Article Contents
| Clin Exp Pediatr > Volume 64(5); 2021 |
|
Abstract
Acute fulminant myocarditis (AFM) occurs as an inflammatory response to an initial myocardial insult. Its rapid and deadly progression calls for prompt diagnosis with aggressive treatment measures. The demonstration of its excellent recovery potential has led to increasing use of mechanical circulatory support, especially extracorporeal membrane oxygenation (ECMO). Arrhythmias, organ failure, elevated cardiac biomarkers, and decreased ventricular function at presentation predict requirement for ECMO. In these patients, ECMO should be considered earlier as the clinical course of AFM can be unpredictable and can lead to rapid haemodynamic collapse. Key uncertainties that clinicians face when managing children with AFM such as timing of initiation of ECMO and left ventricular decompression need further investigation.
Myocarditis is an inflammatory disease caused by an initial insult to the heart which is followed by lymphocytic inflammation of the myocardium [1]. The clinical course of myocarditis ranges from a mild to severe form of heart failure. The severe form of myocarditis that necessitates aggressive and timely treatment to prevent rapid haemodynamic collapse is termed acute fulminant myocarditis (AFM). In certain circumstances, the only way to prevent imminent death is to support the patient on mechanical circulatory support (MCS). Extracorporeal membrane oxygenation (ECMO) is one form of MCS.
ECMO, in the veno-arterial (VA) configuration, is increasingly being used in both pediatric and adult populations for refractory cardiogenic shock [2]. VA-ECMO (simplified as ECMO in this manuscript from here on) represents a modality where blood is drained from a large vein, oxygenated and returned to the circulation through a large artery (Fig. 1).
Given ECMO is only life-sustaining in these rapidly deteriorating patients, a thorough understanding of when and how it can be employed is prudent. This comprehensive review has a detailed account of the characteristics of AFM, the clinical scenarios where ECMO might be employed, basic management strategies of a patient on ECMO, further management options and outcomes.
AFM accounts for 10% to 38% of all cases of acute myocarditis [3-5]. Importantly, AFM may account for approximately 10%–20% of sudden and unexplained deaths in children [6-13]. In a United States study, there was a bimodal age distribution with peaks in infancy and mid-teenage years, with a mean age of 9 years [14]. Interestingly, about 60% of patients with AFM are male, a phenomenon observed perhaps due to pubertal hormonal changes in boys [15]. However, females seem to have worse ventricular function at admission, higher mortality, higher need for cardiac transplants and readmissions to hospital within the first year [16]. In a Japanese survey, only 12% had remarkable comorbidities or past medical history, of which most prevalent was congenital heart disease [13].
The aetiology of myocarditis is diverse, as it can be caused by infectious, autoimmune, or toxic agents. In developed countries, the predominant cause of pediatric acute myocarditis is viral infections [17].
The pathophysiological sequence of myocyte injury in acute myocarditis occurs in 2 distinct phases [18]. During the initial stage, a pathogen causes cell injury, lysis and myocardial necrosis, leading to ventricular dysfunction, heart failure, and arrhythmias. This triggers an inflammatory reaction resulting in lymphocyte infiltration.
Intravenous immunoglobulin (IVIG) has been used to limit this immune response, though there are no convincing data as yet demonstrating a clear survival benefit [19,20]. Even if IVIG were beneficial, the immune-modulatory effects are not immediate. Despite limited and inconclusive evidence, IVIG is still considered a potential therapeutic strategy. Proponents observe that the associated risks are relatively small when compared to the morbidity and mortality from AFM [16,17]. Similarly, steroids have been employed with not much reported benefit [21].
Children with AFM are some of the sickest patients. In these patients, hemodynamic compromise can be sudden. Consequently, early diagnosis and prompt management with aggressive therapies can be lifesaving.
The aims of therapeutic interventions in a patient with AFM is to augment cardiac output, to stabilize hemodynamic variables, and to optimise oxygen delivery-demand mismatch. Positive-pressure ventilation assists by reducing the work of breathing and left ventricular afterload. Although intubation and sedation decrease metabolic demands, it must be done with caution, since these patients are at high risk of severe hemodynamic deterioration and cardiac arrest during intubation [22]. Cautious fluid resuscitation and intravenous inotropic agents are the mainstay of treatment [17]. Some of the predictors of a fulminant myocarditis course is outlined in Table 1.
When a patient is not adequately perfused on maximal medical therapy, ECMO offers a sensible escalation strategy. In these patients, ECMO might be employed as a bridge to decision, bridge to recovery (BTR), or bridge to transplant (BTT). A summary of the main indications for ECMO is presented in Table 2. Retrospective studies that have investigated the use of ECMO in pediatric AFM in the last decade are also reviewed in Table 3.
ECMO is not a definitive treatment for myocarditis as it does not reverse native cardiac dysfunction. But, ECMO can support whole-body perfusion while cardiac function recovers. Although, the time-point when a patient is placed on ECMO varies between centres, there is general consensus that ECMO must be instituted before end-organ dysfunction ensues. Given the decision to place on ECMO and the timing are vitally important for the survival and long-term outcome of the patients, proactive multidisciplinary discussions about goals and strategies should take place in the early phases of illness.
Peripheral cannulation is the preferred approach for children with AFM. The variables to consider in the choice of location are the size of the ECMO cannulae relative to the vessel size and the condition of vessels (i.e., previously accessed/thrombosed). Femoral vessels are not well developed before children start walking. Hence if chosen as a site, there is an increased the risk of limb ischemia and gangrene while access itself may be harder. Crucially, the desired blood flows may not be achieved [32,33]. Consequently, contrary to adult ECMO, the cannulation for neonates, infants, and small children is primarily in the neck (drained from right internal jugular vein and perfused to right common carotid artery) or central cannulation (drained from right atrium and perfused to ascending aorta) via median sternotomy. There are disadvantages with both these approaches. While neck cannulation has a greater risk of neurologic injury, central cannulation has greater risks of mediastinitis and bleeding [34].
The cannulation strategy, therefore, is made as a balance of the risks/benefits of the chosen vessel and the desired blood flow. As a rule of thumb, the desired blood flow is that which perfuses all end organs adequately. Although most units might start at about 120–150 mL/kg/min in a neonate (cardiac index 2.2–2.4 L/m2/min in an older child), this will then be titrated up or down according to the requirements of the child. Ideally, markers of end-organ perfusion such as arterial lactate, mixed venous saturations, preoxygenator arterial oxygen saturations, urine output, biochemical variables of liver and renal function, and mean arterial blood pressure (5th centile for the age) are monitored closely to optimise ECMO support [34].
Left heart decompression (LHD) facilitates myocardial recovery in ECMO patients. ECMO increases the afterload of the left heart and this can lead to left atrial and left ventricular (LV) dilation resulting in pulmonary oedema and thrombus formation within the cardiac chambers [35]. Additionally, the volume overload increases myocardial wall stress and induces inflammatory reactions. Consequently, in inflammatory diseases such as AFM, the combination of LV overloading and inflammation may lead to unfavourable cardiac remodelling. By unloading the LV, wall stress and inflammatory responses are reduced [36].
As LHD is needed in 13% to 46% of pediatric VA-ECMO cases [37-39], certain warning signs might enable decision making. In a normally septated heart with no native ejection (a continuously flat arterial line waveform), ECG trace of ventricular tachycardia, ventricular fibrillation, pulseless electric activity, agonal heart rhythm, or asystole could be critical signals. A persistently closed aortic valve, LV dilatation, sludge, or thrombus formation in LV revealed by echocardiography are additional pointers [39].
LHD can be achieved by percutaneous techniques or surgery. It can be performed by atrial septostomy or by placing a venting catheter in the left atrium or left ventricle. Although LHD demonstrated no survival benefits in some studies [38,40], a recent meta-analysis of adult cardiogenic shock reported an association with decreased mortality. LHD was also associated with a higher rate of LV recovery, relief of pulmonary oedema, weaning off, and duration of ECMO [38,41,42]. Studies that investigated LHD are summarised in Table 3.
ECMO creates interactions between blood and the biomaterial interface, which results in activation of inflammatory and coagulation cascades [43,44]. Thus, anticoagulation becomes mandatory. Optimal anticoagulation is required to limit thrombotic and bleeding complications. Achieving this ‘optimal’ goal has been challenging.
The optimal management of the coagulation system in children is unclear [45-47]. Developmental haemostasis precludes extrapolation of adult anticoagulation strategies. Unfractionated Heparin (UFH) remains the standard of care in pediatric ECMO. However, reports of age-dependent variation in UFH activity with paucity of pediatric research in this area yield varying practices between institutions [48-50]. Research to find a better alternative is underway. Direct thrombin inhibitors (e.g., bivaluridin) have shown promise in early studies [51,52]. Although there are no standard methods for monitoring the effects of anticoagulants in neonatal and pediatric patients on ECMO, the 3 common assays are commonly used which are activated clotting time, activated partial thromboplastin time, and Anti-Xa [53-55]. Table 3 summarises studies that investigate anticoagulation practices in paediatric ECMO.
Thromboembolic and hemorrhagic complications are a frequent cause of death in children supported on ECMO. According to the ELSO registry, the most common mechanical complications are clots in circuits, complicating 22% of neonatal and 17% of pediatric cardiac ECMO runs [2]. Unfortunately, neurologic complications (secondary to bleeding or emboli) are more frequently observed in children than adults [56]. Of note, intracranial haemorrhage, cerebral infarction, and seizures are all seen twice as often in children compared to adults [57]. In addition, regardless of age, neck cannulation increases the risk of neurologic injury in children. The derangement of cerebral autoregulation, the decrease in cerebral venous drainage, and an increased risk of thromboembolic events are possible hazards of this approach [58]. Due to these potentially significant complications, patients on ECMO are closely monitored with regular anticoagulation assays. In the near future, routine neuromonitoring strategy inclusive of techniques such as near infrared spectroscopy and electro-encephalograms might become standard of care [59]. Table 4 summarises studies that explored bleeding, thrombosis, and neurologic complications during pediatric ECMO.
When cardiac function has recovered adequately, the patient is assessed for separation from ECMO support. The main variables that dictate successful weaning are echocardiographic evidence of myocardial recovery, with a good lung function (adequate gas exchange on less than moderate ventilatory support), resolution of end-organ dysfunction, and optimal pulse pressure/end tidal CO2 [34,62]. Echocardiographic evaluation should ideally be made under low-flow conditions. The specific parameters shown to predict are aortic velocity time integral >10 cm, LV EF >20%–25%, and lateral mitral annulus peak systolic velocity ≥6 cm/sec [63].
In a small proportion of AFM patients on ECMO, cardiac function might not adequately recover within 2 weeks. In these instances, ventricular assist devices (VADs) or listing for a heart transplantation maybe considered [14,16,28,37,64,65]. While discussion with parents and preparation should start early, the diagnosis and decision need carful thought. Following prerequisites lend themselves a guide for the decision making around transition to VAD or heart transplantation [66,67]:
•Potential recovery of heart function has been quantified
•End-organ dysfunction has been shown to be reversible
•Lung function is adequate
•Right ventricle is expected to cope with no support (in case of left VAD placement)
•Neurological function has been quantified to best of ability
•Recovery of cardiac function is expected to take more a few weeks (in BTR strategy)
•Patient has been listed for heart transplant (BTT strategy) and age/weight/blood group considerations might delay potential for organ availability
Although survival rate in transplant patients is also favourable [14,28,37,64], the role of heart transplantation has been questioned, as it has become more apparent that native ventricular function is ultimately expected to return, and prolonged duration of extracorporeal support can salvage these patients to recovery [64,68].
Even though AFM often presents as cardiogenic shock or cardiac arrest, the long-term survival is favourable [69]. Similarly, patients with AFM on ECMO have relatively good survival rates ranging from 58% in neonates to 63%–81% in pediatric patients [2,30,58,70]. The parameters associated with mortality are female gender, arrhythmia during ECMO support, renal failure requiring dialysis, and higher stage of end-organ damage [40,64,71].
Similar to neuromonitoring, the long-term neurological outcome in pediatric AFM patients supported on ECMO has not been reported. But some of this can be extrapolated from reports on children with other conditions who survived with ECMO. Alarmingly, lower intelligence quotient scores, behaviour problems, and severe neurodevelopmental delays have been reported in post ECMO pediatric survivors [69,72]. These findings warrant long-term follow-up not only to monitor recovery of cardiac function but also to delineate neurological functional deficits.
The decision to place a patient on ECMO is not easy. The clinician has to balance the risk and the benefit of such an invasive intervention. Clear guidelines cannot be stipulated for this scenario as each patient, each clinical setting, and each team bring in a myriad of nuances. With a haemodynamic profile similar to the AFM patient, data from patients in septic shock requiring ECMO could provide some insights. Schlapbach et al. [73] created a predicted mortality risk for septic controls. This was then compared to the observed mortality for those who were placed on ECMO. They derived a treatment benefit threshold of 47.1% predicted risk of mortality. Above this cutoff, patients were better off on ECMO, while below this value patients did well without ECMO. These results, if extrapolated to other groups, suggest that too early application of ECMO leads to higher mortality due to complications of the therapy, while too late application leads to higher mortality due progression of the underlying disease with inherent complications [73,74].
The early recognition of the clinical signs of a failing heart is paramount. Outcomes in children with AFM are favourable when ECMO is instituted before end-organ dysfunction ensues. Hence, early discussion and implementation of ECMO are mandatory to avoid potentially lifelong sequelae through distant organ injury. The clinician must be cognisant of the complications related to ECMO. This should guide decision making and parental counselling. Finally, a multidisciplinary team approach along with parents is needed to achieve better survival and quality of life. Key uncertainties that clinicians face when managing this patient cohort are the timing of initiation of ECMO and LV decompression. These need to be urgently investigated with strong research methodologies.
Acknowledgments
Hwa Jin Cho acknowledges the support of a grant NRF-2018R1D1A1A02085975 from National Research Foundation of Korea and a grant BCRI18028 from the Chonnam National University Hospital Biomedical Research Institute. Silver Heinsar received a PhD Scholarship from The Prince Charles Hospital Foundation.
Authors acknowledge Sarfaraz Rahiman and In Seok Jeong for reviewing the paper before the submission and Dr. Samia Farah, MD for the illustration, which was created with BioRender.com.
Fig. 1.
Treatment of acute fulminant myocarditis with veno-arterial extracorporeal membrane oxygenation (VA-ECMO) in children acute fulminant myocarditis manifests as lymphocytic infiltration in the myocardium. Rapid, severe disease course, and excellent long-term outcomes justify early use of aggressive management such as ECMO.
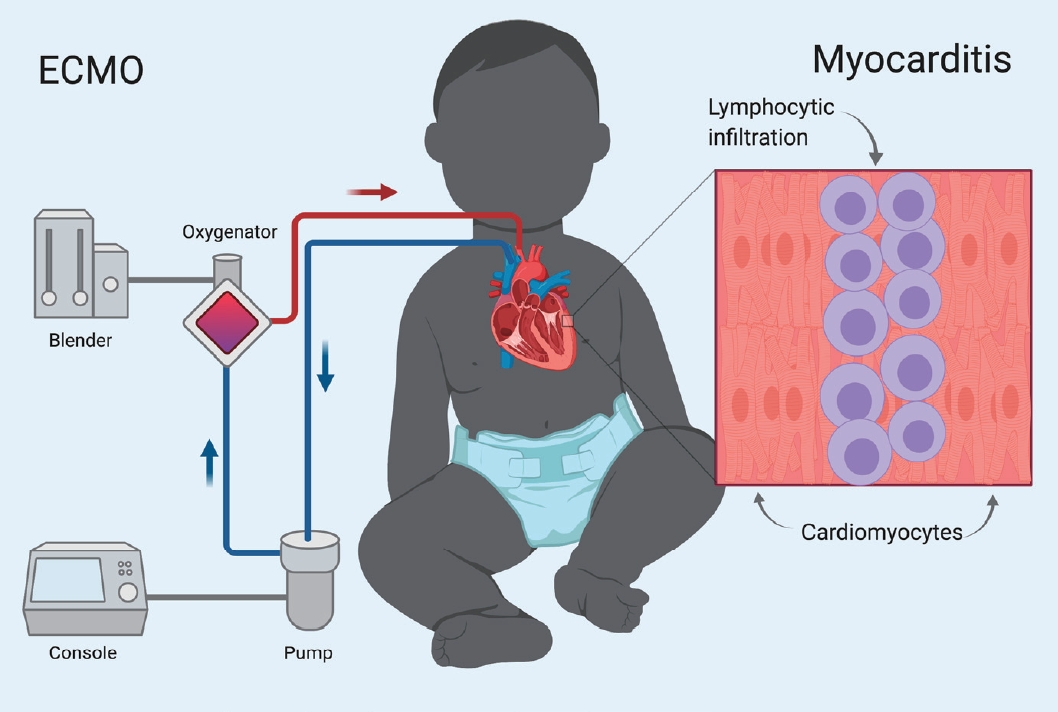
Table 1.
Predictors of a fulminant course
Table 2.
Table 3.
Overview of studies in ECMO use for children with acute fulminant myocarditis (2010–2020)
| Study | Year | Study design | Age (yr)a) | ECMO / totalb) | ECPR, n (%) | ECMO duration (hr)c) | Anticoagulation (monitoring) | LV decompression | ECMO survivald), n (%) | BTT, n (%) | Overall survivale), n (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Nahum et al. [30] | 2010 | Retrospective single centre | 0–8 | 12 | 0 (0) | 209.9±82.4 (range, 100–408) | N/A | N/A | 10/12 (83.3) | 0 (0) | 10/12 (083) |
| Hsu et al. [28] | 2011 | Retrospective single centre | 29.6±18.6 | 75 | 23/75 (31) | 171.5±121 | Heparin, CI (ACT, 180–200 sec) | LV drainage (7, 9%) | 48/75 (64) | 4 (17) | 48/75 (64) |
| ASD creation (1, 1%) | |||||||||||
| LA drainage (15, 20%) | |||||||||||
| Teele et al. [58] | 2011 | Retrospective single centre | 12.7 | 10/20 | 5/10 (50) | 151 (115–187) | N/A | LA drainage (3, 30%) | 7/10 (70) | 1 (10) | 17/20 (85) |
| (0–17.4) | BAS (5, 50%) | ||||||||||
| Wilmot et al. [71] | 2011 | Retrospective single centre | 6 (0–16) | 6/16 | 3/6 (50) | 120 (48–6,096) | N/A | N/A | 4/6 (66.7) | 1 (17) | 12/16 (75) |
| Ghelani et al. [14] | 2012 | Retrospective databasef) | 9±6.8 | 97/514 | N/A | N/A | N/A | N/A | 72/97 (74.2) | 21/514 (4.1) | 477/514 (93) |
| Lee et al. [75] | 2014 | Retrospective single centre | 4.33 (0.17–14) | 13/21 | 4/13 (31) | 168 (96–264) | Heparin, CI (ACT, 180–200 sec) | LA drainage (1, 8%) | 7/13 (53.8) | 0 (0) | 11/21 (64) |
| Nosaka et al. [76] | 2015 | Retrospective single centre | 5 (0–14) | 16/29 | 4/16 (25) | N/A | Heparin, CI (ACT, 180–200 sec) | N/A | 11/16 (68.8) | N/A | 24/29 (83) |
| Lin et al. [29] | 2016 | Retrospective single centre | 11.0 (4.0–15.3) | 7/13 | 3/7 (43) | 98 (70–141) | N/A | N/A | 7/7 (100) | 0 | 14/18 (78) |
| Jung et al. [37] | 2016 | Retrospective single centre | 2 (2.9 mo–11.5) | 13 | 3/13 (23) | 140 (51–425) | Heparin, CI (ACT, 160–180 sec) | ASD creation (6, 46%) | 12/13 (92.3) | 2 (15.4) | 12/13 (92.3) |
| Wu et al. [31] | 2017 | Retrospective single centre | 8.8±6.32 | 10/60 | N/A | N/A | N/A | N/A | 6/10 (60) | N/A | 54/60 (90) |
| Şık et al. [77] | 2018 | Retrospective single centre | 4.4 (1.1–11.7) | 6 | 1/6 (17) | 164 (79–402) | Heparin, CI (ACT, 170–220 sec) | N/A | 5/6 (83) | 0 (0) | 5/6 (83) |
ACT, activated clotting time; ASD, atrial septal defect; AV, atrioventricular; BAS, balloon-atrial septostomy; BTT, Bridge to transplant; CI, continuous infusion; ECMO, extracorporeal membrane oxygenation; ECPR, extracorporeal cardiopulmonary resuscitation; LA, left atrium; LV, left ventricle; N/A, not applicable.
Table 4.
Bleeding, thrombosis, and neurologic complications in children treated with ECMO
| Study | Design | Total population | Frequency (%) | Types | Risk factors |
|---|---|---|---|---|---|
| Bleeding | |||||
| Werho et al. [60] | Registry review | 21,845 | 39% | N/A | ↑ Age |
| Chest cannulation | |||||
| Longer ECMO run | |||||
| Dalton et al. [43] | Observational | 514 | 70% | Surgical site (36%) | ECPR |
| Chest tube (35%) | Organ failure | ||||
| Cannula site (34%) | |||||
| Thrombosis | |||||
| Dalton et al. [43] | Observational | 514 | 38% | Circuit thrombosis (31%) | VA-configuration |
| ICI (4%) | Haemolysis | ||||
| Limb ischemia (3%) | |||||
| Intracardiac clot (2%) | |||||
| Neurologic complications | |||||
| Said et al. [61] | Narrative review | N/A | N/A | Stroke (33%) | ECPR |
| Seizures (18%–23%) | Carotid cannulation | ||||
| ICH (11%–16%) | Younger age | ||||
| Brain death (10%) | Renal/hepatic failure | ||||
| Sepsis and acidosis | |||||
| Inotropic support | |||||
| ↓ PLT |
References
2. Extracorporeal Life Support Organization. ECLS Registry Report. International Summary 2020 January.
3. Mounts AW, Amr S, Jamshidi R, Groves C, Dwyer D, Guarner J, et al. A cluster of fulminant myocarditis cases in children, Baltimore, Maryland, 1997. Pediatr Cardiol 2001;22:34–9.


4. Amabile N, Fraisse A, Bouvenot J, Chetaille P, Ovaert C. Outcome of acute fulminant myocarditis in children. Heart 2006;92:1269–73.



5. Sankar J, Khalil S, Jeeva Sankar M, Kumar D, Dubey N. Short-term outcomes of acute fulminant myocarditis in children. Pediatr Cardiol 2011;32:885–90.


6. Forcada P, Beigelman R, Milei J. Inapparent myocarditis and sudden death in pediatrics. Diagnosis by immunohistochemical staining. Int J Cardiol 1996;56:93–7.


7. Weber MA, Ashworth MT, Risdon RA, Malone M, Burch M, Sebire NJ. Clinicopathological features of paediatric deaths due to myocarditis: an autopsy series. Arch Dis Child 2008;93:594–8.


8. Doolan A, Langlois N, Semsarian C. Causes of sudden cardiac death in young Australians. Med J Aust 2004;180:110–2.


9. deSa DJ. Isolated myocarditis as a cause of sudden death in the first year of life. Forensic Sci Int 1986;30:113–7.


10. Rasten-Almqvist P, Eksborg S, Rajs J. Myocarditis and sudden infant death syndrome. Apmis 2002;110:469–80.


11. Shimizu C, Rambaud C, Cheron G, Rouzioux C, Lozinski GM, Rao A, et al. Molecular identification of viruses in sudden infant death associated with myocarditis and pericarditis. Pediatr Infect Dis J 1995;14:584–8.


12. Dettmeyer R, Baasner A, Schlamann M, Haag C, Madea B. Coxsackie B3 myocarditis in 4 cases of suspected sudden infant death syndrome: diagnosis by immunohistochemical and molecular-pathologic investigations. Pathol Res Pract 2002;198:689–96.


13. Matsuura H, Ichida F, Saji T, Ogawa S, Waki K, Kaneko M, et al. Clinical features of acute and fulminant myocarditis in children - 2nd Nationwide Survey by Japanese Society of Pediatric Cardiology and Cardiac Surgery. Circ J 2016;80:2362–8.


14. Ghelani SJ, Spaeder MC, Pastor W, Spurney CF, Klugman D. Demographics, trends, and outcomes in pediatric acute myocarditis in the United States, 2006 to 2011. Circ Cardiovasc Qual Outcomes 2012;5:622–7.


15. Lyden DC, Olszewski J, Feran M, Job LP, Huber SA. Coxsackievirus B-3induced myocarditis. Effect of sex steroids on viremia and infectivity of cardiocytes. Am J Pathol 1987;126:432–8.


16. utts RJ, Boyle GJ, Deshpande SR, Gambetta K, Knecht KR, Prada-Ruiz CA, et al. Characteristics of clinically diagnosed pediatric myocarditis in a contemporary multi-center cohort. Pediatr Cardiol 2017;38:1175–82.


17. Canter CE, Simpson KE. Diagnosis and treatment of myocarditis in children in the current era. Circulation 2014;129:115–28.


18. Olinde KD, O'Connell JB. Inflammatory heart disease: pathogenesis, clinical manifestations, and treatment of myocarditis. Annu Rev Med 1994;45:481–90.


19. Klugman D, Berger JT, Sable CA, He J, Khandelwal SG, Slonim AD. Pediatric patients hospitalized with myocarditis: a multi-institutional analysis. Pediatr Cardiol 2010;31:222–8.


20. Isogai T, Yasunaga H, Matsui H, Tanaka H, Horiguchi H, Fushimi K. Effect of intravenous immunoglobulin for fulminant myocarditis on in-hospital mortality: propensity score analyses. J Card Fail 2015;21:391–7.


21. Li Y, Yu Y, Chen S, Liao Y, Du J. Corticosteroids and intravenous immunoglobulin in pediatric myocarditis: a meta-analysis. Front Pediatr 2019;7:342



22. Bejiqi R, Retkoceri R, Maloku A, Mustafa A, Bejiqi H, Bejiqi R. The diagnostic and clinical approach to pediatric myocarditis: a review of the current literature. Open Access Maced J Med Sci 2019;7:162–73.



23. Casadonte JR, Mazwi ML, Gambetta KE, Palac HL, McBride ME, Eltayeb OM, et al. Risk factors for cardiac arrest or mechanical circulatory support in children with fulminant myocarditis. Pediatr Cardiol 2017;38:128–34.


24. Chang YJ, Hsiao HJ, Hsia SH, Lin JJ, Hwang MS, Chung HT, et al. Analysis of clinical parameters and echocardiography as predictors of fatal pediatric myocarditis. PLoS One 2019;14:e0214087.



25. Inaba O, Satoh Y, Isobe M, Yamamoto T, Nagao K, Takayama M. Factors and values at admission that predict a fulminant course of acute myocarditis: data from Tokyo CCU network database. Heart Vessels 2017;32:952–9.


26. Schubert S, Opgen-Rhein B, Boehne M, Weigelt A, Wagner R, Müller G, et al. Severe heart failure and the need for mechanical circulatory support and heart transplantation in pediatric patients with myocarditis: Results from the prospective multicenter registry "MYKKE". Pediatr Transplant 2019;23:e13548.



27. Yang F, Wang J, Li W, Xu Y, Wan K, Zeng R, et al. The prognostic value of late gadolinium enhancement in myocarditis and clinically suspected myocarditis: systematic review and meta-analysis. Eur Radiol 2020;30:2616–26.


28. Hsu KH, Chi NH, Yu HY, Wang CH, Huang SC, Wang SS, et al. Extracorporeal membranous oxygenation support for acute fulminant myocarditis: analysis of a single center's experience. Eur J Cardiothorac Surg 2011;40:682–8.


29. Lin KM, Li MH, Hsieh KS, Kuo HC, Cheng MC, Sheu JJ, et al. Impact of extracorporeal membrane oxygenation on acute fulminant myocarditis-related hemodynamic compromise arrhythmia in children. Pediatr Neonatol 2016;57:480–7.


30. Nahum E, Dagan O, Lev A, Shukrun G, Amir G, Frenkel G, et al. Favorable outcome of pediatric fulminant myocarditis supported by extracorporeal membranous oxygenation. Pediatr Cardiol 2010;31:1059–63.


31. Wu HP, Lin MJ, Yang WC, Wu KH, Chen CY. Predictors of extracorporeal membrane oxygenation support for children with acute myocarditis. Biomed Res Int 2017;2017:2510695



32. Kurkluoglu M, Hynes CF, Alfares FA, El-Sayed Ahmed MM, Peer SM, Zurakowski D, et al. Choice of peripheral venoarterial extra-corporeal membrane oxygenation cannulation site in patients above 15 kilograms. J Card Surg 2015;30:461–5.


33. Gander JW, Fisher JC, Reichstein AR, Gross ER, Aspelund G, Middlesworth W, et al. Limb ischemia after common femoral artery cannulation for venoarterial extracorporeal membrane oxygenation: an unresolved problem. J Pediatr Surg 2010;45:2136–40.



34. Butt W, Heard M, Peek GJ. Clinical management of the extracorporeal membrane oxygenation circuit. Pediatr Crit Care Med 2013;14(5 Suppl 1): S13–9.


35. Ostadal P, Mlcek M, Kruger A, Hala P, Lacko S, Mates M, et al. Increasing venoarterial extracorporeal membrane oxygenation flow negatively affects left ventricular performance in a porcine model of cardiogenic shock. J Transl Med 2015;13:266



36. Van Linthout S, Tschöpe C. Inflammation - cause or consequence of heart failure or both? Curr Heart Fail Rep 2017;14:251–65.



37. Jung SY, Shin HJ, Jung JW, Park HK, Shin YR, Park YH, et al. Extracorporeal life support can be a first-line treatment in children with acute fulminant myocarditis. Interact Cardiovasc Thorac Surg 2016;23:24752

38. Kotani Y, Chetan D, Rodrigues W, Sivarajan VB, Gruenwald C, Guerguerian AM, et al. Left atrial decompression during venoarterial extracorporeal membrane oxygenation for left ventricular failure in children: current strategy and clinical outcomes. Artif Organs 2013;37:29–36.


39. Lin YJ, Liu HY, Kuo HC, Huang CF, Hsu MH, Cheng MC, et al. Left ventricle decompression strategies in pediatric peripheral extracorporeal membrane oxygenation. Acta Cardiol Sin 2019;35:335–41.


40. Rajagopal SK, Almond CS, Laussen PC, Rycus PT, Wypij D, Thiagarajan RR. Extracorporeal membrane oxygenation for the support of infants, children, and young adults with acute myocarditis: a review of the Extracorporeal Life Support Organization registry. Crit Care Med 2010;38:382–7.



41. Eastaugh LJ, Thiagarajan RR, Darst JR, McElhinney DB, Lock JE, Marshall AC. Percutaneous left atrial decompression in patients supported with extracorporeal membrane oxygenation for cardiac disease. Pediatr Crit Care Med 2015;16:59–65.


42. Russo JJ, Aleksova N, Pitcher I, Couture E, Parlow S, Faraz M, et al. Left ventricular unloading during extracorporeal membrane oxygenation in patients with cardiogenic shock. J Am Coll Cardiol 2019;73:654–62.


43. Dalton HJ, Reeder R, Garcia-Filion P, Holubkov R, Berg RA, Zuppa A, et al. Factors associated with bleeding and thrombosis in children receiving extracorporeal membrane oxygenation. Am J Respir Crit Care Med 2017;196:762–71.



44. Millar JE, Fanning JP, McDonald CI, McAuley DF, Fraser JF. The inflammatory response to extracorporeal membrane oxygenation (ECMO): a review of the pathophysiology. Crit Care 2016;20:387



45. Monagle P, Chan A, Massicotte P, Chalmers E, Michelson AD. Antithrombotic therapy in children: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest 2004;126(3 Suppl): 645S–687S.


46. Barton R, Ignjatovic V, Monagle P. Anticoagulation during ECMO in neonatal and paediatric patients. Thromb Res 2019;173:172–7.


47. Monagle P, Newall F, Campbell J. Anticoagulation in neonates and children: pitfalls and dilemmas. Blood Rev 2010;24:151–62.


48. O'Meara LC, Alten JA, Goldberg KG, Timpa JG, Phillips J, Laney D, et al. Anti-xa directed protocol for anticoagulation management in children supported with extracorporeal membrane oxygenation. ASAIO J 2015;61:339–44.


49. Ignjatovic V, Summerhayes R, Than J, Gan A, Monagle P. Therapeutic range for unfractionated heparin therapy: age-related differences in response in children. J Thromb Haemost 2006;4:2280–2.


50. Monagle P, Barnes C, Ignjatovic V, Furmedge J, Newall F, Chan A, et al. Developmental haemostasis. Impact for clinical haemostasis laboratories. Thromb Haemost 2006;95:362–72.


51. Cho HJ, Kim DW, Kim GS, Jeong IS. Anticoagulation therapy during extracorporeal membrane oxygenator support in pediatric patients. Chonnam Med J 2017;53:110–7.



52. Pieri M, Agracheva N, Bonaveglio E, Greco T, De Bonis M, Covello RD, et al. Bivalirudin versus heparin as an anticoagulant during extracorporeal membrane oxygenation: a case-control study. J Cardiothorac Vasc Anesth 2013;27:30–4.


53. Bembea MM, Annich G, Rycus P, Oldenburg G, Berkowitz I, Pronovost P. Variability in anticoagulation management of patients on extracorporeal membrane oxygenation: an international survey. Pediatr Crit Care Med 2013;14:e77–84.


54. Liveris A, Bello RA, Friedmann P, Duffy MA, Manwani D, Killinger JS, et al. Anti-factor Xa assay is a superior correlate of heparin dose than activated partial thromboplastin time or activated clotting time in pediatric extracorporeal membrane oxygenation*. Pediatr Crit Care Med 2014;15:e72–9.


55. Northrop MS, Sidonio RF, Phillips SE, Smith AH, Daphne HC, Pietsch JB, et al. The use of an extracorporeal membrane oxygenation anticoagulation laboratory protocol is associated with decreased blood product use, decreased hemorrhagic complications, and increased circuit life. Pediatr Crit Care Med 2015;16:66–74.


56. Hervey-Jumper SL, Annich GM, Yancon AR, Garton HJ, Muraszko KM, Maher CO. Neurological complications of extracorporeal membrane oxygenation in children. J Neurosurg Pediatr 2011;7:338–44.


57. Hardart GE, Fackler JC. Predictors of intracranial hemorrhage during neonatal extracorporeal membrane oxygenation. J Pediatr 1999;134:156–9.


58. Teele SA, Allan CK, Laussen PC, Newburger JW, Gauvreau K, Thiagarajan RR. Management and outcomes in pediatric patients presenting with acute fulminant myocarditis. J Pediatr 2011;158:638–43. e1.


59. Cho SM, Farrokh S, Whitman G, Bleck TP, Geocadin RG. Neurocritical care for extracorporeal membrane oxygenation patients. Crit Care Med 2019;47:1773–81.


60. Werho DK, Pasquali SK, Yu S, Donohue J, Annich GM, Thiagarajan RR, et al. Hemorrhagic complications in pediatric cardiac patients on extracorporeal membrane oxygenation: an analysis of the Extracorporeal Life Support Organization Registry. Pediatr Crit Care Med 2015;16:27688
61. Said AS, Guilliams KP, Bembea MM. Neurological monitoring and complications of pediatric extracorporeal membrane oxygenation support. Pediatr Neurol 2020;108:31–9.



62. Extracorporeal Life Support Organization. Guidelines for Pediatric Cardiac Failure [Internet]. Ann Arbor (MI), Extracorporeal Life Support Organization. [cited Year Month Day]. Available from: http://www.elso.org/resources/guidelines.aspx.
63. Aissaoui N, Luyt CE, Leprince P, Trouillet JL, Leger P, Pavie A, et al. Predictors of successful extracorporeal membrane oxygenation (ECMO) weaning after assistance for refractory cardiogenic shock. Intensive Care Med 2011;37:1738–45.


64. Duncan BW, Bohn DJ, Atz AM, French JW, Laussen PC, Wessel DL. Mechanical circulatory support for the treatment of children with acute fulminant myocarditis. J Thorac Cardiovasc Surg 2001;122:440–8.


65. Foerster SR, Canter CE, Cinar A, Sleeper LA, Webber SA, Pahl E, et al. Ventricular remodeling and survival are more favorable for myocarditis than for idiopathic dilated cardiomyopathy in childhood: an outcomes study from the Pediatric Cardiomyopathy Registry. Circ Heart Fail 2010;3:689–97.


66. Morales DLS, Rossano JW, VanderPluym C, Lorts A, Cantor R, St Louis JD, et al. Third Annual Pediatric Interagency Registry for Mechanical Circulatory Support (Pedimacs) report: preimplant characteristics and outcomes. Ann Thorac Surg 2019;107:993–1004.


67. Yoshioka D, Takeda K, Takayama H, Naka AY. Low INTERMACS profiles: one-stage durable LVAD implantation for INTERMACS level 1: indications and contraindications. Montalto A, Loforte A, Musumeci F, Krabatsch T, Slaughter MS, editors. Mechanical circulatory support in end-stage heart failure: a practical manual. Cham: Springer International Publishing, 2017;:115–9.
68. McCarthy RE 3rd, Boehmer JP, Hruban RH, Hutchins GM, Kasper EK, Hare JM, et al. Long-term outcome of fulminant myocarditis as compared with acute (nonfulminant) myocarditis. N Engl J Med 2000;342:690–5.


69. Boyle K, Felling R, Yiu A, Battarjee W, Schwartz JM, Salorio C, et al. Neurologic outcomes after extracorporeal membrane oxygenation: a systematic review. Pediatr Crit Care Med 2018;19:760–6.



70. Xiong H, Xia B, Zhu J, Li B, Huang W. Clinical outcomes in pediatric patients hospitalized with fulminant myocarditis requiring extracorporeal membrane oxygenation: a meta-analysis. Pediatr Cardiol 2017;38:20914

71. Wilmot I, Morales DL, Price JF, Rossano JW, Kim JJ, Decker JA, et al. Effectiveness of mechanical circulatory support in children with acute fulminant and persistent myocarditis. J Card Fail 2011;17:487–94.


72. Sadhwani A, Cheng H, Stopp C, Rollins CK, Jolley MA, Dunbar-Masterson C, et al. Early neurodevelopmental outcomes in children supported with ECMO for cardiac indications. Pediatr Cardiol 2019;40:1072–83.



73. Schlapbach LJ, Chiletti R, Straney L, Festa M, Alexander D, Butt W, et al. Defining benefit threshold for extracorporeal membrane oxygenation in children with sepsis-a binational multicenter cohort study. Critical Care (London, England) 2019;23:429



74. Kociol RD, Cooper LT, Fang JC, Moslehi JJ, Pang PS, Sabe MA, et al. Recognition and initial management of fulminant myocarditis: a scientific statement from the American Heart Association. Circulation 2020;141:e69–92.

75. Lee EY, Lee HL, Kim HT, Lee HD, Park JA. Clinical features and shortterm outcomes of pediatric acute fulminant myocarditis in a single center. Korean J Pediatr 2014;57:489–95.








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


