Article Contents
| Clin Exp Pediatr > Volume 63(10); 2020 |
|
A 6-year-old boy was admitted with concerns of abdominal pain, high-grade fever, and increasing generalized swelling of the body for 20 days. The abdominal pain was severe in intensity, diffuse in nature and the child was not able to sit up or stand. It was associated with decreased urine output. Child had no previous history of similar complains, there was no history of any recurrent infections, hospitalizations, or contact with a tuberculosis case. The child was hemodynamically stable with heart rate of 90 beats/min, respiratory rate of 26 breaths/min, and blood pressure of 104/70 mm Hg (50th–90th percentile). The weight was 19.6 kg (body mass index, 15.08 kg/m2; World Health Organization weight for age z score, -0.17). General physical examination revealed generalized oedema. Abdominal examination revealed a distended abdomen with severe tenderness in the left iliac fossa and left lumbar region associated with guarding of the abdomen. Child kept his left thigh in flexed position and attempted extension elicited severe pain. However, movements at right hip joint were normal. The rest of the examination was normal. Given the history of generalized edema in a well-nourished child, in the absence of any cardiovascular or liver manifestations, a provisional diagnosis of nephrotic syndrome was made. Urine analysis showed spot urine protein to creatinine ratio of 4.7 mg/mg. Serum albumin levels was low (1.5 g/dL), serum triglycerides (230 mg/dL), and total cholesterol levels (237 mg/dL) were elevated for age. Laboratory results are listed in Table 1.
1. What could be the most likely cause of pain abdomen in this case scenario?
(a) Urinary tract infection (UTI)
(b) Peritonitis
(c) Septic arthritis
(d) Psoas abscess
A good history and clinical examination with specific investigations are crucial in managing such cases. The child was diagnosed to have nephrotic syndrome by history of generalized edema with supporting laboratory findings of proteinuria, hypoalbuminemia, and hypercholesterolemia. The possible etiologies of pain abdomen in children with nephrotic syndrome include severe hypovolemia leading to mesenteric ischemia [1], pain due to distension as a result of massive ascites, spontaneous bacterial peritonitis (SBP), gastroenteritis, UTI [1], adverse effect of corticosteroids leading to gastritis, mesenteric ischemia due to thromboembolism [2-4], acute appendicitis, intussusception due to severe gut wall oedema [5-7] and rarely even pancreatitis or retroperitoneal abscesses. Our patient had a striking history of high-grade fever, pain abdomen, flexed position of left hip, and pain exacerbating with attempted extension of the left hip (psoas sign). Psoas abscess is a rare infectious cause of pain abdomen in nephrotic syndrome. The classical triad of the disease: fever, back pain/flank pain and limited movements of hip joint may or may not be observed in each case. Psoas muscle arises from the lateral borders of T12 to the L5 vertebrae and is inserted into the lesser trochanter of the femur. It is a flexor of the hip joint; hence any inflammation of this muscle leads to pain on passively stretching (extension of hip joint) the muscle. A close differential could be septic arthritis. Unlike septic arthritis, hip pain in patients with psoas abscesses is usually diminished with hip flexion. Psoas abscess patients frequently prefer to be in a position of less discomfort that includes hip flexion and lumbar lordosis. We decided to investigate further for diagnosing this clinical entity.
2. Complications of nephrotic syndrome include
(a) Hypovolemia
(b) Thrombosis
(c) Infections
(d) All the above
Nephrotic syndrome is diagnosed due to its classic presentation and laboratory analysis. However, the disease may also be revealed by a complication, like psoas abscess in this patient. Complications inherent to the pathophysiology of nephrotic syndrome include hypovolemia, infections, thrombosis, acute kidney injury, and effects on growth. Pain abdomen is an important presenting complain in most of these complications. Meticulous evaluation of pain abdomen is important, because the treatment is different depending upon the etiology. In a setting of tachycardia, with low volume pulses with a history of pain abdomen and gastrointestinal (GI) losses in the form of vomiting and diarrhoea with high urea, hypovolemia should be considered. UTI if present, causes pain abdomen in nephrotic children. Clinical clues include associated fever, frequency, hesitancy, and burning micturition. In children with ascites with tenderness/guarding or rigidity on palpation; an ascitic tap should be done. Ascitic fluid cytology showing >100 leukocytes/mm3 with >50% neutrophils are suggestive of SBP [1]. Ultrasound abdomen should be done to diagnose any intra-abdominal abscesses, intussusception, or appendicitis. Renal vein thrombosis is suspected in a patient with oliguria, anuria, haematuria, or flank pain, especially following an episode of dehydration. Femoral and mesenteric arterial thrombosis may occasionally occur. Ultrasound doppler may be considered in such cases. More common reasons of pain abdomen include rapid formation of ascites and gastritis due to treatment with steroids. Though, our case of psoas abscess is an exceedingly rare initial presentation of nephrotic syndrome, it is important to correctly diagnose and adequately treat the condition to prevent significant morbidity and mortality. The present case also adds to the clinical spectrum of manifestations in nephrotic syndrome.
3. Imaging modality of choice for psoas abscess
(a) Ultrasound abdomen
(b) Contrast-enhanced computed tomography (CECT)
(c) Magnetic resonance imaging
(d) Abdominal plain radiographs
The diagnosis of a psoas abscess may be suspected on clinical grounds and confirmed on imaging studies. Identification of the etiological organism requires blood cultures or aspirated pus. Complete blood count, inflammatory markers, and complete urine analysis were requested. The laboratory results showed leucocytosis 17.7×103 cells/mm3, positive C-reactive protein (CRP) 20.6 mg/dL, normal erythrocyte sedimentation rate and no growth on urine and blood cultures. An ascitic tap was done which ruled out SBP (25 cells; all lymphocytes). In view of positive psoas sign, ultrasonography (USG) of the abdomen was done which showed moderate ascites with loculated collection with septae in the left psoas muscle, exact extent of which could not be made out. CECT abdomen showed multiloculate collection with peripheral enhancing wall noted in left retroperitoneum, left psoas muscle, and latissimus dorsi, also abutting anterior wall of left iliacus muscle and external oblique muscle (Fig 1). Ultrasound of the hip joint was done which showed no collection which ruled out septic arthritis.
Computed tomography (CT) is the optimal radiographic modality to evaluate psoas abscess. In most cases abscess is well delineated by CT [8]. Though ultrasound may be diagnostic in 50% cases, it is limited by low sensitivity and specificity. Magnetic resonance imaging may be used for improved definition of soft tissues and vertebral bodies [9]. Limited reports exist regarding the use of positron emission tomography-CT in psoas abscess.
4. How will you manage the above patient?
(a) Surgical drainage of the abscess with steroids for nephrotic syndrome
(b) Percutaneous drainage of the abscess with steroids for nephrotic syndrome
(c) Abscess drainage with appropriate antibiotic therapy followed by steroids
(d) Steroids for nephrotic syndrome with appropriate antibiotics for abscess
The standard treatment of first episode nephrotic syndrome (FENS) includes steroid therapy for 12 weeks, started immediately following the diagnosis. However, in scenarios of nephrotic syndrome presenting with complications, treatment must be tailored. Infections must be treated before initiating steroids to prevent the risk of sepsis. Management of psoas abscess consists of drainage and prompt initiation of appropriate antibiotic therapy.
Our child was started on empirical antibiotic therapy (intravenous [IV] antibiotics: ceftriaxone, vancomycin) to cover methicillin resistant staphylococcus aureus and enteric organisms with simultaneously percutaneous abscess drainage. USG guided drainage of abscess was done and 50 mL thick pus aspirated. Pus Cultures showed growth of staphylococcus aureus which was sensitive to vancomycin. IV antibiotic therapy was given for 2 weeks. Daily urine analysis revealed persistent nephrotic range proteinuria (urine dipstick 4+, spot urine protein to creatinine ratio 4.5). Serum albumin levels dropped to below 1 mg/dL with significant fluid overload causing respiratory distress on day 8 of therapy. IV albumin infusions were given control the edema. Supportive care was continued with salt-restricted diet. In due course, patients clinical condition improved. Leucocytosis resolved (8×103 cells/mm3), CRP decreased (5.2 mg/dL). After 2 weeks of IV antibiotics, repeat USG was done which showed resolution of abscess, following which child was shifted to combination of oral linezolid and cefpodoxime for 4 weeks. After 3 weeks of antibiotics, CRP normalised (0.5 md/dL) and he was started on oral prednisolone (2 mg/kg/day). Child attained remission after 2 weeks of daily prednisolone and received standard therapy for FENS. Child was discharged and is in regular follow-up. Patient is in remission, with complete resolution of symptoms and no further relapses. Follow-up CT was not done, as the child was asymptomatic. To the best of our knowledge there is only one such reported case in paediatric age group in a 6-year-old girl who was on alternate day treatment with prednisolone 1.5 mg/kg for 3 months [10]. This child was on immunosuppressants, however our patient (FENS) presented with psoas abscess, and was not on any immunosuppressive medication. The plausible explanation in our patient is abnormal T-lymphocyte function and hypoproteinaemia particularly hypogammaglobulinemia, decreased complement factors B (C3 proactivator) and D, intrinsic to pathophysiology of nephrotic syndrome. Oedematous tissue also behaves as a culture medium for bacterial growth.
Pain abdomen is a common presenting symptom in children with nephrotic syndrome and they are prone to infections due to their immunocompromised status. A high level of suspicion is necessary for diagnosing rare occult causes of pain abdomen in nephrotic syndrome. Psoas abscess, though a rare manifestation can portend significant morbidity and mortality.
Answers
1. (d) 2. (d) 3. (b) 4. (c)
Fig. 1.
Contrast-enhanced computed tomography image of the abdomen revealing a multiloculated collection 44.8 mm×58.8 mm×95.8 mm (anteroposterior × transverse × craniocaudal) with a peripherally enhanced wall noted in the left retroperitoneum, left psoas muscle, and latissimus dorsi abutting the anterior wall of the left iliacus muscle and external oblique muscle. The collection was tracked from the lower pole of the left kidney up to the left iliac fossa region.
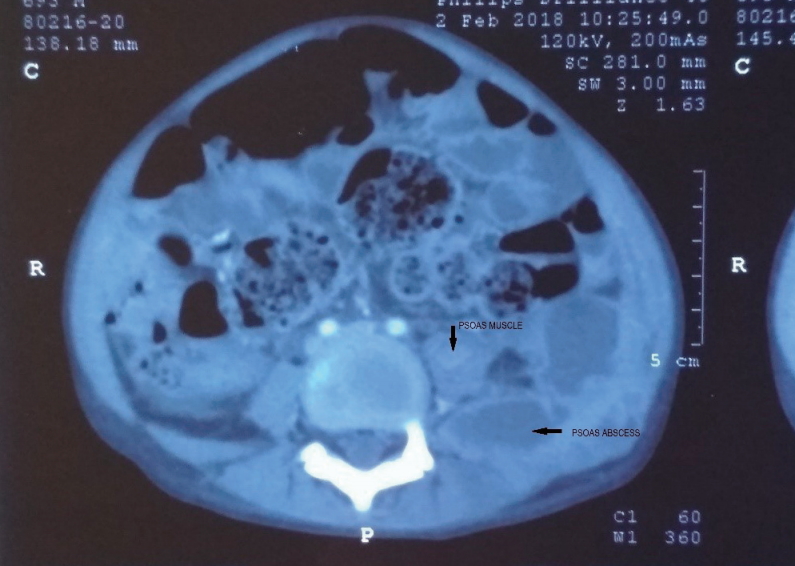
Table 1.
Investigations at presentation
References
1. Indian Pediatric Nephrology Group; Indian Academy of Pediatrics; Bagga A, Ali U, Banerjee S, Kanitkar M, et al. Management of steroid sensitive nephrotic syndrome: revised guidelines. Indian Pediatr 2008;45:203–14.


2. Etoh Y, Ohsawa I, Fujita T, Fuke Y, Endo M, Ohi H, et al. Nephrotic syndrome with portal, splenic and renal vein thrombosis. A Case Report. Nephron 2002;92:680–4.


3. Llach F. Hypercoagulability, renal vein thrombosis, and other thrombotic complications of nephrotic syndrome. Kidney Int 1985;28:429–39.


4. Vaziri ND, Branson HE, Ness R. Changes of coagulation factors IX, VIII, VII, X, and V in nephrotic syndrome. Am J Med Sci 1980;280:167–71.


5. Takano T, Nakagawa M, Ishiwara H. Jaundice, vomiting and abdominal pain (nephrotic syndrome): intussusception (with lipoid nephrosis). Nishin Rinsho 1975;Spec No:978-9:1502–5.
6. Asai K, Tanaka S, Tanaka N, Tsumura K, Kato F, Kikuchi K. Intussusception of the small bowel associated with nephrotic syndrome. Pediatr Nephrol 2005;20:1818–20.



7. Kelly GS, Brady TM. A child with nephrotic syndrome and abdominal pain. Clin Pediatr (Phila) 2016;55:683–5.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


