Introduction
Blunt chest trauma may cause cardiac injury in any of the heart chambers or septal or valvular structures1). Ventricular septal defect (VSD), especially when isolated, is very rare event. There have been about 60 cases of traumatic VSD secondary to blunt chest trauma reported since 19701). Traumatic VSD is variable in their clinical presentation, course, and severity, so one can be difficult to diagnose. Echocardiography is a most effective diagnostic tool, although cardiac catheterization is sometimes required for further assessment. Most patients may require surgical or percutaneous closure. We report the case of a 4-year-old boy who presented with a traumatic VSD, following a car accident.
Case report
A previously healthy 4-year-old boy was hit in the anterior trunk by a car wheel. He was soon transferred to a nearby hospital and diagnosed as pneumothorax with contusional pulmonary hemorrhage and hemoperitoneum secondary to liver laceration of segment 7 (S7). Initial chest computed tomography (Fig. 1A) and echocardiography revealed normal cardiac structure without any injury. He was hospitalized in the department of general surgery and was treated for pneumothorax and hemoperitoneum.
After 8 days, his chest radiography showed progressive pleural effusion, and he was noted to have grade IV/VI pansystolic murmur at the left upper sternal border. Echocardiography and chest computed tomography (Fig. 1B) was performed again, and revealed a newly developed large VSD with pericardial effusion.
He was transferred to our hospital for further assessment and surgical repair. On arrival at our hospital, vital signs showed a blood pressure of 80/53 mmHg, a heart rate of 110 beats/minute, a respiratory rate of 30 breaths/minute, and an oxygen saturation of 95% on room air.
Initial laboratory studies showed the following: white blood cell count of 15,100/µL, platelet count of 659×103 µL and hemoglobin level of 12.4 g/dL. The level of cardiac enzymes such as creatinine kinase, creatinine kinase-myocardial fraction, and lactate dehydrogenase were in the normal range.
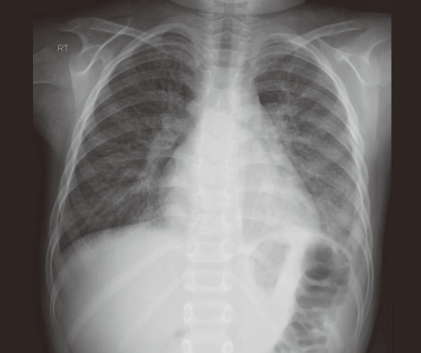
Chest radiography (Fig. 2) showed cardiomegaly and diffuse interstitial thickenings of both lungs, probably secondary to lung contusions. His electrocardiogram (ECG) showed normal sinus rhythm with a rate of 115 beats/min and nonspecific T-wave abnormality.
Echocardiography clearly revealed a VSD with left-to-right shunt flow with minimal pericardial effusion. The VSD was a mid-muscular defect near the apex, with a diameter of about 1.2cm and a pressure gradient of 37 mmHg (Fig. 3). Grade I mitral regurgitation and tricuspid regurgitation were detected, and the aorta appeared normal. The ejection fraction was in the normal range.
His mild congestive heart failure was cared for with diuretics and digitalis. We decided to delay the repair of the VSD until the fibrosis formed around the defect. On the 24th day after the car accident, open repair of the traumatic VSD was performed. An incision was made in the left ventricle, and there was a VSD about 1.5 cm in diameter. The VSD was repaired successfully using double velour. Fibrotic change around the VSD and a thinned posterior ventricular wall were detected, and the left ventricle was plicated with Teflon felt. Postoperative echocardiography showed very small residual VSD shunt and normal ventricular function. His clinical course gradually improved after the operation, and he was discharged on the 10th post-operative day.
About one month after discharge, he was asymptomatic, and a repeat echocardiography showed no residual VSD shunt flow and an ejection fraction of 66%.
Discussion
VSD is mostly congenital in origin, but could be acquired as result of penetrating trauma, blunt trauma, post-surgical contusion, myocardial infarction, or perforation during cardiac catheterization1). Hewett2) first reported rupture of the interventricular septum caused by blunt trauma in 1847, and the first surgical repair was reported in 19553). The most common cause is traffic accidents. Other causes, such as falls from high places or kicks to the chest, have been reported1).
The mechanisms of traumatic VSD occurring from blunt trauma have been of little concern until now, but two dominant theories have been described. The first postulates that the rupture occurs due to acute compression of the heart between the sternum and the vertebrae during the late diastolic phase when the ventricles are filled and the atrioventricular valves are closed4). The second proposes that myocardial injury causes a microvascular disruption, leading to infarction and liquefaction of the septum5). In our case, the patient showed normal cardiac structure at the time of injury, but after 8 days, he was diagnosed as having a VSD, after repeated echocardiography. We can speculate that there was progressive myocardial necrosis secondary to localized interruption of coronary blood flow within the ventricular septum. This progressive myocardial destruction then resulted in a gradual increase in the defect size and left-to-right shunt.
The diagnosis of traumatic VSD might be difficult or delayed due to variable symptoms and the timing of presentation. The timing of traumatic VSD occurring is variable, from immediately after the accident to several years later, according to the mechanism6). Also, clinical symptoms range from mild overall features to fulminant cardiogenic shock6). Besides, traumatic VSD can be masked by concomitant injuries such as intra-abdominal injuries, rib fractures, and fractures of the extremities.
As with congenital VSD, pansystolic murmurs are auscultated at the left sternal border, and chest radiography may be normal or show only mild cardiomegaly or pulmonary edema7). Cardiac enzymes lack the sensitivity or specificity for the diagnosis of cardiac injury, because other skeletal injuries may cause elevations of cardiac enzymes, with or without cardiac injury8). Recently, some authors reported that troponin I and T have proved to be sensitive to cardiac injury and useful in stratifications of patients for risk of complications9).
Transthoracic echocardiography is one of the most effective tools for diagnosing traumatic VSD. It enables the employment of rapid and noninvasive means for evaluating hemodynamically unstable patients at their bedsides10). But for patients whose echocardiography yields poor image quality, due to pneumothorax, wounds, or chest tubes at the site where the transducer should be placed, it is difficult to gain enough information. In these cases, additional cardiac imaging modalities such as CT or MRI can play an important role11).
As with our patient, most traumatic VSD involves the muscular portion of the septum, but cases of perimembranous or subpulmonary rupture have been reported12). In particular, the case of perimembranous rupture requires more attention because it can often be accompanied by tearing of the tricuspid valve13). Other cardiac lesions reported include mitral and aortic valve injury, ventricular aneurysms, pericardial injury, and acute myocardial infarction1).
Closure of traumatic VSD has been based on a combination of heart failure symptoms, hemodynamics, and defect size. A large VSD more than 1 cm in diameter requires surgical repair as soon as possible because congestive heart failure may develop secondary to increased pulmonary arterial pressures7). However, in some patients, such as those presenting a low left-to-right shunt rate and mild congestive heart failure, medical treatment should be attempted first, followed 2-3 weeks later by surgical repair when the myocardial tissue surrounding the VSD shows signs of formation of a fibrous ring14). Small traumatic VSD may close spontaneously without operation14).
Recently, a small number of reports have been published describing effective percutaneous closure of traumatic VSD alternative to surgery11).
We report here a case of a traumatic VSD related to blunt chest injury showing delayed clinical presentation. In traumatic VSD patients, clinical symptoms and signs are relatively variable, and may be masked by concomitant injuries. Therefore, the clinician should focus a high level of attention on chest injury patients. It should be kept in mind that the apparent absence, upon initial evaluation, of myocardial damage cannot exclude its presence; thus, serial cardiac examinations using echocardiography, CT, and/or MRI are strongly recommended.





 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


