Urinary tract infections (UTIs) are the most common serious bacterial infections in children [1]. The most common pathogens causing UTIs are Enterobacteriaceae, such as Escherichia coli and Klebsiella species [2]. Antimicrobial drug resistance to fluoroquinolone, cephalosporin, and carbapenem among Enterobacteriaceae has spread globally over the past few decades and become a pressing problem [3]. The dissemination of drug-resistant organisms is troublesome for clinicians when selecting empirical antibiotics. Patients with UTIs were historically administered broad-spectrum cephalosporin as the empirical therapy. Carbapenem is the definitive therapy for infections caused by extended-spectrum β-lactamases (ESBL)–producing bacteria. However, carbapenemsparing options are on the rise for mild infections with ESBL producers because its overuse is leading to the emergence of carbapenem-resistant organisms.
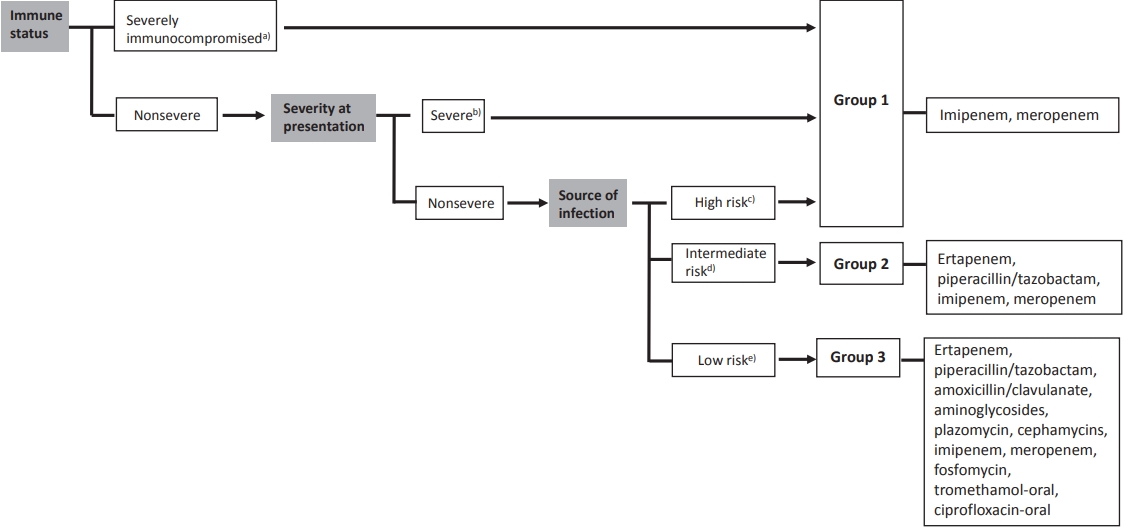
Recent studies have focused on the noninferiority of noncarbapenem therapy for the treatment of ESBL-producing Enterobacteriaceae infections to reduce carbapenem utilization [4-7]. A review article examined noncarbapenem β-lactam (cephamycin, cefepime, piperacillin/tazobactam, and newer β-lactam/β-lactamase inhibitors) therapy for ESBL-producing bacterial infections. The authors suggested that noncarbapenem could be considered in patients with mild to moderate low-inoculum infections [6]. A recent literature review summarized published articles regarding the treatment of ESBL-producing Enterobacteriaceae infections. Patients were divided into 3 groups: group 1, severe or nonsevere infections from high-risk sources and/or severely immunocompromised patients; group 2, nonsevere infections and intermediate-risk sources; and group 3, nonsevere infections and low-risk sources (Fig. 1). They concluded that carbapenem should be the choice of drug for the treatment of ESBL-producing Enterobacteriaceae in severe infections, whereas other antimicrobial agents could be considered for mild infections such as UTIs [7]. Thus, using noncarbapenem therapy for treating UTIs caused by ESBL-producing bacteria could be an effective way to prevent carbapenem overuse.
Furthermore, children with vesicoureteral reflux (VUR) are at high risk for acute and recurrent pyelonephritis [8]. In patients with VUR, it is unknown whether carbapenem therapy can reduce the short-term recurrence. Therefore, a prospective study is needed to compare the treatment outcomes of carbapenemtreated and non–carbapenem-treated patients diagnosed with UTIs due to ESBL producers underlying VUR. To enable a careful conclusion, large samples and multivariate analysis are required.
If UTIs caused by ESBL-producing bacteria are alleviated through empirical noncarbapenem therapy, switching to carbapenem therapy is a difficult decision for clinicians. To solve this challenge and develop management guidelines, additional large-scale randomized controlled trials are required.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


