Article Contents
| Clin Exp Pediatr > Volume 66(3); 2023 |
|
Abstract
Background
Purpose
Methods
Results
Footnotes
Funding
This study was supported by a research grant for the project of the Korean Academy of Pediatric Allergy and Respiratory Disease.
Author contribution
Conceptualization: Kang EK, Kim CK, Yang HJ, Data curation: Sol IS, Lee E, Yang HJ, Yum HY, Lee MH, Chu MA, Kim HB, Seo JH, Shim JY, Ahn JY, Jang YY, Chung HL, Jung EH, Kim CH, Kang EK, Kim CK. Formal analysis: Kim CS. Methodology: Yang HJ, Lee YJ, Kim K, Kim BS, Lee KS, Han MY, Hong SJ, Kim JT; Software: Moon HJ, Sol IS. Investigation: Sol IS, Kang EK, Yang HJ. Writing - original draft: Sol IS, Kang EK. Writing - review & editing: Sol IS, Kang EK, Kim CK
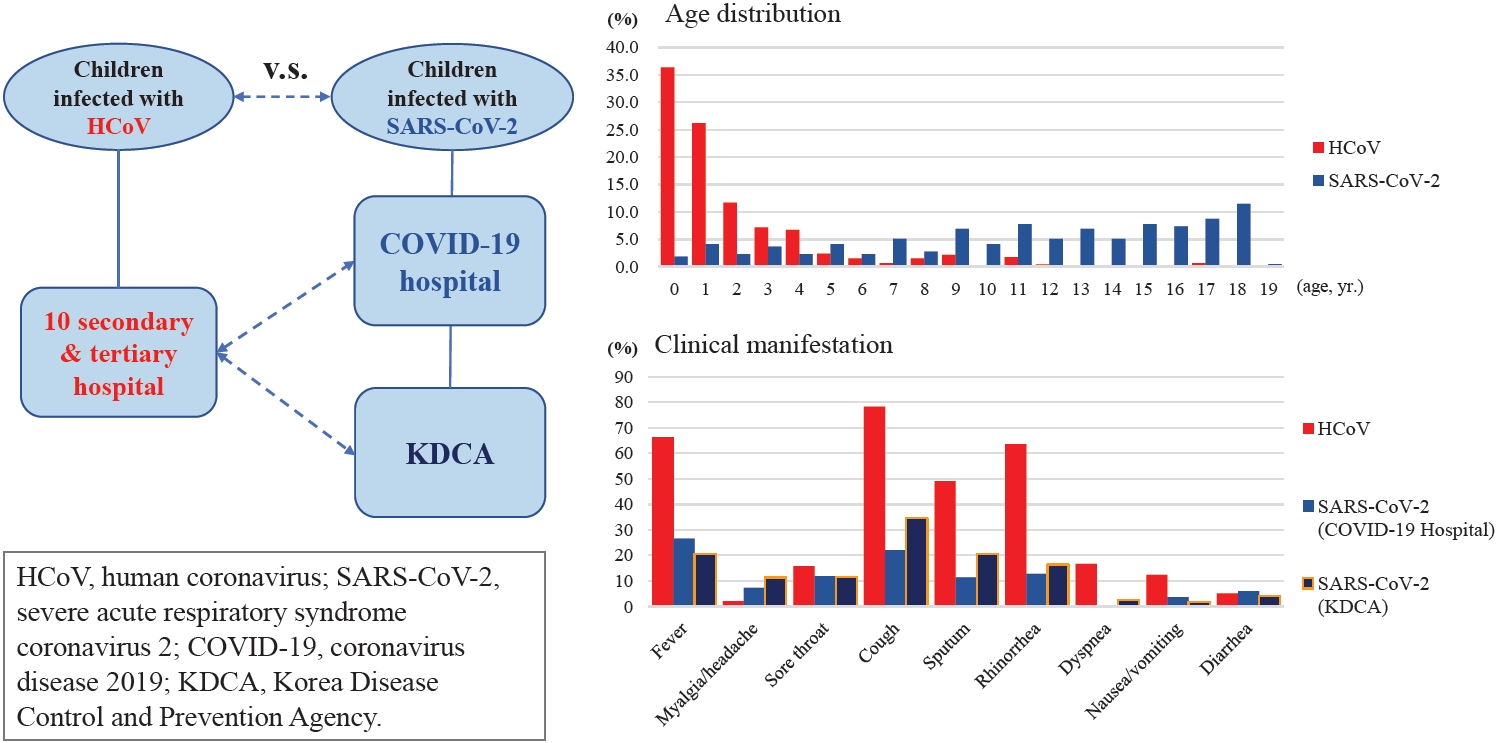
Fig. 1.
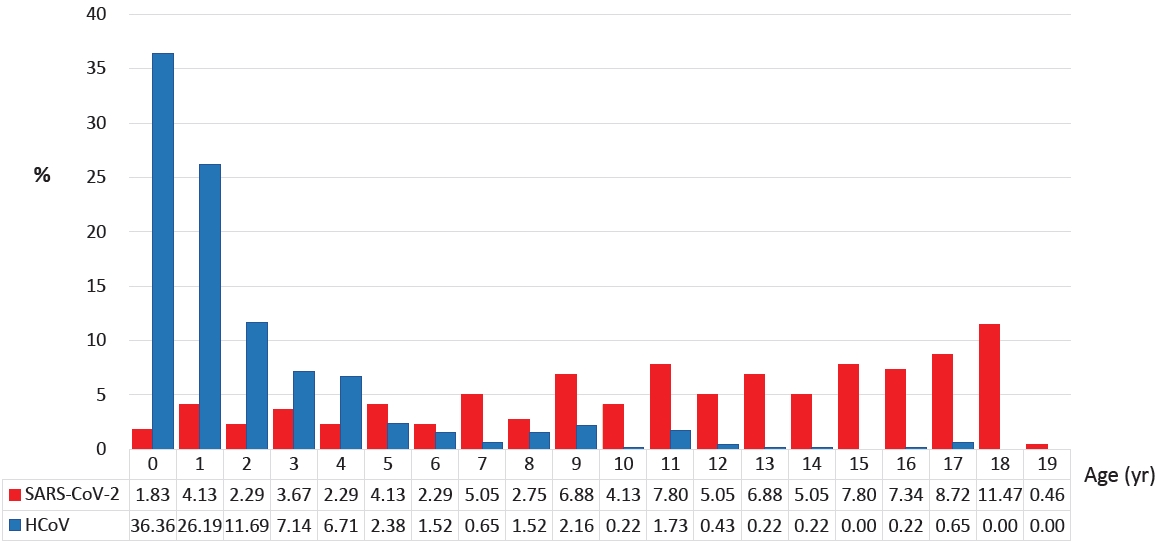
Table 1.
Values are presented as mean±standard deviation or number (%).
HCoV, human coronavirus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; NS, nonspecific; COVID-19, coronavirus disease 2019.
P values were calculated using Student t test or Welch t test for continuous variables and using the chi-squared test or Fisher exact test for categorical variables.
The hospitalization period for SARS-CoV-2 comprised the time from the date of infection notification to the date of quarantine release or death.
Boldface indicates a statistically significant difference with P<0.05.
Table 2.
Values are presented as number (%).
HCoV, human coronavirus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; COPD, chronic obstructive lung disease; NS, nonspecific.
P values were calculated using the chi-square test or Fisher exact test for categorical variables.
Boldface indicates a statistically significant difference with P<0.05.
Table 3.
Table 4.
Values are presented as mean±standard deviation or number (%).
HCoV, human coronavirus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; COVID-19, coronavirus disease 2019.
P values were calculated using the chi-square test or Fisher exact test for categorical variables.
Boldface indicates a statistically significant difference with P<0.05.
Table 5.
Values are presented as mean±standard deviation or median (range).
HCoV, human coronavirus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; COVID-19, coronavirus disease 2019; WBC, white blood cell; PLT, platelets; AST, aspartate aminotransferase; ALT, alanine transferase; CRP, C-reactive protein; LDH, lactate dehydrogenase.
P values were calculated using Student t test or Welch t test for continuous variables.
Boldface indicates a statistically significant difference with P<0.05.
Table 6.
Values are presented as mean±standard deviation or median (range).
HCoV, human coronavirus; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; COVID-19, coronavirus disease 2019; HFNC, high flow nasal cannula; NS, nonspecific.
P values were calculated using the chi-square test or Fisher exact test for categorical variables.
Boldface indicates a statistically significant difference with P<0.05.
References
-
METRICS

-
- 1 Crossref
- 0 Scopus
- 8,035 View
- 217 Download





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation

