Article Contents
| Korean J Pediatr > Volume 61(8); 2018 |
|
Abstract
Purpose
Hypothermia at admission is associated with increased mortality and morbidity in preterm infants. We performed a quality improvement (QI) effort to determine the impact of a decrease in admission hypothermia in preterm infants.
Methods
The study enrolled very low birth weight (VLBW) infants born at Gangnam Severance Hospital between January 2013 and December 2016. This multidisciplinary QI effort included the use of occlusive wraps, warm blankets, and caps; the delivery room temperature was maintained above 23.0˚C, and a check-list was used for feedback.
Results
Among 259 preterm infants, the incidence of hypothermia (defined as body temperature <36.0˚C) decreased significantly from 68% to 41%, and the mean body temperature on neonatal intensive care unit admission increased significantly from 35.5˚C to 36.0˚C. In subgroup analysis of VLBW infants, admission hypothermia and neonatal outcomes were compared between the pre-QI (n=55) and post-QI groups (n=75). Body temperature on admission increased significantly from 35.4˚C to 35.9˚C and the number of infants with hypothermia decreased significantly from 71% to 45%. There were no cases of neonatal hyperthermia. The incidence of pulmonary hemorrhage was significantly decreased (P=0.017). Interaction analysis showed that birth weight and gestational age were not correlated with hypothermia following implementation of the protocol.
Neonatal hypothermia is associated with increased mortality and morbidity [1-4]. Despite the advancements in perinatal intensive care, hypothermia continues to remain a significant challenge, especially in very low birth weight (VLBW) infants. According to the Korean Neonatal Network data, the body temperature in VLBW infants at admission to the neonatal intensive care unit (NICU) is is 36.0°C±0.7°C, the incidence of hypothermia was 77.1% [5].
Infants, especially those that are preterm, are susceptible to cold stress during the perinatal period owing to their large surface area-to-body mass ratio, immature skin [6], decreased subcutaneous fat, poor vasomotor control, and lack of shivering response [7]. Hypothermia is associated with increased glucose consumption, and the resultant hypoglycaemia and hypoxemia contribute to metabolic acidosis, pulmonary vasoconstriction, and respiratory difficulty [8-10]. In addition, neonatal hypothermia is associated with an increased risk of infection , coagulation defects [11], delayed readjustment from foetal to newborn circulation [12], hyaline membrane disease (HMD), acute renal failure, necrotizing enterocolitis, brain haemorrhage, and in extreme cases, mortality [2,13]. The EPICure study identified hypothermia immediately after birth in extremely preterm infants as a risk factor for mortality [2]. Interventions to reduce heat loss in preterm infants have been extensively studied and include polyethylene wraps, plastic bags, and exothermic mattresses [14-17]. Although several studies have demonstrated a reduction in hypothermia in VLBW infants by using standardized protocols, these protocols have failed to completely avoid hypothermia [18-20].
This study was performed as a quality improvement (QI) effort to prevent hypothermia in preterm infants by reducing heat loss. In addition, the study also analyzed the mortality and morbidity in pre-QI group and post-QI group.
All preterm infants between 24 weeks 0 days and 34 weeks 0 days of gestation that were born at the Gangnam Severance Hospital from January 2013 to December 2016 were included in this prepost design QI study. The period from January to February 2015 were excluded as the standardized protocol was being set up. The multidisciplinary standardized protocol was operational from March 2015 and all infants born in this period were included as the post-QI group. We excluded infants that required operation or receiving invasive procedures immediately after delivery. Subgroup analysis to especially VLBW infants was done for comparing the admission hypothermia and neonatal outcomes between the pre-QI and post-QI groups. Ethical approval for this retrospective study was obtained from the ethics committees of Gangnam Severance Hospital and informed consent was waived (approval number: 3-2017-0155).
We formed a multidisciplinary team consisting of a neonatologist, an obstetrician, and a nurse in the NICU, the delivery room (DR), and the quality management cell, respectively. A standardized protocol was developed and the compliance with the protocol was verified by using a checklist. The collected data were analyzed, and used for providing regular feedback to the multidisciplinary team and for guiding the team leaders in providing comparisons and in educating the different team members about thermoregulation.
The routine measures of ensuring thermoregulation postnatal care including the use of radiant warmers, thorough drying of the infant immediately after delivery, and the use of blankets, applied to the pre-QI and the post-QI groups.
The new thermoregulation protocol was introduced into practice in March 2015, and included the use of occlusive wraps, prewarmed blankets, and caps for all infants, in addition to maintaining the temperature of the DR and the operating room (OR) above 23.0°C. Axillary temperature of all infants was measured at their admission to the NICU (Fig. 1).
All data were prospectively collected, and included the following information: date of birth, prenatal interventions such as in vitro fertilization, birth weight, gestational age, sex, Apgar score, axillary temperature taken immediately after admission to the NICU, capillary blood gas analysis at admission (i-STAT System, Abbott Point of Care, Inc., Princeton, NJ, USA), and ambient air temperatures in the DR/OR and the NICU. The axillary and room temperatures were obtained using digital thermometers.
Hypothermia and hyperthermia were defined according to the World Health Organization (WHO) guidelines as body temperature <36.0°C and >38.0°C, respectively. The following definitions were applied in identifying comorbid conditions: patent ductus arteriosus (PDA), if surgery or medication was needed; HMD, requiring exogenous surfactant; bronchopulmonary dysplasia (BPD), as per the National Institutes of Health classification; and intraventricular haemorrhage (IVH), when greater than grade II. Outcome parameters included the incidence of hypothermia, hyperthermia, mortality and morbidity (i.e., PDA, HMD, BPD, IVH, PVL, sepsis, and pulmonary haemorrhage) among all preterm VLBW infants.
IBM SPSS Statistics ver. 23.0 (IBM Co., Armonk, NY, USA) was used to evaluate the data. Categorical data were expressed as numbers and percentages and compared using chi-square or Fisher exact test. Continuous data were expressed as mean±standard deviation and compared using the independent 2 sample t test. Correlations between continuous data were evaluated using the Pearson correlation coefficient. Interaction analysis was carried out to examine the correlation between the temperature and basic characteristics
The new thermoregulation protocol was introduced into practice in March 2015. The bundle approach for postnatal therrmoregulation including the use of radiant warmers, blankets, prepared wrap and cap applied to all infants during post-QI phase. All measured temperature at DR and OR achieved above 23.0°C. All including infants during post-QI phase, used checklist for completion of thermoregulation protocol. Axillary temperature of all infants measured at their admission to the NICU was recorded (Fig. 1).
The number of infants born during the pre-QI and the post-QI phase were 125 and 134, respectively. The average gestational age was 30.1±3.6 weeks (range, 23.1–34.0 weeks), and birth weight was 1,420±510 g (range, 420–2,660 g) without significant difference between pre-QI and post-QI group. The overall incidence of hypothermia was significantly declined from 68% to 41%. The mean body temperature at admission to NICU was significantly increased from 35.5°C±0.9°C to 36.0°C±0.7°C.
Among them, a total of 130 VLBW cases included in the subgroup analysis; 55 (42%) in the preimplementation and 75 (58%) in the postimplementation phase. The baseline characteristics of the study population are shown in Table 1. Both the groups were similar in terms of their gestational age, birth weight, gender, the mode of delivery, and type of pregnancy. The overall incidence of hypothermia in VLBW infants was significantly lesser following the intervention (71% vs. 45%, P=0.003). The mean body temperature at admission to NICU in the post-QI group was 35.9°C±0.5°C compared to 35.4°C±0.7°C in the pre-QI group (P<0.001) (Table 2). There were no cases of hyperthermia during the study period. The mean DR temperature was 25.4°C±3.3°C and mean OR was 24.0°C±1.3°C. The mean duration at DR and OR stay was 9.2±4.0 minutes.
There was no significant difference in the incidence of other secondary outcomes, namely PDA, HMD, BPD, IVH, PVL, and sepsis. However, there was a significant reduction in the incidence of pulmonary haemorrhage in the post-QI group when compared to the pre-QI group (P=0.017) (Table 3).
In the pre-QI group, the body temperature at NICU admission was correlated with birth weight (P=0.025) and gestational age (P=0.016), however, not correlated in the post-QI group (P=0.658, P=0.054, respectively) (Table 4). We concluded assessment of new thermoregulation protocol showed no significant influence of lower birth weight and gestation using interaction analysis.
Additional efforts in preventing newborn hypothermia are required notwithstanding the advancements in perinatal care and the adherence to the WHO guidelines for the routine care for hypothermia [21]. This quality initiative demonstrated that this goal may be achieved by an interdepartmental and interdisciplinary standardized protocol and checklist based feedback [18-20].
Heat loss in newborn infants occurs through 4 mechanisms: conduction, convection, radiation, and evaporation. Maintenance of normothermia in newborns requires strategies that counteract these mechanisms of heat loss [22,23]. The WHO in its revised publication titled, “Thermal protection of the new born (1997),” introduced the warm chain technique, which interlinked procedures carried out at birth and later, in order to prevent hypothermia in all newborns [21]. However, most premature infants continue to have hypothermia on admission to the NICU. Wilson et al. [24] report that hypothermia occurred in 53.4% of 5,697 infants born at <32-week gestational age in a population-based study with samples from 11 European countries. More concerning is that 12.9% of the population had admission temperatures below 35.5°C [25]. We also showed the overall incidence of hypothermia was 68% in preintervention routine care.
The different interventions to maintain normothermia have been extensively investigated. One study demonstrated that wrapped infants had a higher admission temperature (36.5°C) compared to control infants (35.6°C) (P=0.002) [26]. Other randomized controlled trial showed that the use of plastic bag was more likely to result in normothermia when compared to the pre-QI group managed with standard thermoregulation care, among preterm infants with a birth weight of 1,000 to 2,500 g (P=0.007) [14]. The effectiveness of the self-heating acetate gel mattresses was evaluated and the ratio of hypothermia decreased from 22.6% to 3.3% (P<0.001), at the same time hyperthermia rose from 30.1% to 49.6% (P=0.004) [16]. In a study comparing vinyl bag and polyethylene wrap in preterm infants at ≤32-week gestation, the use of vinyl bag was reported to be more effective at maintaining body temperature during the first 60 minutes following birth (P=0.041) [27]. Retrospective observational study was conducted and demonstrated that the hypothermia rate was lesser in the ‘bag and mattress’ group (26%) compared to the ‘bag only’ (69%) and traditional care groups (84%) [17]. More recently, efforts at improving quality have focused on using ‘intervention bundles’ such as staff education, maintenance of consistent room temperature, use of polyethylene wraps or vinyl bags, caps, and warmed incubators for infant transfers [18-20]. In this QI efforts, mixed bundles included the use of polyethylene wraps, prewarmed blankets, caps, and maintenance of ambient air temperature in the DR/OR above 23°C was applied in addition to the involvement of dedicated staff to monitor temperature.
A multidisciplinary practice plan was implemented to reduce hypothermia in preterm infants. One study showed admitting temperature increased from 36.0°C to 36.3°C, to 36.7°C at baseline, phasing in, and full implementation, respectively (P<0.001) [28]. Similar results have also been reported from other centres in Korea. Kim et al. [18] investigated the effect of clinical strategy in preventing hypothermia in infants between 23 and 24 weeks of gestation; in their study, the NICU admission temperature increased from 34.7°C to 35.3°C on implementing measures that included controlling the temperature of the OR/DR and the NICU, covering the body and head with wrap. Our study has demonstrated that by standardizing the management of thermoregulation from the predelivery period resulted in a reduction in the hypothermia rate of VLBW infants from 70.9% to 45.3% and these results are consistent with other QI efforts.
De Almeida et al. [29] identified DR temperature<25°C, gestational age<32 weeks and absence of plastic bag/wrap as independent risk factors for hypothermia at 5 minutes after birth. These factors were strongly associated with hypothermia at NICU admission (odds ratio, 7.45). Exposure to cold air during resuscitation and transport were also independent risk factors for hypothermia at NICU admission. In our study, we maintained the OR/DR temperatures>23°C, and applied wrap and cap to all infants. Strong correlation between NICU admission temperatures and gestational age, and birth weight in the preimplementation phase was noted.
However, after intervention phase, gestation and birth weight showed no more statistical significance according to interaction analysis. In addition, the distance between the OR/DR and the NICU or using warm air during resuscitation and transport should also be taken into consideration.
Admission hypothermia in the neonatal population is associated with increased mortality and morbidity. Hypothermia at NICU admission resulted in a 1.64-fold increase in the chance of early neonatal death (95% confidence interval, 1.03–2.61) [29]. Others showed moderate hypothermia after birth in VLBW infants was associated with PPHN and moderate to severe BPD [30]. Pulmonary haemorrhage is associated with cold injury [31-33]. Most of all, we found the reduction in the admission hypothermia appeared to be associated with a reduction in the incidence of massive pulmonary haemorrhage from 10.9% to 1.3% (P=0.017).
This study had several limitations. The pre-post design of the study showed to makes a connection between intervention and reducing admission hypothermia. However, randomized controlled trial of intervention was needed. Our QI efforts were limited to a single center. Additionally, collecting the body temperature at 5 minutes after birth and analyzing the difference between at 5 minutes and at admission may help in identifying other risk factors.
In conclusion, this study demonstrated that a standardized protocol can help in reducing the incidence of admission hypothermia in preterm infants and improve early neonatal survival. It may also reduce the incidence of massive pulmonary haemorrhage. The QI achieved by this effort can only be maintained with routine surveillance of admission temperatures.
Fig. 1.
Body temperatures recorded at neonatal intensive care unit admission during post-QI phase. NICU, neonatal intensive care unit; VLBW, very low birth weight; QI, quality improvement.
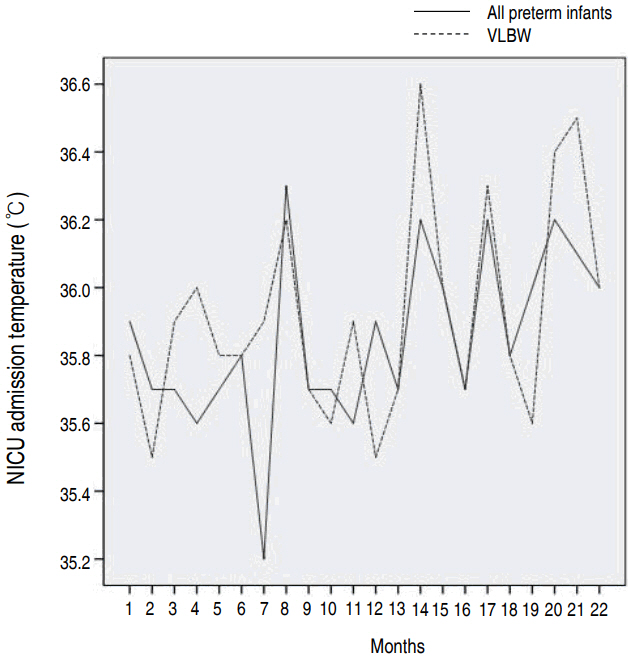
Table 1.
Baseline characteristics
Table 2.
Axillary temperature taken immediately after admission to the neonatal intensive care unit
| Variable | Pre-QI group (n=55) | Post-QI group (n=75) | P value |
|---|---|---|---|
| Body temperature (°C) | 35.4±0.7 | 35.9±0.5 | <0.001 |
| Normothermia* | 16 (29.1) | 41 (54.7) | 0.003 |
Table 3.
Comparison of morbidity and mortality between pre-QI and post-QI groups
Table 4.
Correlation between body temperature and basic characteristics and interaction analysis in the pre-QI and post-QI groups
References
1. Acolet D, Elbourne D, McIntosh N, Weindling M, Korkodilos M, Haviland J, et al. Project 27/28: inquiry into quality of neonatal care and its effect on the survival of infants who were born at 27 and 28 weeks in England, Wales, and Northern Ireland. Pediatrics 2005;116:1457–65.


2. Costeloe K, Hennessy E, Gibson AT, Marlow N, Wilkinson AR. The EPICure study: outcomes to discharge from hospital for infants born at the threshold of viability. Pediatrics 2000;106:659–71.


3. Laptook AR, Salhab W, Bhaskar B, Neonatal Research Network. Admission temperature of low birth weight infants: predictors and associated morbidities. Pediatrics 2007;119:e643–9.


4. Miller SS, Lee HC, Gould JB. Hypothermia in very low birth weight infants: distribution, risk factors and outcomes. J Perinatol 2011;31 Suppl 1:S49–56.



5. Cho SJ, Shin J, Namgung R. Initial resuscitation at delivery and short term neonatal outcomes in very-low-birth-weight infants. J Korean Med Sci 2015;30 Suppl 1:S45–51.


7. Scopes JW, Tizard JP. The effect of intravenous noradrenaline on the oxygen consumption of new-born mammals. J Physiol 1963;165:305–26.



8. Elliott RI, Mann TP. Neonatal cold injury due to accidental exposure to cold. Lancet 1957;272:229–34.

9. Gandy GM, Adamsons K Jr, Cunningham N, Silverman WA, James LS. Thermal environment and acid-base homeostasis in human infants during the first few hours of life. J Clin Invest 1964;43:751–8.



10. McCall EM, Alderdice F, Halliday HL, Jenkins JG, Vohra S. Interventions to prevent hypothermia at birth in preterm and/or low birthweight infants. Cochrane Database Syst Rev 2010;CD004210


11. Chadd MA, Gray OP. Hypothermia and coagulation defects in the newborn. Arch Dis Child 1972;47:819–21.



12. Stephenson JM, Du JN, Oliver TK Jr. The effect of cooling on blood gas tensions in newborn infants. J Pediatr 1970;76:848–52.


13. Wyckoff MH, Aziz K, Escobedo MB, Kapadia VS, Kattwinkel J, Perlman JM, et al. Part 13: Neonatal Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2): S543–60.


14. Leadford AE, Warren JB, Manasyan A, Chomba E, Salas AA, Schelonka R, et al. Plastic bags for prevention of hypothermia in preterm and low birth weight infants. Pediatrics 2013;132:e128–34.



15. Doglioni N, Cavallin F, Mardegan V, Palatron S, Filippone M, Vecchiato L, et al. Total body polyethylene wraps for preventing hypothermia in preterm infants: a randomized trial. J Pediatr 2014;165:261–6.


16. Ibrahim CP, Yoxall CW. Use of self-heating gel mattresses eliminates admission hypothermia in infants born below 28 weeks gestation. Eur J Pediatr 2010;169:795–9.


17. Singh A, Duckett J, Newton T, Watkinson M. Improving neonatal unit admission temperatures in preterm babies: exothermic mattresses, polythene bags or a traditional approach? J Perinatol 2010;30:45–9.



18. Kim JK, Seong SI, Shin JH, Jeong JM, Ahn SY, Kim ES, et al. Effect of clinical strategy for preventing heat loss between 23 and 24 weeks of gestation. J Korean Soc Neonatol 2012;19:26–31.

19. Manani M, Jegatheesan P, DeSandre G, Song D, Showalter L, Govindaswami B. Elimination of admission hypothermia in preterm very low-birth-weight infants by standardization of delivery room management. Perm J 2013;17:8–13.




20. Pinheiro JM, Furdon SA, Boynton S, Dugan R, Reu-Donlon C, Jensen S. Decreasing hypothermia during delivery room stabilization of preterm neonates. Pediatrics 2014;133:e218–26.


21. World Health Organization, Division of Reproductive Health. Thermal protection of the newborn: a practical guide. Geneva (Switzerland): World Health Organization, 1997.
22. Capobianco JA. Keeping the newborn warm: how to safeguard the infant against life-threatening heat loss. Nursing 1980;10:64–7.

23. Thomas K. Thermoregulation in neonates. Neonatal Netw 1994;13:15–22.
24. Wilson E, Maier RF, Norman M, Misselwitz B, Howell EA, Zeitlin J, et al. Admission hypothermia in very preterm infants and neonatal mortality and morbidity. J Pediatr 2016;175:61–7. e4.


26. Vohra S, Roberts RS, Zhang B, Janes M, Schmidt B. Heat Loss Prevention (HeLP) in the delivery room: a randomized controlled trial of polyethylene occlusive skin wrapping in very preterm infants. J Pediatr 2004;145:750–3.


27. Çağlar S, Gözen D, Ince Z. Heat loss prevention (help) after birth in preterm infants using vinyl isolation bag or polyethylene wrap. J Obstet Gynecol Neonatal Nurs 2014;43:216–23.


28. Russo A, McCready M, Torres L, Theuriere C, Venturini S, Spaight M, et al. Reducing hypothermia in preterm infants following delivery. Pediatrics 2014;133:e1055–62.


29. de Almeida MF, Guinsburg R, Sancho GA, Rosa IR, Lamy ZC, Martinez FE, et al. Hypothermia and early neonatal mortality in preterm infants. J Pediatr 2014;164:271–5.


30. Jang JH, Shin SH, Woo HK, Choi EK, Song IG, Shin SH, et al. The association between admission hypothermia and neonatal outcomes in very low birth weight infants. Neonatal Med 2016;23:183–9.

31. Ambalavanan N, Carlo WA. Pulmonary hemorrhage. Kliegman RM, Stanton BF, St. Geme JW, Schor NF, Behrman RE, editors. Nelson textbook of pediatrics. 20th ed. Philadelphia (PA): Elsevier, 2016;:1168





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


