Article Contents
| Clin Exp Pediatr > Volume 66(5); 2023 |
|
Infantile-onset multisystem neurologic, endocrine, and pancreatic disease (IMNEPD) is a rare inherited recessive disorder that is caused by biallelic mutation in PTRH2 gene. The first cases were described in 2014 by Hu et al. [1] who exposed the novel disease phenotype, genetic cause, and the functional analysis in human and mice tissues. The main features of IMNEPD are intellectual disability, global developmental delay, deafness and exocrine and endocrine pancreatic insufficiency. The phenotypic variability was reported in individuals carrying the same mutation [2]. Clinical variability was observed and the frequency of neurological abnormalities was found highest. Diverse types of mutations were discovered having the missense mutation the commonest [3]. Studies conducted in the Middle Eastern and Asia region reported varying genetic mutations [1,4-6]. We presented 2 siblings diagnosed with IMNEPD with phenotype and genotype description.
A 17-year-old female born to consanguineous parents (Table 1). Parents noticed global developmental delay at 1 year of age. Patient had difficulties in gross motor skills, walks in a slow gait and severe pes cavus deformity (Fig. 1A). Patient was diagnosed recently with mild scoliosis. In addition to speech delay and learning difficulties, she was hypotonic with muscular atrophy and depressed deep tendon reflexes (Fig. 1B). Electroencephalogram (EEG) at 1 year of age showed mild to moderate generalized nonspecific disturbance of cerebral activity. She received antiepileptic medications at 1 year for couple of years. On follow-up, her repeated EEGs at 2,11 and 13 years of age were unremarkable. Brain magnetic resonance imaging (MRI) recently reported cerebellar atrophy when compared to normal previous MRI (Fig. 1E, F). At the age of 12 years, patient was diagnosed with insulin-dependent diabetes mellitus when hemoglobin A1c (HbA1c) was 15.3% but was not in ketoacidosis. She was referred to genetic clinic and performed whole exom sequencing after written consent. It showed a novel homozygous variant in PTRH2 gene classified as likely pathogenic according to American College of Medical Genetics classification (Supplementary 1).
An 18-year-old male who is brother of 1st case (Table 1). The patient followed almost the same clinical course as his sister. Parents noticed global developmental delay in the 1st year of life. Currently, he has difficulties in gross motor skills, walks in a slow gait and has severe pes cavus deformity (Fig. 1C). In addition to speech delay and learning difficulties, he is hypotonic with muscular atrophy with depressed deep tendon reflexes (Fig. 1D). EEG showed mild generalized disturbance of cerebral activity with infrequent generalized epileptic discharges. Antiepileptic medications were started for couple of years. His repeated EEGs at 4, 12, and 15 years of age were unremarkable. Targeted genetic testing confirmed the diagnosis (Supplementary 1).
PTRH2 gene produce a mitochondrial protein which has a functional role in human cognitive function and the regulation of cell growth and survival. It involves in regulation of apoptotic signaling and myogenic differentiation [7]. This explains the progressive nature of the disease. Hu et al. [1] reported an affected girl who became dependent on wheel-chair at age of 15 years due to muscle weakness and ataxia. Our case 1 showed progressive difficulty in walking due to demyelinating neuropathy and scoliosis but she is still ambulatory. She showed progression in the hearing impairment. Another study in Syrian patient (age 17 year) showed IMNEPD due to a novel nonsense mutation in PTRH2 gene (c.324G>A (p.W108*) resulted in limb and truncal ataxia [5]. Supplementary 2 showed the clinical variability of IMNEPD and demonstrated neurological manifestation predominate as PTRH2 was highly expressed in brain tissue. In our patients, we discovered homozygous variant in PTRH2 gene which is the second family to have duplication type. This adds to the postulation that protein-truncating variants is more severe [3,5,6].
MRI brain for our first case showed progressive cerebellar atrophy and this has been frequently reported in patients with IMNEPD [1,2,8]. Parida et al. [3] estimated the presence of cerebellar atrophy in 40%. The lack of cerebellar atrophy in some patients with missense mutation is mainly related to residual PTRH2 [2]. In absence of PTRH2, there will be downregulation of the mammalian target of rapamycin (mTOR) signaling and lead to loss of Purkinje cell in cerebellum and progressive cerebellar atrophy [8]. It has been reported that mTOR1 agonists can rescue Purkinje cell and improve mTOR signaling [9]. This raises the hope for a potential therapy.
Our case was diabetic and her affected sibling in prediabetic state. The youngest age when diabetes was discovered was 12 years of age [3]. Another study reported development of antibody negative diabetes (HbA1c value 10.2%) with neuro cognitive impairment and deafness at the age of 13 year emphasized on regular monitoring of diabetes [10].
We herein recommend regular check of glycosylated hemoglobin after the 1st decade of life and encourage to do a neuroimaging at diagnosis and follow-up especially if the patient has neurological manifestation.
Supplementary material
Supplementary 1.
Genetic testing results identifying a likely pathogenic variant in the PTRH2 gene (NM_001015509.2: c.114dup, p. Gly39Trpfs*16) according to American College of Medical Genetics classification.
Supplementary 2.
Clinical presentation of current patients versus previously published cases (Parida et al. [3])
Fig. 1.
Cases 1 (A) and 2 (C). A neurological evaluation showed a severe pes cavus deformity of the foot. Cases 1 (B) and 2 (D) showing muscular atrophy with depressed deep tendon reflexes. (E, F) T2 axial brain magnetic resonance image of case 1 showing prominent bilateral cerebellar folia with mild to moderate cerebellar atrophy.
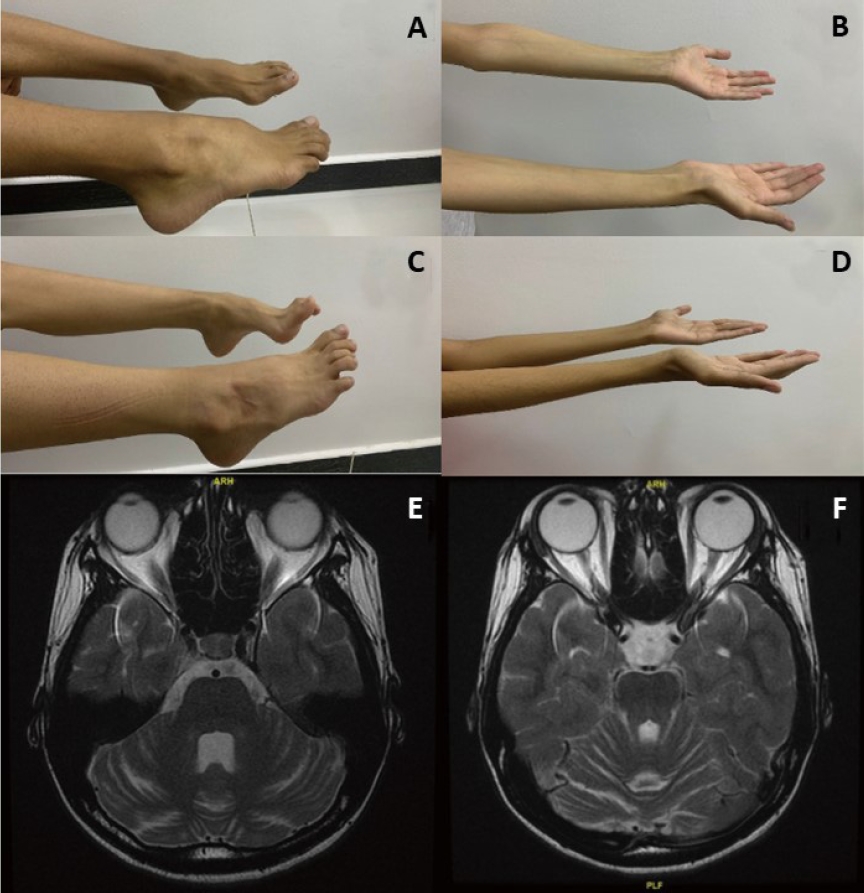
Table 1.
Clinical characteristics of patients with infantile-onset multisystem neurologic, endocrine, and pancreatic disease
| Characteristics | Case 1 | Case 2 |
|---|---|---|
| Age | 17 | 18 |
| Sex | Female | Male |
| Nationality | Saudi | Saudi |
| Birth | At 28 weeks | Full term |
| Genetic mutation | NM_001015509.2: c.114dup, p. Gly39Trpfs*16 (homozygous) | NM_001015509.2: c.114dup, p. Gly39Trpfs*16 (homozygous) |
| Failure to thrive | Present | Absent |
| Onset of global developmental delay | Infancy | Infancy |
| Brain MRI | Cerebellar atrophy | Arachnoid cysta) |
| Progressive bilateral sensorineural hearing loss | Present | Present |
| Nerve conduction studies | Mixed sensorimotor neuropathy mostly axonal at 14 years of age | NA |
| IQ testingb) | 51 Moderate impairment | 50 Moderate impairment |
| Vineland Adaptive Behavior Scale | 20–35 in communication (severe) | 20–35 in communication (severe) |
| 35–40 in adaptation (moderate) | 20–35 in adaptation (severe) | |
| Diabetes mellitus | Diabetic | Prediabetes state |
References
1. Hu H, Matter ML, Issa-Jahns L, Jijiwa M, Kraemer N, Musante L, et al. Mutations in PTRH2 cause novel infantile-onset multisystem disease with intellectual disability, microcephaly, progressive ataxia, and muscle weakness. Ann Clin Transl Neurol 2014;1:1024–35.




2. Bronson SC, Suresh E, Kumar SS, Mythili C, Shanmugam A. A novel synergistic association of variants in PTRH2 and KIF1A relates to a syndrome of hereditary axonopathy, outer hair cell dysfunction, intellectual disability, pancreatic lipomatosis, diabetes, cerebellar atrophy, and vertebral artery hypoplasia. Cureus 2021;13:e13174.


3. Parida P, Dubbudu A, Biswal SR, Sharawat IK, Panda PK. Diabetes mellitus in an adolescent girl with intellectual disability caused by novel single base pair duplication in the PTRH2 gene: expanding the clinical spectrum of IMNEPD. Brain Dev 2021;43:314–9.


4. Khamirani HJ, Zoghi S, Dianatpour M, Jankhah A, Tabei SS, Mohammadi S, et al. A novel PTRH2 missense mutation causing IMNEPD: a case report. Hum Genome Var 2021;8:23.




5. Le C, Prasad AN, Rupar CA, Debicki D, Andrade A, Prasad C. Infantileonset multisystem neurologic, endocrine, and pancreatic disease: case and review. Can J Neurol Sci J Can Sci Neurol 2019;46:459–63.

6. Sharkia R, Shalev SA, Zalan A, Marom-David M, Watemberg N, Urquhart JE, et al. Homozygous mutation in PTRH2 gene causes progressive sensorineural deafness and peripheral neuropathy. Am J Med Genet A 2017;173:1051–5.



7. Doe J, Kaindl AM, Jijiwa M, de la Vega M, Hu H, Griffiths GS, et al. PTRH2 gene mutation causes progressive congenital skeletal muscle pathology. Hum Mol Genet 2017;26:1458–64.



8. Corpuz AD, Ramos JW, Matter ML. PTRH2: an adhesion regulated molecular switch at the nexus of life, death, and differentiation. Cell Death Discov 2020;6:124.




9. Picker-Minh S, Luperi I, Ravindran E, Kraemer N, Zaqout S, Stoltenburg-Didinger G, et al. PTRH2 is necessary for purkinje cell differentiation and survival and its loss recapitulates progressive cerebellar atrophy and ataxia seen in IMNEPD patients. Cerebellum 2022 Oct 11. doi: 10.1007/s12311-022-01488-z. [Epub].


10. Becker M, Seneca S, Schierloh U, Witsch M, De Beaufort C, Scalais E. Diabetes in a child with infantile-onset multisystem neurologic, endocrine, and pancreatic disease (IMNEPD). In: 59th Annual Meeting of the European Society for Paediatric Endocrinology (ESPE 2021 Online); 2021 Sep 22-26; Abstracts 2021;94.





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


