Article Contents
| Korean J Pediatr > Volume 59(3); 2016 |
|
Abstract
We report a case of a 5-year-old girl who developed left hemiparesis and left facial palsy, 6 days after the initiation of fever and respiratory symptoms due to pneumonia. Chest radiography, conducted upon admission, showed pneumonic infiltration and pleural effusion in the left lung field. Brain magnetic resonance imaging showed acute ischemic infarction in the right middle cerebral artery territory. Brain magnetic resonance angiography and transfemoral cerebral angiography revealed complete occlusion of the right middle cerebral artery. Mycoplasma pneumoniae infection was identified by a 4-fold increase in IgG antibodies to M. pneumoniae between acute and convalescent sera by enzyme-linked immunosorbent assay. Fibrinogen and D-dimer levels were elevated, while laboratory exams in order to identify other predisposing factors of pediatric stroke were all negative. This is the first reported pediatric case in English literature of a M. pneumoniae-associated cerebral infarction involving complete occlusion of the right middle cerebral artery.
Extrapulmonary complications of Mycoplasma pneumoniae infection involving the central nervous system (CNS) are not rare. According to previous reports, encephalitis, meningoencephalitis, aseptic meningitis, transverse myelitis, acute disseminated encephalomyelitis, and Guillain-Barré syndrome have been associated with M. pneumoniae infection1,2). Meanwhile, there are less than 20 cases of M. pneumoniae-associated cerebral infraction reported in the pediatric population, and only six cases have identified occlusion of the cerebral arteries3,4,5,6,7,8). We report a case of M. pneumoniae-associated cerebral infraction in a 5-year-old girl in which complete occlusion of the right middle cerebral artery (MCA) was identified by brain magnetic resonance angiography (MRA) and transfemoral cerebral angiography (TFCA).
A 5-year-old girl was admitted due to left hemiparesis and left facial palsy. She had developed symptoms of cough and fever 6 days before. Although she had taken oral antibiotics prescribed at a local clinic, her symptoms did not show improvement. On the fourth day of her illness, she was referred to a secondary hospital and was diagnosed as pneumonia. She was admitted to that hospital and was treated with antibiotics of ceftriaxone and clarithromycin, intravenously. On the sixth day of her illness, her fever subsided. However, she abruptly developed left hemiparesis and left facial palsy, and was referred to Inha University Hospital. Past medical history of the patient and family were unremarkable.
On admission, she was fully conscious and oriented. Vital signs showed a blood pressure of 127/60 mmHg, heart rate of 102 beats/min, respiratory rate of 24 breaths/min, and body temperature of 37.6℃. Her heart beat was regular without murmurs, while breath sounds were coarse with inspiratory crackles auscultated on both lung fields. On neurologic examination, pupils were isocoric with prompt light reflexes. Extraocular movements were full in both eyes, while there were defects in the left visual fields. She was unable to form facial expressions on the lower half of her left face, while she was able to wrinkle her forehead normally, indicating left side central type facial palsy. Motor strengths were decreased to grade III in both the left upper and lower extremities. Sensory was intact, while hemineglect was revealed on the left side. Hyperreflexia was noted on the left biceps and ankle jerks. Pathologic reflexes of Babinski sign and ankle clonus were present on the left.
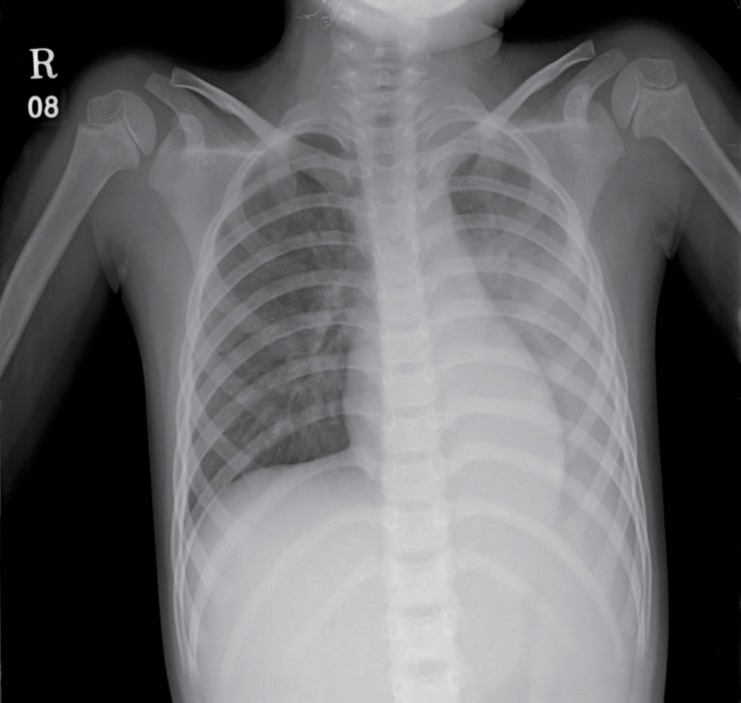
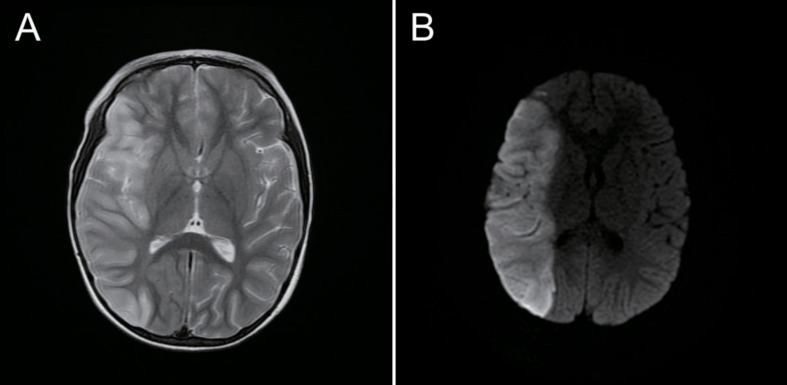
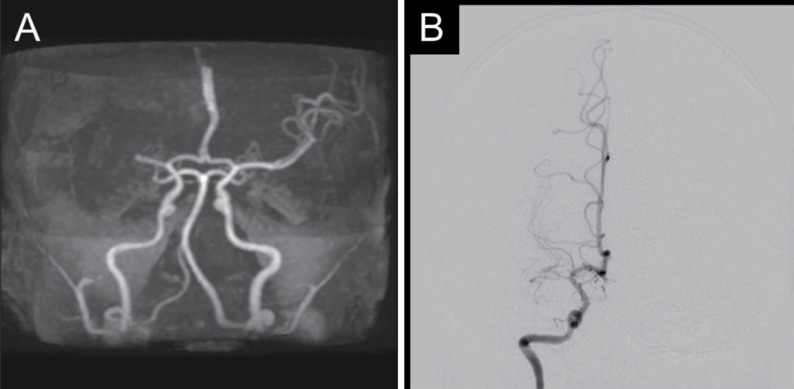
Chest radiography conducted on admission showed pneumonic infiltration and pleural effusion in the left lung field (Fig. 1). Brain magnetic resonance imaging revealed acute ischemic infarction in the right MCA territory (Fig. 2), and brain MRA showed complete occlusion of the right MCA at the M1 portion (Fig. 3A). Transthoracic echocardiography and electrocardiography results were normal. TFCA revealed complete occlusion of the right MCA at the M1 portion, with no evidence of vasculitis or Moyamoya disease (Fig. 3B).
Laboratory exams conducted on the day of admission showed a hemoglobin of 10.5 g/dL, hematocrit 30.1%, white blood cell (WBC) count of 5,420/µL, comprising 82.8% neutrophils, 9.2% lymphocytes, and 4.2% monocytes, and platelet count of 250,000/µL. C-reactive protein was 7.15 mg/dL while other electrolyte and biochemistry laboratory exams were normal. Coagulation studies revealed normal prothrombin and activated partial thromboplastin time, while fibrinogen level was elevated to 674 mg/dL (normal range, 200–400 mg/dL) and D-dimer level to 1.7 µg/mL (normal range, 0–0.4 µg/mL). Analysis of the patient's cerebrospinal fluid (CSF) showed a WBC count of 3/µL, glucose concentration of 90 mg/dL, and protein concentration of 15 mg/dL. Initial serum antibody titers to M. pneumoniae detected by enzyme-linked immunosorbent assays (ELISA) were 5,021 U/mL for IgM (normal range, 0–949 U/mL) and 5.5 AU/mL for IgG (normal range, 0–9.99 AU/mL). Polymerase chain reaction (PCR) of M. pneumoniae, influenza viruses, enteroviruses, and herpes simplex viruses were negative in the CSF.
The patient was diagnosed as pneumonia, strongly suggestive of M. pneumoniae origin associated with acute cerebral infarction. Antibiotics of ceftriaxone and clarithromycin were administered for 14 days, mannitol for 7 days, and methylprednisolone for 3 days, intravenously. Antithrombotic therapy with aspirin at doses of 2 mg/kg was given daily. There was no fever after admission and respiratory symptoms showed gradual improvement. Rehabilitation therapy was initiated on the fifth hospital day. Pneumonic infiltration on chest radiography started to show improvement on the sixth hospital day.
The results of laboratory exams in order to identify possible predisposing causes of the patient's stroke were all negative. Serum analysis of prothrombotic agents and factors including protein C, protein S, antithrombin III, lupus anticoagulant, homocysteine, lipid profiles and plasma lipoprotein levels were normal. Analysis of auto-antibodies associated with autoimmune and rheumatologic diseases, including antiphospholid antibodies, anticardiolipin antibodies, antinuclear antibodies, anticytoplasmic antibodies, anti-DNA antibodies, and rheumatoid factors were all negative. No other bacterial or viral organisms were documented in the serum or CSF on culture studies. Serum antibody titers to M. pneumoniae detected by ELISA 3 weeks after admission were 7,300 U/mL for IgM and 26.65 AU/mL for IgG.
The patient was discharged on the 32nd hospital day after receiving the inpatient rehabilitation schedule. At discharge, both left hemiparesis and left facial palsy had improved, although mild neurologic deficits regarding fine motor skills were observed. Motor strengths of grade V were checked on all extremities and unaided walking was capable.
Although not fully understood, two mechanisms have been proposed in order to explain the pathophysiology of cerebral arterial occlusion associated with M. pneumoniae9,10). The direct-type mechanism is due to the vascular transfer of the organism itself, which locally affect the vascular walls of the cerebral circulation by the induction of cytokines and chemokines such as tumor necrosis factor-α and interleukin-89,10,11). This may cause local vasculitis, thrombotic vascular occlusion, or both, even in the absence of a systemic hypercoagulable state. The indirect-type mechanism is associated with a systemic hypercoagulable state resulting in a generalized thrombotic vascular occlusion, in which the activation of complements are assumed to play a role9,12). This postulation is supported by the elevation of fibrinogen and D-dimer levels reported in some cases of M. pneumoniae-associated cerebral arterial occlusion4,6). In our case, negative M. pneumoniae PCR results of the CSF and laboratory findings of elevated fibrinogen and D-dimer levels indicate that the pathophysiology of cerebral arterial occlusion might be related to the indirect-type mechanism. Thus, a systemic hypercoagulable state induced by M. pneumoniae infection and resultant inflammation may have contributed to the generation of the cerebral arterial occlusion rather than the direct effects of the organism on the affected cerebral artery.
Congenital or acquired heart diseases, sickle cell disease, thrombophilias, and infections are well known etiologies and predisposing factors for acute ischemic cerebral infarction in the pediatric population13). Hence, laboratory studies should include examinations regarding coagulopathies, inflammation, autoimmune diseases, and hyperlipidemia. Predisposing diseases or conditions such as sickle cell trait5), hyperhomocysteinemia8), and antiphospholipid and anticardiolipin antibodies6) have been identified in cases of M. pneumoniae-associated cerebral arterial occlusion in children, besides the organism itself.
Occlusion of cerebral arteries have been documented both in the anterior and posterior circulation in pediatric cases of M. pneumoniae-associated cerebral arterial occlusion, confirmed by conventional angiography or MRA. Internal carotid occlusion has been reported in three cases3,6,7), posterior cerebral artery occlusion in two cases5,8), and MCA occlusion in one case4) among English literature. Our case is the first report of complete arterial occlusion occurring in the right MCA with resultant massive ischemic infarct in its territory which was associated with M. pneumoniae infection in a child.
Treatment of CNS diseases associated with M. pneumoniae infection includes antibiotics, corticosteroids, and intravenous immunoglobulin (IVIG)1). Treatment with steroids and IVIG was reported to improve outcome in a pediatric case of M. pneumoniae-associated stroke14). Low dose aspirin has also been administered in some pediatric cases of M. pneumoniae-associated cerebral arterial occlusion5,8). In general strokes, initial aspirin therapy with doses of 1 to 5 mg/kg is recommended daily followed by long-term aspirin therapy for a minimum of 2 years in children, except those associated with sickle cell disease15). Although there is no standardized treatment, according to our case we assume that the combination of antibiotics, mannitol, corticosteroids, and aspirin may be effective for initial treatment in pediatric cerebral arterial occlusion associated with M. pneumoniae infection. The addition of IVIG may be considered in cases that do not respond well to initial treatment.
In conclusion, we report a case of complete occlusion of the right MCA in a child with M. pneumoniae pneumonia. Thorough evaluation of possible predisposing factors for stroke should always be conducted in children and close attention is required on determining the potential systemic hypercoagulable status associated with M. pneumoniae-associated cerebral arterial occlusion. The administration of corticosteroids and aspirin should also be considered additionally to proper intravenous antibiotics.
Conflicts of interest
Conflict of interest:
No potential conflict of interest relevant to this article was reported.
References
1. Tsiodras S, Kelesidis I, Kelesidis T, Stamboulis E, Giamarellou H. Central nervous system manifestations of Mycoplasma pneumoniae infections. J Infect 2005;51:343–354.


2. Daxboeck F. Mycoplasma pneumoniae central nervous system infections. Curr Opin Neurol 2006;19:374–378.


3. Visudhiphan P, Chiemchanya S, Sirinavin S. Internal carotid artery occlusion associated with Mycoplasma pneumoniae infection. Pediatr Neurol 1992;8:237–239.


4. Fu M, Wong KS, Lam WW, Wong GW. Middle cerebral artery occlusion after recent Mycoplasma pneumoniae infection. J Neurol Sci 1998;157:113–115.


5. Antachopoulos C, Liakopoulou T, Palamidou F, Papathanassiou D, Youroukos S. Posterior cerebral artery occlusion associated with Mycoplasma pneumoniae infection. J Child Neurol 2002;17:55–57.


6. Tanir G, Aydemir C, Yilmaz D, Tuygun N. Internal carotid artery occlusion associated with Mycoplasma pneumoniae infection in a child. Turk J Pediatr 2006;48:166–171.

7. Lee CY, Huang YY, Huang FL, Liu FC, Chen PY. Mycoplasma pneumoniae-associated cerebral infarction in a child. J Trop Pediatr 2009;55:272–275.


8. Ryu JS, Kim HJ, Sung IY, Ko TS. Posterior cerebral artery occlusion after Mycoplasma pneumoniae infection associated with genetic defect of MTHFR C677T. J Child Neurol 2009;24:891–894.


9. Narita M. Pathogenesis of neurologic manifestations of Mycoplasma pneumoniae infection. Pediatr Neurol 2009;41:159–166.


10. Bitnun A, Richardson SE. Mycoplasma pneumoniae: innocent bystander or a true cause of central nervous system disease? Curr Infect Dis Rep 2010;12:282–290.


11. Atkinson TP, Balish MF, Waites KB. Epidemiology, clinical manifestations, pathogenesis and laboratory detection of Mycoplasma pneumoniae infections. FEMS Microbiol Rev 2008;32:956–973.


12. Narita M. Pathogenesis of extrapulmonary manifestations of Mycoplasma pneumoniae infection with special reference to pneumonia. J Infect Chemother 2010;16:162–169.


13. Jordan LC, Hillis AE. Challenges in the diagnosis and treatment of pediatric stroke. Nat Rev Neurol 2011;7:199–208.



Fig. 1
Simple chest radiograph of the patient. Pneumonic infiltration with pleural effusion in the left lung was observed on the first day of admission.



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link PubMed
PubMed Download Citation
Download Citation


