Introduction
Rhinovirus (RV) is a common cause of asthma exacerbation. Viral infections trigger 80% of asthma exacerbation in children, with RV being the most common viral cause1). However, the mechanisms for RV-induced exacerbation of asthma are still uncertain. According to current theory, it is thought that infection of airway epithelial cells trigger epithelial responses that contribute to increased airway inflammation2), generating a variety of proinflammatory mediators to attract inflammatory cells to the airway with a subsequent worsening of underlying disease3). As part of efforts to clarify this theory, many trials have investigated mediators related to the lower airways immune responses in RV infected asthmatics. Proper comparison groups must be considered in these studies to show that the observed immune responses in an RV infected asthma group are related to exacerbation mechanisms. The results of noninfected asthma and infected normal control groups must be different from those of the RV infected asthma group. Induced sputum has been widely sampled for these studies, which have shown that this sample can provide information regarding the cellular and molecular process involved in asthma4). However, induced sputum may not reflect the mucosal inflammation occurring in asthma and may not show a correlation with bronchoalveolar lavage fluid (BALF) for many inflammatory cell types5). As far as we know, there have been relatively few reports using BALF as lower airway samples to analyze inflammatory mediators for the following groups: RV infected asthmatics with exacerbation; noninfected asthmatics; RV infected normal controls; and noninfected normal controls6-8). Therefore, we analyzed the inflammatory immune responses in BALF and in tissues from Dermatophagoides farinae (Df) sensitized and challenged mice (Df mice) and phosphate buffered saline (PBS) treated mice (PBS mice) with or without RV infection and investigated the different responses after RV infection in asthma by comparing the 4 groups above.
Materials and methods
1. Preparation of a murine asthma model
Six-week-old female BALB/c mice weighing 20-22 g (Orient Bio Inc., Seongnam, Korea) were injected intraperitoneally with 200 µL of a 2 mg/mL solution of aluminum potassium sulfate and 250 µg crude extract of Df (Arthropods of Medical Importance Resource Bank, Seoul, Korea) or PBS on day 0 and 14. Mice were given 50 µg crude extract of Df intranasally in a volume of 50 µL PBS on days 14, 15, and 16. We examined airway responsiveness, Th2 cytokine expression levels, and numbers of eosinophils in BALF. We also performed lung histopathological analysis of the lungs of the mice. This study was approved by the Institutional Animal Care and Use Committee.
2. RV exposure
RV 1B stocks were generated by infecting monolayer cultures of HeLa cells (KCLB No. 10002, Korean Cell Line Bank, Seoul, Korea) until cytopathic effects were fully developed. Cells and supernatants were harvested. The cells were disrupted by freezing and thawing, and the cell debris was pelleted by low speed centrifugation. The resulting clarified supernatants were then frozen at -70℃. The cytopathic effect was determined by visual inspection and by assessment of the continuity of the monolayer after fixation. Fifty percent tissue culture infectivity doses (TCID50) were determined by the Spearman-Karber method. Immediately following the last Df or PBS treatment, mice were inoculated intranasally with 5×106 TCID50/µL RV1B on day 16 (Fig. 1). Airway responsiveness to metacholine (12.5 mg/mL, 25 mg/mL, 50 mg/mL, and 100 mg/mL) was measured and BAL was performed 24 hours after RV inoculation. Lung digests were obtained by mincing the tissue to verify viral existence by polymerase chain reaction.
3. Determination of airway responsiveness
Airway responsiveness was measured with an OCP3000 insrument (Allmedicus, Anyang, Korea) using 4 different concentrations of metacholine. An Ultra-Neb ulrasonography nebulizer (3650p; Pulmo-Aide LT Lompressor, Someret, PA, USA) was used to administer an 3 mL aerolized volumes of each concentration of metacholine over a period of 3 minutes. The enhanced pause (Penh) variable was measured over a period of 3 minutes.
4. Preparation of BALF
After measuring airway responsiveness, the main bronchus was ligated and BAL was performed on the entire lung by using a 22 gauge needle inserted into the trachea. PBS (1 mL) was injected, and BALF was harvested once and centrifuged at 2,000 rpm for 10 minutes at 4℃. Supernatants were stored at -70℃.
5. Analysis of cells in BALF
Cell precipitates were suspended in 0.4 mL PBS and a 10 µL volume mixed with 10 µL of trypan blue solution (Gibco, Grand Island, NY, USA). The total number of cells in each sample was counted using a hemocytometer. The concentration of cells was adjusted to 1×106 cells/mL. Slides were prepared using a cytocentrifuge (Cytospin 2; Shandon, Runcorn, UK) and stained with Diff-Quick (Sysmex Corporation, Kobe, Japan). The differential counts of macrophages, eosinophils, lymphocytes, and neutrophils (500 minimum) were counted at ×400 magnification.
6. Histological studies of lung
Once lavage was complete, the lungs were immediately removed, fixed in cold formalin, embedded in paraffin, and cut into 5 µm thick sections, which were stained with hematoxylin and eosin and examined under light microscopy. Pathological change was described by a board certified pathologist, using a system previously described and modified9). The eosinophil and neutrophil infiltration score was evaluated on a subjective scale of 0 to 3. A value of 0 was assigned when no infiltration was detectable, a value of 1 for mild infiltration, a value of 2 for moderate infiltration, and a value of 3 for marked infiltration. The degree of peribronchial and perivascular inflammation was evaluated on a subjective scale of 0 to 3. A value of 0 was assigned when no inflammation was detectable, a value of 1 for occasional cuffing with inflammatory cells, a value of 2 for most bronchi or vessels surrounded by a thin layer (1-5 cells thick) of inflammatory cells, and a value of 3 when most bronchi or vessels were surrounded by a thick layer (>5 cells thick) of inflammatory cells. The degree of edema was evaluated on a subjective scale of 0 to 3. A value of 0 was assigned when no edema was detectable, a value of 1 for mild edema, a value of 2 for moderate edema, and a value of 3 for marked edema. The epithelial impairment score was evaluated on a subjective scale of 0 to 3. A value of 0 was assigned for a normal state, 1 for focal cell loss, 2 for moderate cell loss, and 3 for diffuse cell loss and epithelial metaplasia. The total inflammation score was defined as the sum of all of these scores.
7. Measurements of cytokines and chemokines
The levels of the following cytokines were quantified in the supernatants of BALF by enzyme-linked immunosorbent assay (Bio-Plex Pro Mouse Cytokine assay kit, Bio-Rad Laboratories Inc., Hercules, CA, USA). 1) Th1, Th2 and regulatory cytokine: interferon gamma (IFN-γ), IL-4, IL-5, IL-13, IL-10; 2) proinflammatory cytokine: IL-6, IL-1β, tumor necrosis factor alpha (TNF-α); 3) chemokine: regulated on activation, normal T cells expressed and secreted (RANTES), macrophage inflammatory protein-1-alpha (MIP-1α), eotaxin, keratinocyte derived chemokine (KC); and 4) granulocyte-macrophage colony stimulating factor (GM-CSF). The 4 groups for comparison are as follows: PBS treated mice (PBS mice), RV infected PBS treated mice (RV infected PBS mice), Df sensitized and challenged mice (Df mice), and RV infected Df sensitized and challenged mice (RV infected Df mice).
Results
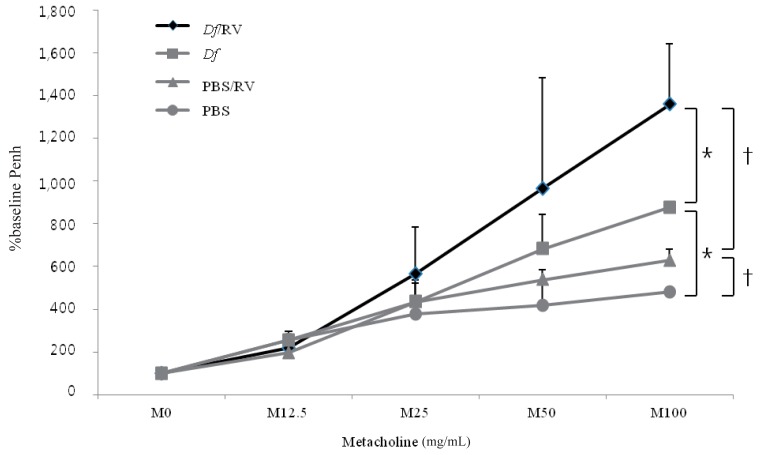
1. RV infection increases Penh in both Df mice and PBS mice
RV infection increased Penh in both Df mice and PBS mice (P<0.05). However, RV infected Df mice showed higher Penh levels at a metacholine dose of 100 mg/mL than RV infected PBS mice (P<0.05) (Fig. 2).
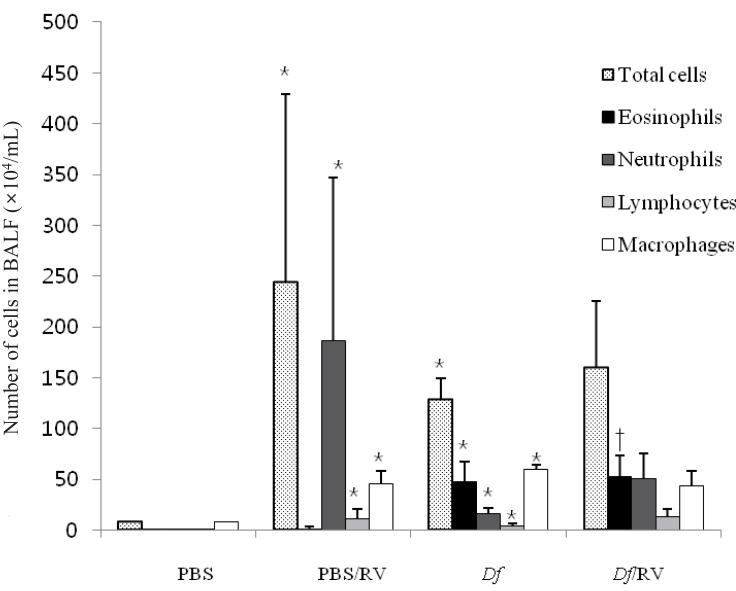
2. Neutrophils and eosinophils are dominant cells in BALF from Df mice after RV infection
The number of eosinophils, neutrophils, lymphocytes, and macrophages was higher in Df mice than in PBS mice (P<0.05). RV infection caused neutrophil to trend higher in Df mice (P=0.05) and increased neutrophil, lymphocyte, and macrophage count in PBS mice (P<0.05). The largest number of cells after RV infection was neutrophils (32.7%) and eosinophils (32.7%) in Df mice and neutrophils (76.5%) in PBS mice. RV infected Df mice showed a higher eosinophil count compared with RV infected PBS mice (P=0.046) (Fig. 3).
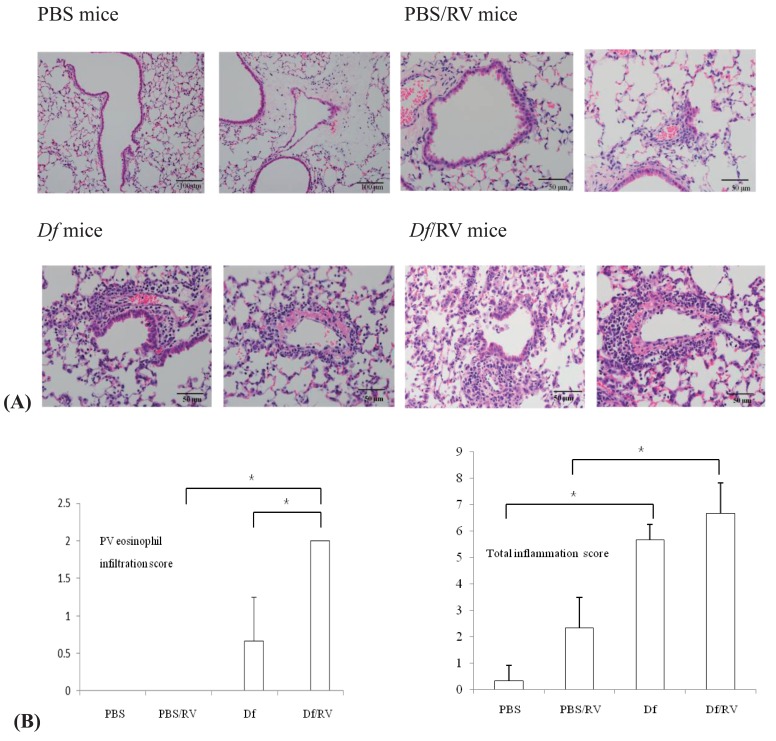
3. RV infection increases airway eosinophil infiltration and inflammation in Df mice
Peribronchial and perivascular inflammatory cell infiltration were observed in all 3 groups, but not in the PBS mice (Fig. 4A). Perivascular and peribronchial inflammatory cell infiltration and total inflammation score were elevated in Df mice, compared with PBS mice (P<0.05). There was no significant increase in total inflammation score in both Df mice and PBS mice after RV infection. However, RV infection significantly increased the perivascular eosinophil infiltration score in Df mice (P=0.034) but not in PBS mice (Fig. 4B).
4. Effects of RV infection on BALF cytokines and chemokines
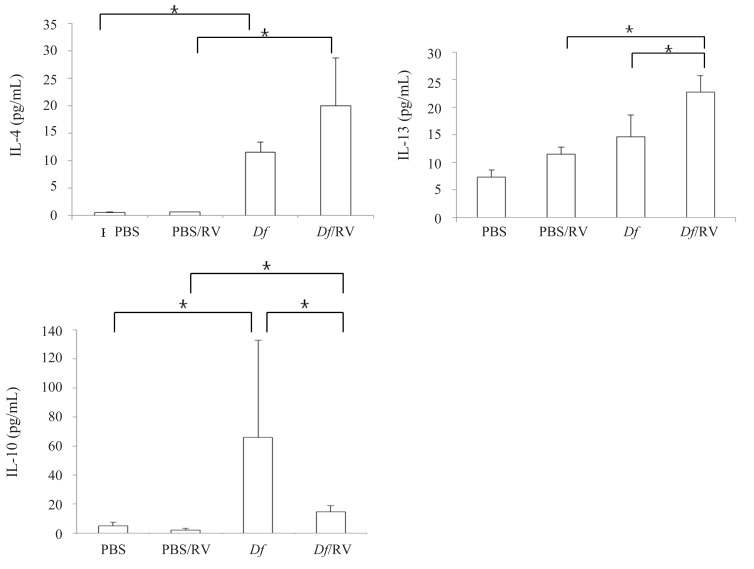
1) IFN-γ, IL-4, IL-13, and IL-10 : IL-13 increase and IL-10 decrease in Df mice after RV infection
There were no significant differences in IFN-γ levels between the 4 mice groups (data not shown). The levels of IL-4 was higher in Df mice than in PBS mice (P=0.046). The levels of IL-13 were higher, but not significatnly, in Df mice than in PBS mice (P=0.05). In Df mice, RV infection increased the levels of IL-13 (P=0.046) and caused the levels of IL-4 to trend higher (P=0.05). RV infected Df mice showed higher levels of IL-4 (P=0.009) and IL-13 (P=0.046) than RV infected PBS mice. The levels of IL-10 were higher in Df mice than in PBS mice (P=0.009). RV infection decreased the levels of IL-10 in Df mice (P=0.016). RV infected Df mice showed higher levels of IL-10 than RV infected PBS mice (P=0.008) (Fig. 5).
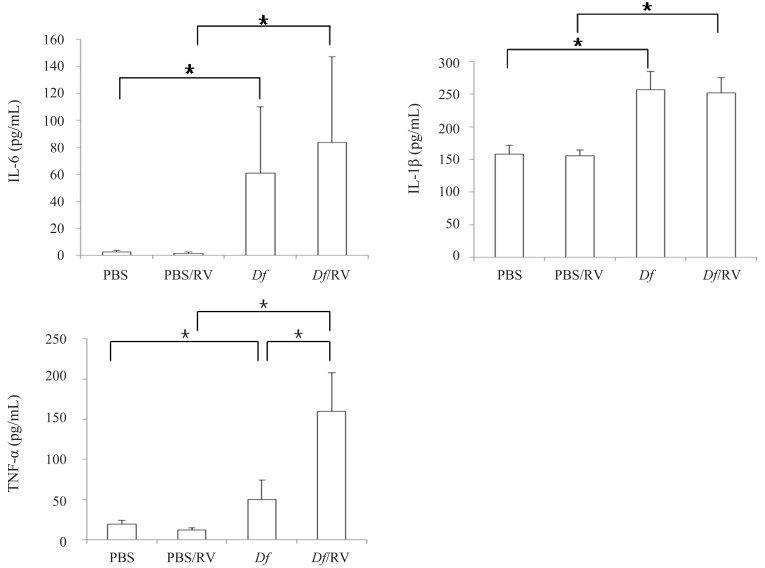
2) Proinflammatory cytokines (IL-6, IL-1β, and TNF-α): TNF-α increase in Df mice after RV infection
The levels of IL-6, IL-1β, and TNF-α were higher in Df mice than in PBS mice (P=0.009, 0.009, and 0.016, respectively). RV infection increased the levels of TNF-α in Df mice (P=0.009) but not in PBS mice. RV infected Df mice showed higher levels of IL-6, IL-1β, and TNF-α than those in RV infected PBS mice (all P=0.009) (Fig. 6).
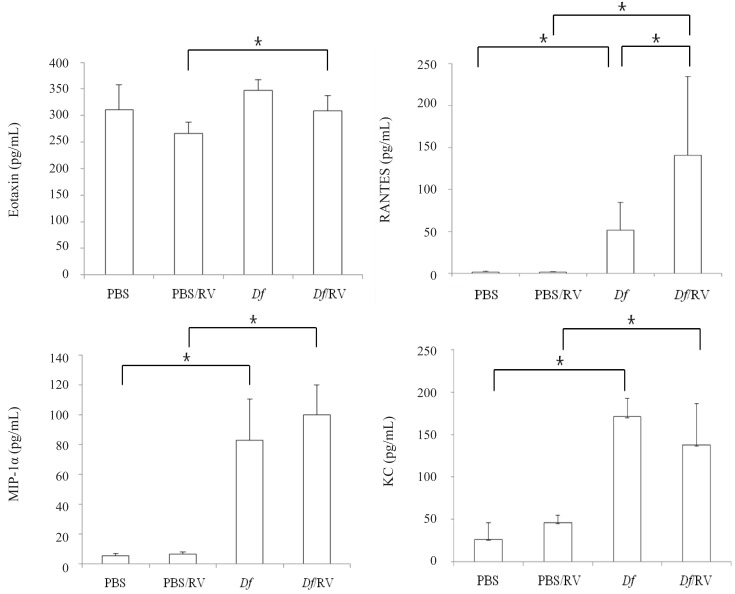
3) Chemokines (RANTES, MIP-1α, eotaxin, and KC): RANTES increase in Df mice after RV infection
The levels of RANTES, MIP-1α, and KC were higher in Df mice than in PBS mice (P=0.009 for all three chemokines). RV infection increased the levels of RANTES in Df mice (P=0.028) but not in PBS mice. RV infected Df mice showed higher levels of RANTES, MIP-1α, eotaxin, and KC than RV infected PBS mice (P=0.009, 0.009, 0.034, and 0.009, respectively) (Fig. 7).
Discussion
In this study, RV infection induced an increase of IL-13, TNF-α, and RANTES levels in BALF in Df mice, but not in PBS mice. These data are consistent with the notion that the allergic environment qualitatively alters the response to RV7). IL-13 induces mucus metaplasia, airway hyperresponsiveness (AHR), and eosinophilic inflammation, as well as Th2 cytokine and eotaxin production. Further, an IL-13 gene deletion protects asthma mice models from allergic airway inflammation10), suggesting an important role in exacerbating allergic airway disease. RANTES is an important factor in the activation of eosinophils in RV induced asthma exacerbations3). RV infection of bronchial epithelium results in induced eosinophil recruitment, through the production of chemokines such as RANTES, MIP-1α, and eotaxin11). The increase in airway responsiveness in asthma patients correlates with the increase in eosinophil cationic protein and with the change in the eosinophil percentage in sputum, suggesting that RV enhanced AHR is associated with eosinophilic inflammation12). Our results did not show a direct link between these cytokine/chemokine and AHR or airway eosinophil infiltration. However, significantly higher level of IL-13 and RANTES in RV infected Df mice was accompanied with greater airway eosinophil infiltration and higher Penh values, compared with Df mice, which is in agreement with the findings of previous studies10,12,13). This suggests that IL-13 and RANTES has a possible role in an RV induced increase of airway eosinophil infiltration and Penh level in Df mice.
TNF-α, one of the increased cytokines in asthmatic airways, is produced by macrophages, T cells, mast cells, and epithelial cells. Its secretion is stimulated by airway macrophages after RV infection3,14). Besides intercellular adhesion molecule 1 (ICAM-1) related roles, TNF-α also functions as a chemoattractant for neutrophils and stimulates the production of IL-8 and RANTES by airway epithelial cells15). There have been a few reports on interactions between RV infection and TNF-α14,16,17). RV infection and TNF-α stimulation induced a cooperative increase in airway epithelial cell chemokine expression, which is not fully accounted for by an increase in ICAM-1 expression, suggesting its role in a cellular mechanism for RV-induced exacerbations of asthma14). Moreover, in an experimental RV infected cell study, TNF-α was able to down-regulate the antiviral action of INF-α completely, even at very low levels. Furthermore RV replication in lung macrophages induces TNF-α production and secretion16). However, these previous findings came from cell experiments, and interactions between RV infection and TNF-α from in vivo asthma models is still not well understood. Based on our findings, we cannot determine whether airway cell infiltration or an increased level of Penh in RV infected Df mice was directly affected by an elevated TNF-α level. Nevertheless, it is notable that the TNF-α level increased in Df mice, but not in PBS mice in response to RV infection using an in vivo experiment. A recent report showed a TNF-α dependent response early in the second phase of AHR in the murine asthma model18). Although it is not clear whether this biphasic AHR is a universal phenomenon, the significant relationship of TNF-α and AHR cannot be ignored. Taken altogether, TNF-α may be of importance in RV-induced asthma exacerbation mechanisms through the regulation of RV infection.
In asthma, while these above mentioned mediators are known to play an important role in airway inflammation, the function of IL-10, an anti-inflammatory or regulatory cytokine, is not still clear. IL-10 can be produced by most cells of the immune system, including dendritic cells, B cells, macrophages, CD4 T cells, CD8 T cells, NK cells, and regulatory T cells19). The level of IL-10 in the present study decreased after RV infection in Df mice, but not in PBS mice. The reports of the effects of RV infection on IL-10 in asthma models differ between studies6,7). A deficient induction of IL-10 from BAL cells of asthmatics compared with normal subjects was reported6), while RV treatment of macrophages from ovalbumin (OVA) sensitized mice, but not from PBS mice, induced significant expression of IL-107). Apart from the fact that previous studies have used different cells or subjects from this study, the differing results can also be partially explained by the various functions of IL-10. That is, it is possible that in allergen sensitized virus infected mice, there is more complicated pattern of IL-10 secretion, given that it is an M2 polarization marker and has diverse functions. Although generally immunosuppressive, many of its functions may be pathogen specific19).
There was disagreement among some of our results. Firstly, in the present study, eotaxin in RV infected Df mice did not show higher levels compared to Df mice in spite of increase in IL-13. A recent study suggested the importance of alveolar macrophages as a source of increased production of eotaxin in allergen sensitized and RV infected mice. It also showed increase in BALF macrophages in allergen treated mice after RV infection7). Although our experiment did not measure eotaxin from isolated macrophages, the fact that there was no significant increase in BALF macrophage in RV infected Df mice might have contributed to the discordance between patterns of change in IL-13 and eotaxin concentration. Secondly, unlike tissue eosinophil count, in the BALF, RV infection did not increase eosinophils in Df mice; instead it caused a trend toward an increase in the neutrophils. We analyzed BALF 1 day after infection, which is the time over which that neutrophil count increases8). Indeed, a previous study reported a significant increase in BALF eosinophils in Df mice 4 days after RV infection7). Furthermore, in the lungs of our murine asthma model, the largest absolute number of cells was neutrophils and eosinophils. Taken together, this suggests that in RV infected allergen sensitized mice, an increase of neutrophils was the first response, followed by an increase in eosinophils7,8). In the present study, neutrophil infiltration was significantly higher in Df mice than that in PBS mice. This is conceivable, considering the fact that a significant proportion of asthma is associated with neutrophilic airway inflammation, and that this pattern is not limited to individuals with severe symptoms20).
Finally, we must acknowledge the limitations of our experiment. We could not conduct further study on the above mentioned cytokines of significance. For example, to determine the direct link between a specific cytokine and airway responsiveness or tissue findings, a deletion study or antibody blocking trial for the cytokine of interest will be needed. Thus, on the basis of the experiments reported here, it is not possible to determine whether these cytokines are essential for asthma exacerbations in our mouse model. Nevertheless, it is clear that some lower airway cytokines were enhanced after RV infection in allergen sensitized mice only and not in control mice. Moreover, there are differences between this report and previous ones6-8), which investigated inflammatory mediators in BALF from 4 groups like our study. The results in the present study were obtained, not by stimulating BAL cells with phytohemagglutinin (PHA)6), but by infecting murine models with RV1B. Unlike previous studies14,16,17), we also observed the increase in TNF-α levels in an in vivo asthma model as one of different responses in post RV infection compared to a control model.
In conclusion, we have shown that RV infection upregulates IL-13, TNF-α, and RANTES from BALF in Df mice, but not in control animals. The level of IL-10 was decreased in Df mice. These findings are accompanied by increased levels of Penh with airway eosinophil infiltration. These data suggest that modulation of secretion of these cytokines and chemokines may be one of important differences in the lower airway reaction to RV infection between allergen-treated mice and control mice, leading to eosinophilic airway inflammation and increased airway responsiveness in allergen-treated mice. Further studies on the role of IL-13, TNF-α, RANTES, and IL-10 in RV induced asthma exacerbation will contribute to prevention and treatment of acute asthma attacks.




 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


