Article Contents
| Clin Exp Pediatr > Volume 64(9); 2021 |
|
Abstract
Background
Malnutrition has a high incidence among pediatric surgical patients and contributes to increased risks of postoperative complications and extended hospital stays.
Purpose
The present study aimed to determine the influence of preoperative nutritional status on the postoperative outcomes of pediatric patients who underwent elective gastrointestinal (GI) surgery.
Methods
This prospective observational study was conducted at Cairo University Specialized Pediatric Hospital. According to the designated inclusion criteria, 75 surgical cases of both sexes were included. A structured questionnaire was developed and administered. This questionnaire included 3 main sections: demographic data and nutritional status parameters at admission and discharge. Pre- and postoperative nutritional statuses were compared.
Results
According to both the subjective global nutritional assessment and STRONGKIDS score Questioner, more than 60% of patients in the upper GI patient group were at risk of malnutrition. Wasting status was most common in the upper GI patient group (67%; vs. 39.1% in the lower GI group). Underweight status was more common in the hepatobiliary and upper GI patient groups (nearly 50% for each group) than in the lower GI group (30.4%). On the other hand, stunted patients had a higher incidence of complications and a prolonged hospital stay (P=0.003 and P=0.037, respectively), while underweight lower GI patients experienced a prolonged hospital stay (P=0.02). A higher proportion of patients with preoperative anemia than those without preoperative anemia required a blood transfusion (P=0.003).
Nutrition is crucial to maintain normal growth, physiologic homeostasis, and substantial resistance to infection [1]. Malnutrition is a highly-occurring incident among pediatric surgical patients particularly; patients undergoing gastrointestinal (GI) surgery as a result of anorexia, dietary restriction, malabsorption, increased intestinal losses, or altered nutrient requirements [2]. Moreover, the hypermetabolic state induced by surgical stress and postoperative restriction of oral intake can exaggerate malnutrition [3]; this results in debilitating immunity and stress resistance. Therefore, hypermetabolic states are regarded as a commutative risk factor for complications such as infection, poor wound healing, GI tract bacterial overgrowth, prolonged hospital stay, and mortality [4-6]. Parameters for the assessment of body energy and protein reserves have been developed over the years providing a reliable assessment of nutritional status with various methods that could be objective or subjective [7]. There are 2 main classes of objective assessment: anthropometric measures and serum proteins levels. Subjective assessments included questionnaires which depend on patient history and recall [8,9]. The present study aims to figure out the preoperative nutritional status, and its influence on postoperative outcome of pediatric patients who underwent elective GI surgery.
This prospective observational study took place at Cairo University Specialized Pediatric Hospital (CUSPH) during the period from July 2017 to January 2018. To achieve the aim of the study, we adopted the following methodology: forming study population, and developing and administering a questionnaire.
The candidates for our study were recruited from the Pediatric Surgery Department at CUSPH during the study period. We included patients whose age ranged from 1 to 60 months (this age group was vulnerable for malnutrition). The neonate had another neonatal surgical department. Based on the designated inclusion criteria, 75 surgical cases were included in our study from both genders. We excluded from the study the following patients: (I) emergency surgical cases
A structured questionnaire was developed after reviewing the literature to determine the parameters of nutritional assessment and clinical outcomes (included Supplementary material).
The questionnaire was composed of 3 main sections: demographic data of the cases, nutritional status parameters (upon admission and upon discharge), and postoperative clinical outcomes.
The demographic data identified the enrolled patients’ age, sex, and associated comorbid conditions. Nutritional history involved presence of breast feeding, problems with weaning, and previous nutritional support.
The nutritional assessment tools were divided into objective and subjective. The objective parameters included: measurements of weight, length, mid arm circumference (MAC), triceps skin fold thickness (TSF), mid arm muscle circumference (MAMC) as well as 2 biochemical markers; albumin and hemoglobin levels. The subjective methods were based on history and clinical judgment, and they included subjective global nutritional assessment (SGNA) and STRONGKIDS score.
The mechanical scale and stadiometer were used to measure weight and length, respectively. Z scorees for weight-for-age, length-for-age, and weight for length-for-age of infants from 1 to 60 months of age were reported using 2000 CDC growth charts [10].
MAC was taken at a midpoint between the acromion and the olecranon with the arm flexed at 90° angle and with the tape measure perpendicular to the long axis of the arm. TSF was measured using a skin fold caliper at the point previously marked for the MAC, while MAMC was calculated through the following formula (MAC–[3.14×TSF]). Z scorees for arm circumference-for-age and triceps skin fold-for-age of infants from 3 to 60 months of age were reported using 2007 WHO Child Growth Standards [11].
SGNA included: weight change, dietary intake, oral motor skill development, feeding tolerance, functional capacity, muscle wasting, and ankle edema. In assigning overall score, all items were considered in the context of each other. The patients were categorized as either well-nourished if normal ratings in all or most items were achieved or as malnourished in case of moderate to severe ratings in most or all items [8].
STRONGKIDS included the following. Firstly, we considered the presence of an underlying illness, the prospect of a major surgery, and clinical nutritional assessment. Secondly, we considered the presence of one of the following items: (1) excessive diarrhea; (2) reduction of food intake during the last few days; (3) preexisting nutritional intervention; and (4) inability to consume adequate nutritional intake due to pain, weight loss or stationary weight. Assessment score ranged from 0 to 5. Patients with a score of 0 were classified as being at low risk for malnutrition; those with a score between 1 and 3 were at moderate risk of malnutrition; those with a score more than 4 were at high risk of malnutrition [9].
In the third section, the postoperative outcome assessment included the existence of complications and length of hospital stay.
Complications were described according to the presence of postoperative infections that could be local, systemic, or both. Noninfectious complications involved the need of postoperative blood transfusion and intensive care unit admission. A standardized complication classification system of Clavien-Dindo was used to monitor postoperative complications [12]. Patients were categorized according to the length of hospital stay into 2 groups: short stay (less than 7 days) and long stay (more than 7 days).
Throughout the study, we did not interfere with nutritional support provided by the clinical nutrition team which relies on encouraging breast feeding or providing artificial formula or complementary diet with proper calculation of caloric and protein requirements according to age nutritional support provided orally except for gastrostomy patients. Micronutrients supplied if indicated.
Both patients’ nutritional assessment and reporting postoperative outcomes were carried out by a single investigator who received an adequate training in the Pediatric Department at CUSPH. The training included the different methods of assessment of the nutritional parameters (Supplementary material).
We obtained an informed consent from caregivers before conducting the questionnaire. Moreover, we also obtained an ethical approval from the Ethical Committee of Cairo University (approval number and date: I-151015/2017).
Statistical analysis was performed using SPSS ver. 16.0 (SPSS Inc., Chicago, IL, USA) and Microsoft Excel 2013 (15.0.4420.1017) 32-bit software. Normally distributed continuous variables were expressed as mean and standard deviations. Nonnormally distributed data were expressed by median and range. Comparison of numerical variables was done using Student t test and Fisher exact test for independent samples in comparing 2 groups of normally distributed data, and Mann Whitney U test was used for nonnormal data. Comparison of numerical variables measured before and after surgical intervention was done using paired t test for normally distributed data. To examine the relationship among the 3 groups or more, an analysis of variance was performed. Correlation between continuous data was analyzed by Spearman correlation. P value less than 0.05 was considered statistically significant.
Each preoperative subjective and objective parameter was correlated discretely to the postoperative complications and length of hospital stay to determine the impact of the nutritional status on the postoperative outcomes. The postoperative nutritional status was also considered.
Seventy-five patients were enrolled in the study. Including 47 males (63%) and 28 females (37%) with a median age of 10 months (range, 1–60 months). Diagnoses of the patients are displayed in Table 1. Eight patients (11%) had associated comorbid conditions, 2 cases are obese, 2 cases had atrial septal defect, 1 case is asthmatic, 1 had cerebral palsy, 1 is Down syndrome, and 1 had meromilia.
Preoperative weight-for-age z score was less than -2 (underweight) in 28 patients while 17 patients had length-for-age z score less than -2 were (stunted), By using the subjective global nutritional assessment (SGNA) score, it showed that 61 patients (81%) were well-nourished and 14 patients (19%) were malnourished. On the other hand, the STRONGKIDS score found that 13 patients (18%) were at moderate risk of malnutrition, while 25 patients (33%) were at high risk for malnutrition. TSF and MAC were applicable to be interpreted in 62 patients. TSF z score less than -2 was found in 40 patients, while MAC z score less than -2 was found in 19 patients. Interpreting the MAMC with percentiles, we found that 15 patients were ≤5th percentile. The preoperative laboratory assessment revealed the mean of the serum albumin was 3.8 (standard deviation [SD], ±0.5), mean of serum hemoglobin level was 10.5 (SD, ±1.3), the anemic patients were 48 (64% of the included patients).
We evaluated included patients on day 7 postoperative because this is the standard duration for hospital stay of most of uncomplicated surgical cases. We compared the different nutritional assessment parameters preoperatively and postoperatively in each group. Accordingly, upper GI patients’ group showed a significant lower difference in postoperative as regard MAC (P=0.01) while STRONGKIDS score showed lower risk in postoperative with statistically significant difference (P=0.0007). Furthermore, a significant lower difference between pre- and postoperative values TSF and MAC (P=0.01, P=0.04, respectively) in the hepatobiliary patients’ group (Table 2).
Depicting the postoperative complications using Clavien-Dindo classification is featured in (Table 3). Grade I represented surgical site infection (SSI) with no systemic illness; this was obvious among upper and lower GI patients as we found 2 and 9 patients, respectively. Grade II represented systemic infections and blood transfusion. It occurred among the patients of upper GI, lower GI, and hepatobiliary, 3, 7, and 4 patients were detected, respectively. Grade IIIb represented patients who needed surgical intervention. It occurred with 4 patients in the lower GI group and with 1 patient in the hepatobiliary group. Grade IV represented the need for admission in the intensive care unit; this was experienced by 1 patient among the hepatobiliary patients’ group. Grade V represented death; this occurred in 1 case among upper GI patients’ group and another case among hepatobiliary patients’ group.
The overall incidence of postoperative infectious complications was 33.3% among the 3 different anatomical patients’ group. The detected cases among the lower GI patients’ group were 16 cases, while among the upper GI and the hepatobiliary patients’ groups were 5 and 4, respectively.
We noticed that 17.3% (n=13) of included patients required postoperative blood transfusion. All of them had moderate anemia except only 3 cases had mild degree of anemia preoperatively. Two cases among the upper GI, 8 cases among the lower GI, and 3 cases among hepatobiliary patients’ groups received postoperative blood transfusion.
We classified the patients into 2 groups according to the postoperative hospital stay. Twenty-eight experienced long hospital stay for more than 7 days. We found that 8 cases (53%) among the upper GI patients’ group, 14 cases (30%) among the lower GI group, and 6 cases (43%) among the hepatobiliary patients’ group.
When we compared preoperative anthropometric measures for all patients to postoperative complications, we found a significantly lower difference with the preoperative length-for-age z score and weight-for-age z score in complicated cases (P=0.001, P=0.046 respectively) (Table 4; Fig. 1A, B). We also realized that the preoperative length-for-age z score was significantly lower in cases with prolonged hospital stay (P=0.022) (Table 5, Fig. 2).
When comparing anemic and nonanemic patients as regard the postoperative blood transfusion revealed that preoperative anemic patients had higher incidence of blood transfusion than nonanemic group with statistically significant difference (P=0.003). Neither presence of postoperative complications and duration of hospital stay were related to history of blood transfusion.
Furthermore, STRONGKIDS and SGNA scores were not correlated for both postoperative complications and length of hospital stay. Neither weight for length, TSF, MAC, nor MAMC were correlated to postoperative complications and length of hospital stay.
A remarkable prolonged length of hospital stay was noticed with increasing the Clavien-Dindo classification grade of complication (P=0.001).
SGNA is a method of nutritional assessment based on clinical judgment, and it can be adapted to identify presurgical malnutrition, while STRONGKIDS is a nutritional screening tool that has been developed according to the newest ESPEN (European Society for Clinical Nutrition and Metabolism) guidelines [8,9]. The upper GI group of patients showed the highest prevalence of preoperative malnourishment by both SGNA and STRONGKIDS scoring system.
In this study, the incidence of wasting in included patients preoperatively was 46.6%. The highest percentage was found in upper GI group and hepatobiliary group. Furthermore, we found that the rate of underweight was higher among upper GI and hepatobiliary patients’ groups nearly 50% of each group. Upper GI group had the highest percentage of wasting as well as underweight. This could be attributed to periods of decreased intake, interrupted feeding, defective intake, malabsorption, and intestinal losses imposed by the variety of surgical interventions that contributed to the development of undernutrition despite efforts to provide adequate energy and other nutrients [2,13].
The nutritional assessment is the cornerstone in management of surgical patients, should not rely on weight evaluation only, other anthropometric measurements should be used such as TSF, MAC, and MAMC. Because of these are not affected by fluid accumulation. Some patients experienced substantial third space fluid accumulation [11], particularly if they had decreased serum albumin levels in certain surgical procedure such as the correction of pure esophageal atresia, endo-anal pull through for Hirschsprung disease, Kasai procedure, and excision of choledochal cyst with hepatoduodenostomy [1].
Weight is not the best parameter in patients with chronic liver diseases since visceromegaly and ascites may mask malnutrition. Almost 50% of our hepatobiliary patients had abnormal MAMC and TSF measurements. It could be attributed to the increased energy expenditure in this category of patients and to fat malabsorption [14].
The highest incidence of hypoalbuminemia was among hepatobiliary patients’ group. This could be explained by increasing the utilization of protein as source of energy and the reduction in liver capacity for protein synthesis [14]. Serum albumin is considered a negative acute phase reactant with longer half-life. This does not reflect the nutritional status, and it is worthy to be assessed with prealbumin that has shorter half-life [2,15,16]. Therefore, albumin is considered a predictor of morbidity except in hepatic patients [8,17].
Nearly 64% of our patients were anemic. One-third of anemic patients required blood transfusion so the preoperative assessment of hemoglobin is mandatory. The highest prevalence of anemia was among hepatobiliary and lower GI patient’ groups (76% and 66%, respectively). This could be multifactorial due to the underlying surgical procedures, associated morbidity, micronutrient deficiencies, and recurrent infections [18].
Stunting is indicator of chronic malnutrition which is major risk for morbidity hence postoperative complication [5]. It was recorded in 2014 by Egypt Demographic and Health Survey that the prevalence of stunting among children under 5% was 21% [19]. This could be explained by the fact of low diet quality due to low socio-economic status of the population. Furthermore, mothers start lactation with relatively poor energy stores, which is insufficient to produce a sufficient amount of milk for an optimal infant growth.
When pre- and postoperative anthropometric data were compared to each other, no momentous difference was detected. However, there was a significant reduction in postoperative TSF, MAC, and MAMC among both upper GI and hepatobiliary patients’ groups reflecting the changes in muscles and fat stores. This could denote the hypercatabolic state due to surgical stress [3]. On the other hand, there were no notable changes in TSF, MAC, and MAMC among lower GI patients’ group. It is worth mentioning that this category started feeding within the first 24 hours after surgery. Patients in the lower GI group experienced few hours of minimal nutritional intake which preserved their nutritional state postoperatively. Indeed, good caloric and protein intake postoperatively help to maintain these parameters in the normal range [20-22].
SSI is considered the most common complication following major GI surgery, affecting between 25%–40% of patients. The rate of SSI doubles from low-income to high-income settings, persisting after risk adjustment for patients and hospital confounders [23]. We found that one-third [24] of our patients experienced infectious complications postoperatively. The effects of SSI can be life-threatening. They are related to one-third of postoperative deaths. Furthermore, SSI can cause pain and discomfort, increase length of hospital stay, and further amplify the risk of potential nosocomial infection. Blood transfusion is accounted a noninfectious complication, its incidence with similar surgeries in middle-income countries is about 31.1% [23].
One-third of our patients who had infectious complications are wasted. Preoperative malnutrition has shown to increase complications following surgery and the length of hospital stay. Therefore, identifying patients who are at risk of malnutrition prior to surgery is important to optimize their nutritional status prior to the procedure and to minimize suboptimal outcomes. The lack of a universally accepted definition for malnutrition is considered a restriction in evaluating preoperative patients for malnutrition. Furthermore, because the aims of the tools differ, judging the inferiority or superiority of one tool over the other is out of question. Therefore, much of the literature evaluates a variety of nutritional markers using numerous scoring methods [21,24]. We followed the same principle and used different markers to correlate with the postoperative outcome.
The preoperative length-for-age was significantly related to both postoperative complications either infectious or noninfectious as well as postoperative length of hospital stay. This goes with the finding of Secker and Jeejeebhoy [8] as the length-for-age of patients who underwent abdominal and thoracic surgeries was correlated significantly with length of hospital stay. Lengthfor-age is generally considered an indicator of the patient’s long-term or previous nutritional status; thus, these results suggest that patients with chronic malnutrition had increased length of hospital stay [8,25]. Nevertheless, we found a relation between preoperative weight-for-age and postoperative complications with statistical significance (P=0.046). Incidence of complications was substantially related to the length of hospital stay [25,26,27].
It is worth mentioning that one of limitations we faced in this study was that the patients’ assessment was performed by a single investigator. This resulted in limited study duration and a small size of patients’ population.
In pediatric surgical patients, nutritional assessment is an indispensable aspect of an initial evaluation. Postoperative complications have a higher incidence among stunted patients, while long hospital stay occurs with both stunted and underweight patients. Anemic patients should be corrected before the procedure as it significantly indicator for postoperative complication.
Supplementary material
Supplementary questionnaire can be found via https://doi.org/10.3345/cep.2020.00458.
Supplementary material.
Questionnaire of assessing preoperative nutritional status and postoperative outcomes
Fig. 1.
Box plots of the z scores of the entire study population with or without complications including normally growing as well as underweight (A) or stunted patients (B).
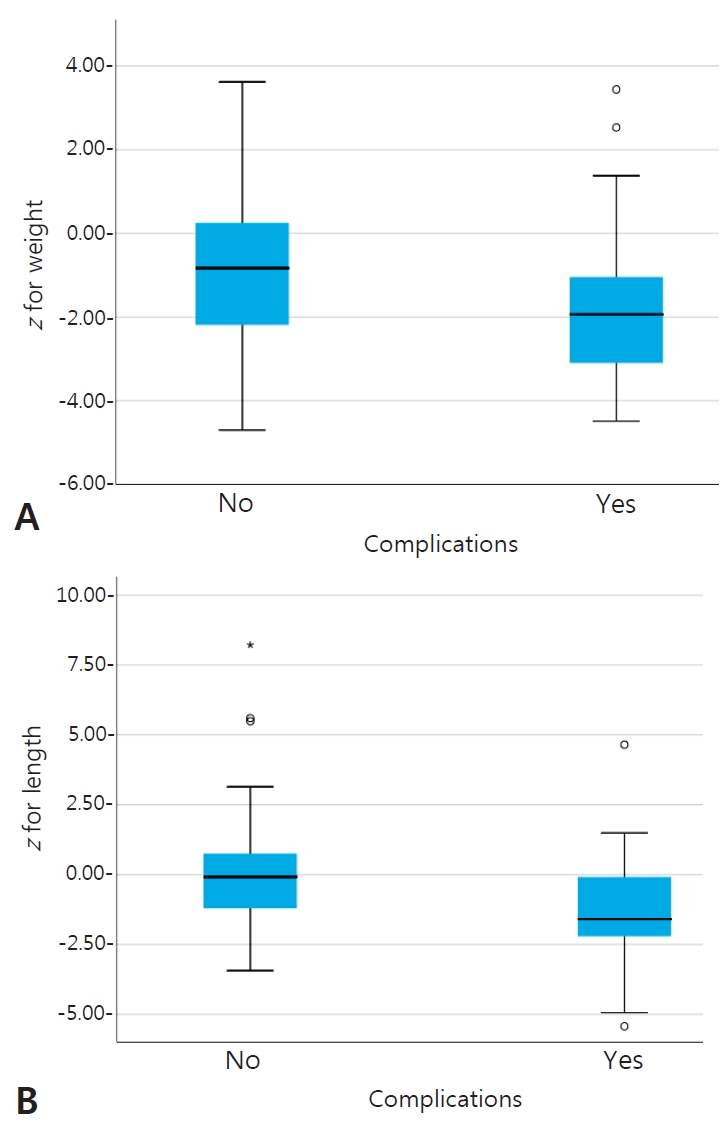
Fig. 2.
Box plot of the z scores of the entire study population with a short or long hospital stay including normally growing as well as underweight or stunted patients.

Table 1.
Distribution of cases among the 3 anatomical groups by disease and surgical procedure
Table 2.
Pre- and postoperative nutritional assessments of patients by surgical group
| Variable |
Upper GI group (n=15) |
P value |
Lower GI group (n=46) |
P value |
Hepatobiliary group (n=14) |
P value | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Pre | Post | |||||
| Weight-for-age z score | 1 | 0.8 | 0.2 | |||||||
| Average | 8 | 7 | 32 | 30 | 7 | 11 | ||||
| Underweighta) | 7 | 8 | 14 | 16 | 7 | 3 | ||||
| Length-for-age z score | ||||||||||
| Average | 12 | 34 | 12 | |||||||
| Stuntedb) | 3 | 12 | 2 | |||||||
| Weight for Length z score | 1 | 1 | 0.7 | |||||||
| Average | 5 | 5 | 28 | 28 | 7 | 9 | ||||
| Wastedc) | 10 | 10 | 18 | 18 | 7 | 5 | ||||
| TSF z score | 0.09 | 0.2 | 0.01 | |||||||
| Average | 7 | 6 | 15 | 16 | 0 | 0 | ||||
| z score less than -2d) | 5 | 6 | 28 | 27 | 7 | 7 | ||||
| MAC z score | 0.01 | 0.17 | 0.04 | |||||||
| Average | 8 | 7 | 32 | 33 | 3 | 1 | ||||
| z score less than -2d) | 4 | 5 | 11 | 10 | 4 | 6 | ||||
| MAMC percentile | 0.6 | 0.4 | 1 | |||||||
| Average | 11 | 9 | 41 | 37 | 8 | 7 | ||||
| Percentile less than 5th | 4 | 6 | 6 | 10 | 5 | 6 | ||||
| Hemoglobin | 1 | 0.1 | 0.1 | |||||||
| Normal | 9 | 8 | 15 | 8 | 4 | 1 | ||||
| Anemiae) | 6 | 7 | 31 | 38 | 10 | 13 | ||||
| Albumin | 0.13 | 0.08 | 0.07 | |||||||
| Normal | 11 | 6 | 39 | 31 | 6 | 1 | ||||
| Hypoalbuminemiaf) | 4 | 9 | 7 | 15 | 8 | 13 | ||||
| STRONGKIDS | 0.0007 | 1 | 1 | |||||||
| Low risk of malnutrition | 6 | 15 | 42 | 43 | 14 | 13 | ||||
| Moderate risk of malnutritiong) | 9 | 0 | 4 | 3 | 0 | 1 | ||||
| SGNAh) | 0.27 | 0.7 | 0.2 | |||||||
| Nonmalnourished | 5 | 9 | 42 | 40 | 14 | 11 | ||||
| Malnourished | 10 | 6 | 4 | 6 | 0 | 3 | ||||
Table 3.
Postoperative complications by Clavien-Dindo classification
| Complication | Upper GI | Lower GI | Hepatobiliary |
|---|---|---|---|
| Grade I | 2 | 9 | 0 |
| Grade II | 3 | 7 | 4 |
| Grade IIIa | 0 | 0 | 0 |
| Grade IIIb | 0 | 4 | 1 |
| Grade IV | 0 | 0 | 1 |
| Grade V | 1 | 0 | 1 |
Table 4.
Comparison of anthropometric measurements and hospital stay durations of the included patients classified by postoperative complication status
Table 5.
Comparison of anthropometric measurements and complications of the included patients classified by hospital stay duration
References
1. Falcão MC, Tannuri U. Nutrition for the pediatric surgical patient: approach in the peri-operative period. Rev Hosp Clin Fac Med Sao Paulo 2005;57:299-308.

2. Mazaki T, Ebisawa K. Enteral versus parenteral nutrition after gastrointestinal surgery: a systematic review and meta-analysis of randomized controlled trials in the English literature. J Gastrointest Surg 2008;12:739-55.


3. Cardinale F, Chinellato I, Caimmi S, Peroni DG, Franceschini F, Miraglia Del Giudice M, et al. Perioperative period: immunological modifications. Int J Immunopathol Pharmacol 2011;24(3 Suppl): S3-12.


5. Sungurtekin H, Sungurtekin U, Balci C, Zencir M, Erdem E. The influence of nutritional status on complications after major intraabdominal surgery. J Am Coll Nutr 2004;23:227-32.


6. Cano NJ, Heng AE, Pison C. Multimodal approach to malnutrition in malnourished maintenance hemodialysis patients. J Ren Nutr 2011;21:23-6.


7. Bauer K, Bovermann G, Roithmaier A, Götz M, Pröiss A, Versmold HT. Body composition, nutrition, and fluid balance during the first two weeks of life in preterm neonates weighing less than 1500 grams. J Pediatr 1991;118:615-20.


8. Secker D, Jeejeebhoy K. Subjective Global Assessment for children. Am J Clin Nutr 2007;85:1083-9.

9. Huysentruyt K, Alliet P, Muyshont L, Rossignol R, Devreker T, Bontems P, et al. The STRONG kids nutritional screening tool in hospitalized children: a validation study. Nutrition 2013;29:1356-61.


10. Kuczmarski RJ, Kuczmarski MF, Roche AF. 2000 CDC growth charts: background for clinical application. Top Clin Nutr 2002;17:15-26.
11. Van den Broeck J, Willie D, Younger N. The World Health Organization child growth standards: expected implications for clinical and epidemiological research. Eur J Pediatr 2009;168:247-51.


12. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13.


13. Rogers EJ, Gilbertson HR, Heine RG, Henning R. Barriers to adequate nutrition in critically ill children. Nutrition 2003;19:865-8.


14. Yang CH, Perumpail BJ, Yoo ER, Ahmed A, Kerner JA Jr. Nutritional needs and support for children with chronic liver disease. Nutrients 2017;9:1127.



15. Fuhrman MP. The Albumin-nutrition connection: separating myth from fact. Nutrition 2002;18:199-200.


16. Wessner S, Burjonrappa S. Review of nutritional assessment and clinical outcomes in pediatric surgical patients: does preoperative nutritional assessment impact clinical outcomes? J Pediatr Surg 2014;49:823-30.


17. Leite HP, Fisberg M, de Carvalho WB, de Camargo Carvalho AC. Serum albumin and clinical outcome in pediatric cardiac surgery. Nutrition 2005;21:553-8.


18. Fowler AJ, Ahmad T, Phull MK, Allard S, Gillies MA, Pearse RM. Meta-analysis of the association between preoperative anaemia and mortality after surgery. Br J Surg 2015;102:1314-24.


19. Mahmoud AO, Zayed KM, Shawky NA. Stunting among children attending a Pediatrics Outpatient Clinic in Cairo, Egypt. Egyp J Community Med 2017;35:33-42.

20. Neumann CG, Harrison GG. Onset and evolution of stunting in infants and children. Examples from the Human Nutrition Collaborative Research Support Program. Kenya and Egypt studies. Eur J Clin Nutr 1994;48 Suppl 1:S90-102.

21. Canada NL, Mullins L, Pearo B, Spoede E. Optimizing perioperative nutrition in pediatric populations. Nutr Clin Pract 2016;31:49-58.


22. Lambert E, Carey S. Practice guideline recommendations on perioperative fasting: a systematic review. J Parenter Enter Nutr 2016;40:1158-65.
23. GlobalSurg Collaborative. Determining the worldwide epidemiology of surgical site infections after gastrointestinal resection surgery: protocol for a multicentre, international, prospective cohort study (GlobalSurg 2). BMJ Open 2017;7:e012150.



24. Schiesser M, Müller S, Kirchhoff P, Breitenstein S, Schäfer M, Clavien PA. Assessment of a novel screening score for nutritional risk in predicting complications in gastro-intestinal surgery. Clin Nutr 2008;27:565-70.


25. Toole BJ, Toole LE, Kyle UG, Cabrera AG, Orellana RA, Coss-Bu JA. Perioperative nutritional support and malnutrition in infants and children with congenital heart disease. Congenit Heart Dis 2014;9:15-25.


26. Leite HP, Fisberg M, Novo NF, Nogueira EB, Ueda IK. Nutritional assessment and surgical risk markers in children submitted to cardiac surgery. Sao Paulo Med J 1995;113:706-14.


27. Brugnara C, Oski FA, Nathan DG. Diagnostic approach to the anemic patient. In: Orkin SH, Nathan DG, Ginsburg D, Look AT, Fisher DE, editors. Nathan and Oski's hematology and oncology of infancy and childhood. 8th ed. Philadelphia (PA): WB Saunders, 2015:293.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


