboratory work-ups [57]. However, the difficulty obtaining an accurate reading from crying children [58], high interobserver variability [59], and unspecified cutoff and test characteristics [30] complicate the use of ultrasonography in this setting.
Regarding overtreatment, a meta-analysis of different clinical scales to measure severe dehydration in children with acute gastroenteritis found insufficient evidence to warrant routine ultrasonography usage [30]. However, only 1 study in this meta-analysis analyzed ultrasonography, while the other four did not. These results strengthen the findings of a previous meta-analysis that the IVC/Ao ratio may not be clinically valuable for detecting significant dehydration in pediatric patients.
There are several explanations for the shortcomings of ultrasonography in identifying significant dehydration in pediatric patients. First, the best view, mode, and site of measurement were not established. Studies were underpowered, targeted different populations, or had different test characteristics to determine those three characteristics [60,61]. Second, IVC diameter changes with age, and the corresponding normal value for age has not been established in multicenter large-cohort studies of pediatric patients [62]. Pediatric patients with significant dehydration present with inconsolable crying, which affects respiratory variability.
A significant inspiratory effort will introduce more false positives into the results, while shallow breathing during crying will introduce false negatives [63]. Conflicting results have been reported regarding fluid responsiveness detected by ultrasonography in spontaneously breathing patients [64,65]. Finally, the optimal IVC/Ao cutoff ratio has not been determined, contributing to different results using different cutoffs [8,44-47].
The findings of our meta-analysis have some limitations. We found only five studies despite our thorough and systematic search. Meta-analyses of diagnostic test accuracy are notorious for missing relevant studies due to the use of nonspecific keywords [32]. However, we conducted a manual citation and study search to ensure that relevant studies were not missed. Different studies also employed different IVC/Ao ratio cutoffs, contributing to threshold variability that affects diagnostic test parameters. In our meta-analysis, the I2 index was high, which indicated heterogeneity. Combining different studies will introduce bias and variations in our meta-analysis, which may also introduce heterogeneity. Clinical variations, such as in the experience and expertise of ultrasonography operators, varying IVC/Ao ratios, and sampling methodology, may introduce some unexplained heterogeneity. However, the included studies were all conducted in a resource-rich setting with ultrasonography performed in the emergency department. In addition, almost all studies used >5% as the cutoff for significant dehydration.
Despite this heterogeneity, our results remain valid among these populations. The utility of the IVC/Ao ratio for identifying substantial dehydration in pediatric patients with acute diarrhea, gastroenteritis, or vomiting has never been adequately evaluated. We believe that this is the first meta-analysis to systematically examine this research question. Despite the small number of studies, Deeks’ test revealed no publication bias. Despite its low power, use of this test is suggested for diagnostic test accuracy meta-analyses [66]. Most importantly, this meta-analysis found that the IVC/Ao ratio is not valuable for excluding or confirming significant dehydration in pediatric patients. Although it may be too premature to conclude this finding, especially with a small number of studies and patients examined herein, it indeed guides researchers to shift their studies from test research to multicenter adequately powered diagnostic research to help establish the usefulness of the IVC/Ao ratio [55].
The IVC/Ao ratio had a positive posttest probability of 76% and negative posttest probability of 16%. This means that the IVC/Ao ratio is insufficient to exclude or confirm significant dehydration in pediatric patients. More studies are needed, especially multicenter adequately powered diagnostic research, to help establish the usefulness of the IVC/Ao ratio. More research is needed to determine the most suitable mode, view, and location to detect IVC in pediatric patients, normal IVC diameter for age, and the optimal IVC/Ao ratio cutoff. Further studies must identify whether this ratio is useful as a replacement, triage, or add-on test in clinical settings [57].
Supplementary materials
Supplementary Tables 1-4 and Figs. 1-9 can be found via https://doi.org/10.3345/cep.2022.01445.
Supplementary Table 1.
Medical subject heading (MeSH) terms used in each database
Supplementary Table 3.
Quality assessment of diagnostic accuracy studies tool result of each study
Supplementary Fig. 1.
PRISMA (preferred reporting items for systematic reviews and meta-analyses) flowchart for selection of included studies.
Supplementary Fig. 2.
Quality assessment of diagnostic accuracy studies graphical representation of risk of bias.
QUADAS-2, Quality Assessment of Diagnostic Accuracy Studies.
Supplementary Fig. 5.
Summary receiver operating curve with confidence and prediction regions. Sens, Sensitiviy; Spec, Specificity; SROC, Summary Receiver Operating Characteristic; AUC, Area Under the Curve
Supplementary Fig. 6.
Study-specific sensitivity and specificity. CI, confidence interval.
Supplementary Fig. 7.
Fagan’s nomogram of IVC/Ao ratio in detecting significant dehydration in pediatric patients. Prob, probability; LR, Likelihood ratio; Pos: Positive; Neg, negative.
Supplementary Fig. 8.
Likelihood ratio scattergram. LUQ, Left upper quadrant; LRP, Likelihood ratio positive; LRN, Likelihood ratio negative; RUQ, Right upper quadrant; LLQ, Left lower quadrant; RLQ, Right lower quadrant.
Supplementary Fig. 9.
Linear regression test of funnel plot asymmetry.
Footnotes
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contribution
Conceptualization: GSO, MW; Data curation: GSO, MI, JW, NKH; Formal analysis: GSO; Funding acquisition: None; Methodology: GSO, MI, JW, NKH, MW; Project administration: MI, JW, NKH; Visualization: GSO; Writing - original draft: GSO, MI, JW, NKH, MW; Writing - review & editing: GSO, MI, JW, NKH, MW
Fig. 1.
Probability modifying plot. LR, Likelihood ratio; NPV, negative predictive value; PPV, positive predictive value.
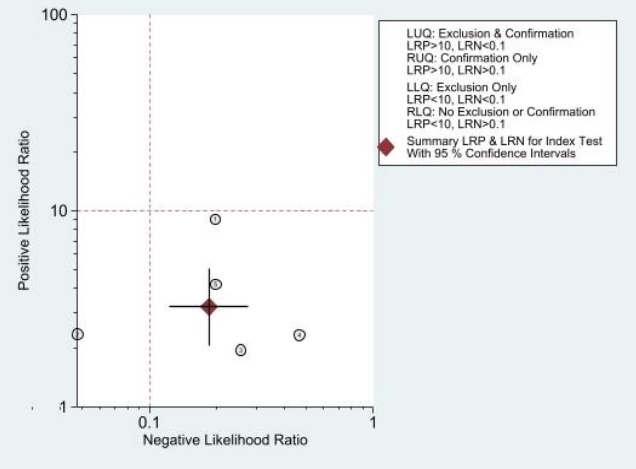
Table 1.
Descriptive findings of each study
| Study | Study period | Setting | Sampling method | Population | Age (yr) | Cutoff for significant dehydration | USG machine | Measurement site | Performing the USG |
|---|---|---|---|---|---|---|---|---|---|
| El Amrousy et al. (2017) [44] | January 2016 to May 2017 | ED | Consecutive | Infants (<24 months) with acute diarrhea and dehydration who required IV fluid for treatment | 12.33±5.14 years for nonsignificant dehydration and 11.37±6.09 years for significant dehydration | Significant (>5%) | Vivid 7(GE Healthcare, Horten, Norway) machine with 7 and 4S probes | Inferior to the xiphoid region | A pediatric cardiologist or expert radiologist |
| Chen et al. (2007) [47] | June 2006 to July 2007 | ED | Not mentioned | Children (6 months and 16 years of age) if they presented with clinical evidence of dehydration and were judged to need IV fluids as determined by the pediatric emergency attending physician on duty | 0.8–16 Years old | Notmentioned | The Sonosite 180+ ultrasound machine (Sonosite, Bothell, WA) with the C60 curvilinear probe | Subxiphoid region, just rostral or caudal to the insertion of the left renal vein into the IVC | One trained pediatric emergency attending physician and one trained medical student |
| Chen et al. (2010) [45] | November 2007 to June 2009 | ED | Consecutive | Patients between 6 months and 18 years of age who presented to the ED with gastroenteritis | 5.3±4.3 Years old | Significant (>5%) | Sonosite MicroMAXX US (Sonosite, Bothell, WA, USA), using the C60 curvilinear probe | Subxiphoid region, just rostral or caudal to the insertion of the left renal vein into the IVC | Five pediatric emergency physicians |
| Jauregui et al. (2014) [8] | June 2011 to February 2013 | ED | Consecutive | All children less than or equal to 18 years of age presenting with a chief complaint of vomiting and/ or diarrhea, or suspicion of dehydration | 6 Years (1 month–18 years) | Significant (>5%) | Sonosite M-Turbo ultrasound machine (Bothell, WA, USA) and curved footprint C-60 abdominal probe or a phased array P21 probe at the time of enrollment (beginning of the ED visit) | Below the level of the xiphoid bone | Trained study staff |
| Kim et al. (2010) [46] | January to August 2008 | ED | Not mentioned | Children (6 months to 6 years) who were clinically judged to have a medical history, signs, or symptoms suspected of dehydration by our emergency medicine specialist (showing symptoms such as vomiting, diarrhea, high fever, inactivity, and impaired oral intake) were targeted | 31±20.2 Months | Significant (>5%) | C60 of Sonosite 180+ (Sonosite Inc.) (5–2 MHz) curved transducer | One cm below the middle hepatic vein in the respiratory cycle | Four emergency medicine majors with ≥2 years of experience |
Table 2.
Diagnostic test parameters of included studies
| Study | Samples recruited | Total sample size (% from samples recruited) | Prevalence of significant dehydration (%) | IVC/Ao ratio cutoff point | Sensitivity | Specificity | Positive likelihood ratio | Negative likelihood ratio | Positive predictive value (%) | Negative predictive value (%) | The area under the curve |
|---|---|---|---|---|---|---|---|---|---|---|---|
| El Amrousy et al. (2017) [44] | N/A | 200 (N/A) | 68 | 0.75 | 82 | 91 | 7.88 | 3.12 | N/A | N/A | 0.87 |
| Chen et al. (2007) [47] | N/A | 36 Pairs of subjects and controls (N/A) | 50 | 1 | 97 | 58 | N/A | N/A | N/A | N/A | 0.91 (0.84–0.98) |
| Chen et al. (2010) [45] | 112 | 71 (63.4) | 39 | 0.8 | 86 | 56 | N/A | N/A | 56 | 86 | 0.73 (0.61–0.84) |
| Jauregui et al. (2014) [8] | 209 | 113 (54.1) | 89.4 | 0.8 | 67 | 71 | 2.32 | N/A | N/A | N/A | 0.72 (0.53–0.91) |
| Kim et al. (2010) [46] | 83 | 59 (71.1) | 32.2 | 0.879 | 85 | 79.9 | N/A | N/A | 76 | 50 | 0.81 for moderate dehydration and 0.69 for severe dehydration |
References
2. World Health Organization. Diarrhoeal disease [Internet]. Geneva (Switzerland): World Health Organization; 2017 [cited 2022 Oct 20]. Available from: https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease.
3. Nakagomi T, Nakagomi O, Takahashi Y, Enoki M, Suzuki T, Kilgore PE. Incidence and burden of rotavirus gastroenteritis in Japan, as estimated from a prospective sentinel hospital study. J Infect Dis 2005;192 Suppl 1:S106–10.


4. Soenarto Y, Aman AT, Bakri A, Waluya H, Firmansyah A, Kadim M, et al. Burden of severe rotavirus diarrhea in Indonesia. J Infect Dis 2009;200 Suppl 1:S188–94.


5. Moritz ML, Ayus JC. Prevention of hospital-acquired hyponatremia: a case for using isotonic saline. Pediatrics 2003;111:227–30.



6. Holliday MA, Friedman AL, Segar WE, Chesney R, Finberg L. Acute hospital-induced hyponatremia in children: a physiologic approach. J Pediatr 2004;145:584–7.


8. Jauregui J, Nelson D, Choo E, Stearns B, Levine AC, Liebmann O, et al. The BUDDY (Bedside Ultrasound to Detect Dehydration in Youth) study. Crit Ultrasound J 2014;6:15.




9. Van den Berg J, Berger MY. Guidelines on acute gastroenteritis in children: a critical appraisal of their quality and applicability in primary care. BMC Fam Pract 2011;12:134.


10. Shavit I, Brant R, Nijssen-Jordan C, Galbraith R, Johnson DW. A novel imaging technique to measure capillary-refill time: improving diagnostic accuracy for dehydration in young children with gastroenteritis. Pediatrics 2006;118:2402–8.



11. Fleming S, Gill P, Jones C, Taylor JA, Van den Bruel A, Heneghan C, et al. The diagnostic value of capillary refill time for detecting serious illness in children: a systematic review and meta-analysis. PLoS One 2015;10:e0138155.



12. Lee GO, Richard SA, Kang G, Houpt ER, Seidman JC, Pendergast LL, et al. A comparison of diarrheal severity scores in the MAL-ED multisite community-based cohort study. J Pediatr Gastroenterol Nutr 2016;63:466–73.



13. Clark HF, Borian FE, Bell LM, Modesto K, Gouvea V, Plotkin SA. Protective effect of WC3 vaccine against rotavirus diarrhea in infants during a predominantly serotype 1 rotavirus season. J Infect Dis 1988;158:570–87.



14. Lee G, Peñataro Yori P, Paredes Olortegui M, Caulfield LE, Sack DA, Fischer-Walker C, et al. An instrument for the assessment of diarrhoeal severity based on a longitudinal community-based study. BMJ Open 2014;4:e004816.



15. Parkin PC, Macarthur C, Khambalia A, Goldman RD, Friedman JN. Clinical and laboratory assessment of dehydration severity in children with acute gastroenteritis. Clin Pediatr 2009;49:235–9.


16. Gorelick MH, Shaw KN, Murphy KO. Validity and reliability of clinical signs in the diagnosis of dehydration in children. Pediatrics 1997;99:E6.


17. World Health Organization. The treatment of diarrhoea: a manual for physicians and other senior health workers [Internet]. Geneva (Switzerland): World Health Organization; 2005 [cited 2022 Oct 20]. Available from: http://apps.who.int/iris/bitstream/handle/10665/43209/9241593180.pdf?sequence=1.
18. Schnadower D, Tarr PI, Gorelick MH, O’Connell K, Roskind CG, Powell EC, et al. Validation of the modified Vesikari score in children with gastroenteritis in 5 US emergency departments. J Pediatr Gastroenterol Nutr 2013;57:514–9.



19. Bailey B, Gravel J, Goldman RD, Friedman JN, Parkin PC. External validation of the clinical dehydration scale for children with acute gastroenteritis. Acad Emerg Med 2010;17:583–8.


20. Pringle K, Shah SP, Umulisa I, Mark Munyaneza RB, Dushimiyimana JM, Stegmann K, et al. Comparing the accuracy of the three popular clinical dehydration scales in children with diarrhea. Intl J Emerg Med 2011;4:58.




21. Wathen JE, MacKenzie T, Bothner JP. Usefulness of the serum electrolyte panel in the management of pediatric dehydration treated with intravenously administered fluids. Pediatrics 2004;114:1227–34.



22. Vega RM, Avner JR. A prospective study of the usefulness of clinical and laboratory parameters for predicting percentage of dehydration in children. Pediatr Emerg Care 1997;13:179–82.


23. Teach SJ, Yates EW, Feld LG. Laboratory predictors of fluid deficit in acutely dehydrated children. Clin Pediatr (Phila) 1997;36:395–400.



24. Steiner MJ, Nager AL, Wang VJ. Urine specific gravity and other urinary indices: inaccurate tests for dehydration. Pediatr Emerg Care 2007;23:298–303.

25. Conlon TW, Nishisaki A, Singh Y, Bhombal S, De Luca D, Kessler DO, et al. Moving beyond the stethoscope: diagnostic point-of-care ultrasound in pediatric practice. Pediatrics 2019;144:e20191402.



26. Di Pietro S, Falaschi F, Bruno A, Perrone T, Musella V, Perlini S. The learning curve of sonographic inferior vena cava evaluation by novice medical students: the Pavia experience. J Ultrasound 2018;21:137–44.




27. De Lorenzo RA, Holbrook-Emmons VL. Ultrasound measurement of inferior vena cava diameters by emergency department nurses. Adv Emerg Nurs J 2014;36:271–8.


29. Kosiak W, Swieton D, Piskunowicz M. Sonographic inferior vena cava/ aorta diameter index, a new approach to the body fluid status assessment in children and young adults in emergency ultrasound--preliminary study. Am J Emerg Med 2008;26:320–5.


30. Freedman SB, Vandermeer B, Milne A, Hartling L. Diagnosing clinically significant dehydration in children with acute gastroenteritis using noninvasive methods: a meta-analysis. J Pediatr 2015;166:908. –16. e1-6.


31. Salameh JP, Bossuyt PM, McGrath TA, Thombs BD, Hyde CJ, Macaskill P, et al. Preferred reporting items for systematic review and meta-analysis of diagnostic test accuracy studies (PRISMA-DTA): explanation, elaboration, and checklist. BMJ 2020;370:m2632.


32. Whiting P, Westwood M, Beynon R, Burke M, Sterne JA, Glanville J. Inclusion of methodological filters in searches for diagnostic test accuracy studies misses relevant studies. J Clin Epidemiol 2011;64:602–7.


33. Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan—a web and mobile app for systematic reviews. Syst Rev 2016;5:210.




34. Reitsma JB, Glas AS, Rutjes AW, Scholten RJ, Bossuyt PM, Zwinderman AH. Bivariate analysis of sensitivity and specificity produces informative summary measures in diagnostic reviews. J Clin Epidemiol 2005;58:982–90.


35. Rutter CM, Gatsonis CA. A hierarchical regression approach to metaanalysis of diagnostic test accuracy evaluations. Stat Med 2001;20:2865–84.


36. Young Ho L. Overview of the process of conducting meta-analyses of the diagnostic test accuracy. J Rheum Dis 2018;25:3–10.


37. Higgins JPT, Thompson SG. Quantifying heterogeneity in a metaanalysis. Stat Med 2002;21:1539–58.


38. McConnochie KM, Conners GP, Lu E, Wilson C. How commonly are children hospitalized for dehydration eligible for care in alternative settings? Arch Pediatr Adolesc Med 1999;153:1233–41.


39. Jauregui J, Nelson D, Choo E, Stearns B, Levine AC, Liebmann O, et al. External validation and comparison of three pediatric clinical dehydration scales. PLoS One 2014;9:e95739.



40. Sharma A, Mathur A, Stålsby Lundborg C, Pathak A. Incidence and risk factors for severe dehydration in hospitalized children in Ujjain, India. Int J Environ Res Public Health 2020;17:616.



41. Stengel D, Bauwens K, Sehouli J, Ekkernkamp A, Porzsolt F. A likelihood ratio approach to meta-analysis of diagnostic studies. J Med Screen 2003;10:47–51.



42. Li J, Fine JP, Safdar N. Prevalence-dependent diagnostic accuracy measures. Stat Med 2007;26:3258–73.


43. Harbord RM, Whiting P. Metandi: meta-analysis of diagnostic accuracy using hierarchical logistic regression. Stata J 2009;9:211–29.


44. El Amrousy D, Gamal R, Elrifaey S, Hassan S. Non-invasive assessment of significant dehydration in infants using the inferior vena cava to aortic ratio: is it useful? J Pediatr Gastroenterol Nutr 2018;66:882–6.


45. Chen L, Hsiao A, Langhan M, Riera A, Santucci KA. Use of bedside ultrasound to assess degree of dehydration in children with gastroenteritis. Acad Emerg Med 2010;17:1042–7.



46. Kim JS, Lee Yg, Cho Hy, Park Dh, Jun J, Shin Ty, et al. Usefulness of bedside emergency ultrasound for detecting significant dehydration in children. J Korean Soc Emerg Med 2010;21:696–703.
47. Chen L, Kim Y, Santucci KA. Use of ultrasound measurement of the inferior vena cava diameter as an objective tool in the assessment of children with clinical dehydration. Acad Emerg Med 2007;14:841–5.


48. Trevethan R. Sensitivity, specificity, and predictive values: foundations, pliabilities, and pitfalls in research and practice. Front Pub Health 2017;5:307.



49. Trikalinos TA, Balion CM, Coleman CI, Griffith L, Santaguida PL, Vandermeer B, et al. Chapter 8: meta-analysis of test performance when there is a “gold standard”. J Gen Intern Med 2012;27 Suppl 1(Suppl 1): S56–66.




50. Whiting P, Rutjes AW, Reitsma JB, Glas AS, Bossuyt PM, Kleijnen J. Sources of variation and bias in studies of diagnostic accuracy: a systematic review. Ann Intern Med 2004;140:189–202.


51. Sedighi I. Interpretation of diagnostic tests: likelihood ratio vs. predictive value. Iran J Pediatr 2013;23:717.


52. Ebell. Likelihood ratios, predictive values, and post-test probabilities [Internet] Leawood (KS): American Academy of Family Physicians; c2005 [cited 2022 Oct 18]. Available from: https://www.aafp.org/dam/AAFP/documents/journals/afp/Likelihood_Ratios.pdf.
53. Moons KG, Harrell FE. Sensitivity and specificity should be deemphasized in diagnostic accuracy studies. Acad Radiol 2003;10:670–2.


54. Leeflang MM, Bossuyt PM, Irwig L. Diagnostic test accuracy may vary with prevalence: implications for evidence-based diagnosis. J Clin Epidemiol 2009;62:5–12.


55. Moons KGM, Biesheuvel CJ, Grobbee DE. Test research versus diagnostic research. Clin Chem 2004;50:473–6.



56. Koeller E, Robertson SE, Garbern SC, Monk G, Nasrin S, Alam NH, et al. Ultrasound adds no benefit to clinical exam for predicting dehydration in children with acute diarrhea in a resource-limited setting. J Ultrasound Med 2019;38:685–93.



57. Bossuyt PM, Irwig L, Craig J, Glasziou P. Comparative accuracy: assessing new tests against existing diagnostic pathways. BMJ 2006;332:1089–92.



58. Levine AC, Shah SP, Umulisa I, Munyaneza RB, Dushimiyimana JM, Stegmann K, et al. Ultrasound assessment of severe dehydration in children with diarrhea and vomiting. Acad Emerg Med 2010;17:1035–41.


59. Zhou AZ, Green RS, Haines EJ, Vazquez MN, Tay ET, Tsung JW. Interobserver agreement of inferior vena cava ultrasound collapse duration and correlated outcomes in children with dehydration. Pediatr Emerg Care 2022;38:13–6.


60. Horoz OO, Yildizdas D, Aslan N, Coban Y, Misirlioglu M, Haytoglu Z, et al. Sonographic measurements of inferior vena cava, aorta, anda IVC/aorta ratio in healthy children. Niger J Clin Pract 2022;25:825–32.


61. Finnerty NM, Panchal AR, Boulger C, Vira A, Bischof JJ, Amick C, et al. Inferior vena cava measurement with ultrasound: what is the best view and best mode? West J Emerg Med 2017;18:496–501.



62. Kathuria N, Ng L, Saul T, Lewiss RE. The baseline diameter of the inferior vena cava measured by sonography increases with age in normovolemic children. J Ultrasound Med 2015;34:1091–6.



63. Via G, Tavazzi G, Price S. Ten situations where inferior vena cava ultrasound may fail to accurately predict fluid responsiveness: a physiologically based point of view. Intensive Care Med 2016;42:1164–7.



64. Airapetian N, Maizel J, Alyamani O, Mahjoub Y, Lorne E, Levrard M, et al. Does inferior vena cava respiratory variability predict fluid responsiveness in spontaneously breathing patients? Crit Care 2015;19:400.




65. Caplan M, Durand A, Bortolotti P, Colling D, Goutay J, Duburcq T, et al. Measurement site of inferior vena cava diameter affects the accuracy with which fluid responsiveness can be predicted in spontaneously breathing patients: a post hoc analysis of two prospective cohorts. Ann Intensive Care 2020;10:168.










 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


