Introduction
Kawasaki disease (KD) causes severe vasculitis of all blood vessels but predominantly affects the medium-sized arteries, with a striking predilection for the coronary arteries. Head and neck manifestation of KD includes cervical lymphadenitis, parotitis, otitis media, epiglottitis, peritonsillar abscess, or peritonsillar cellulitis and less commonly retropharyngeal abscess (RPA). There have been several reports in English on KD with RPA since 19941-6). Needle aspirations from these patients established the nonsuppurative nature of the illness. However, there have been no reported cases of KD with confirmed retropharyngeal or parapharyngeal abscesses.
We describe here a three-year-old boy with KD with a coexistent parapharyngeal abscess.
Case report
A previously healthy three-year-old boy presented with a 5-day history of fever (up to 39.5-40Ōäā) and a 1 day history of bilateral conjunctival injection and increasing left neck pain. A physical examination upon admission revealed bilateral conjunctival injection without discharge, fissured red lips, strawberry tongue and tender enlarged left cervical lymph nodes. The largest node, measuring 3├Ś4 cm, was in the left posterior triangle of the neck. Breathing sounds were clear without crackles, and no cardiac murmur was audible. A chest radiograph was unremarkable with a normal appearance of mediastinum and no infiltrates. Initial laboratory results were as follows: erythrocyte sedimentation rate (ESR) 62 mm/h, haemoglobin 12.3 g/dL, white blood cell (WBC) count 16,400/┬ĄL (polymorphonuclear leukocytes, 73%; lymphocytes, 19%; monocytes, 5%), platelet count 597├Ś103/┬ĄL, C-reactive protein (CRP) 5.8 mg/dL, sodium 139 mmol/L, N-terminal fragment of B-type natriuretic peptide (NT-proBNP) 312 pg/mL. Echocardiography revealed perivascular brightness around both coronary arteries and mild mitral regurgitation. The diagnosis of KD was made at this point, and treatment with intravenous immunoglobulin (IVIG) (2 g/kg/day) and aspirin (80 mg/kg/day) was begun.
Consequently, the fever subsided and conjunctival injection disappeared within 24 hours of IVIG infusion, but the painful left neck swelling persisted and severe torticollis developed. On the 3rd hospital day, the fever reappeared with persistent Kawasaki features. The neck lesion, measuring 4├Ś5 cm, showed diffuse erythematous swelling. It was hard, fixed and hot with extreme tenderness. The follow-up of laboratory results were as follows: ESR 84 mm/h, haemoglobin 10.3 g/L, WBC count 13,510/┬ĄL (polymorphonuclear leukocytes, 67%; lymphocytes, 25%; monocytes, 4%), platelets 612├Ś103/┬ĄL, CRP 6.9 mg/dL, sodium 134 mmol/L, and NT-proBNP 404 pg/mL. Initial blood, throat, and urine cultures were unremarkable. Normal peripheral blood smear result was noted. A purified protein derivative (PPD) test as well as serologies for Epstein-Barr virus (EBV), and cytomegalovirus (CMV) were also negative. On the fourth hospital day, IVIG was administered again. The fever subsided and his symptoms (injected bulbar conjunctiva, fissured lips and strawberry tongue) slowly resolved until the 6th hospital day. A repeat echocardiogram on the hospital day 6 showed mild dilation of the left main coronary artery (2.5 mm) with trivial mitral regurgitation. Despite defervescence, the patient complained of painful neck swelling and severe torticollis.
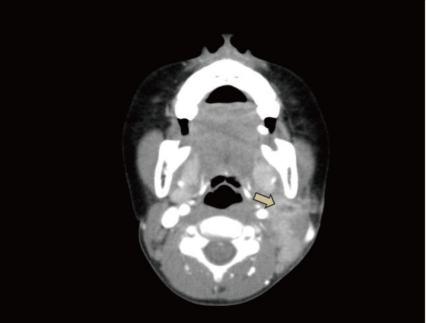
On the 6th hospital day, neck computed tomography (CT) showed a low density lesion with an irregular thick wall in the left lateral node, suggesting an abscess, and multiple lymph node enlargements in the left posterior cervical space (Fig. 1). He was thus diagnosed with a combined parapharyngeal abscess and was treated with antibiotics, including ceftriaxone, until the 9th hospital day. However, there was no improvement of the left cervical lymphadenitis.
Because of the lack of improvement of tender cervical adenopathy despite the use of antibiotics, incision and drainage of the presumed parapharyngeal abscess was planned with the patient under general anesthesia on the 9th hospital day. Purulent fluid collection in parapharyngeal space was drained. Shortly thereafter, the cervical adenopathy decreased in size and his torticollis began to resolve. A culture of the abscess revealed the growth of Staphylococcus aureus which is susceptible to clindamycin, erythromycin, and vancomycin. However, ceftriaxone was not included in the antibiotic susceptibilty test. No coronary artery lesion was noted on repeat echocardiogram. On the 14th hospital day, desquamation of the fingertips was noted. A repeat CT scan on hospital day 16 showed decrease of fluid collection (about 0.5 cm) in the left parapharyngeal space compared with the previous CT scan (Fig. 2). The patient was discharged with low dose aspirin and oral antibiotics on hospital day 16.
Discussion
Lymphadenopathy is the least common diagnostic feature in patients with KD, whereas the other criteria occur in about 90%7). Although KD mimicking a deep neck infection appears to be rare, the true incidence of a parapharyngeal or retropharyngeal pathology is unknown due to the general lack of routine neck imaging. Hence the likely prevalence of unreported cases also remains unknown.
RPAs fall into the group of deep space infections of the neck, which include parapharyngeal abscesses and Ludwig's angina, and multiple abscess types can sometimes coexist8). A Medline search revealed at least six reported cases of KD with an initial diagnosis of a retropharyngeal abscess1-6). Symptoms of all the patients in thoses cases improved dramatically, including their cervical adenopathy after IVIG infusion. In addition, the hypodense core on the initial CT scan which was first interpreted as an abscess revealed a reactive lymphadenopathy on follow up sonogram of neck or on surgical finding. However, in this case, the size of the tender neck swelling increased even after defervescence with the treatment of IVIG. Therefore, the antibiotic therapy was begun on the sixth hospital day lasting until the 16th day of hospitalization. It is noteworthy that no previous report has been issued about the coexistence of these two diseases.
Clinical suspicion should always take precedence, but common sense dictates that the decision to operate should always be made in conjunction with the radiologic evidence, be that ultrasound or CT scan9). Holt et al.10) defined the characteristics of a deep neck abscess as a cystic appearance, low-density CT number, air or fluid at the center of the suspected area, and "rim enhancement". CT scan was quite sensitive (91%) in terms of differentiating abscesses from cellulitis or adenopathy in other reported groups11). However, some studies cast doubts about the complete reliability of CT scans and reported specificity was rather low (60%)12). Several factors may affect the sensitivity and specificity of CT scans. Glasier et al.13) reported that there may be overlapping CT density numbers between cellulitis, adenopathy and abscess, which may lead to false interpretations. On the other hand, inadequate surgical exploration in a patient with a CT scan showing an abscess may result in a false-negative exploration. Their false-positive cases with abscesses seen on CT scan, had undergone needle aspiration only, without formal incision and drainage4,14).
In this case, because of the lack of clinical improvement of cervical adenitis on intravenous antibiotics, the patient underwent an operation on the 9th hospital day for drainage and culture of the parapharyngeal fluid. An abscess cavity could be identified intraoperatively. In other words, the CT scan that showed a parapharyngeal abscess-like lesion was not a false positive finding. This was in contrast to previous reported cases.
Atypical KD is associated with an increased risk of coronary abnormality15). In this case, he did not fulfill all of the diagnostic criteria for KD. High NT-pro BNP concentration and early echocardiogram established the diagnosis of KD16). Later onset of the desquamation was confirmatory. Our child had recrudescent fever after an afebrile period of 24 hours and required additional IVIG retreatment. Recently, Cho et al.17) reported four cases of KD presenting as retropharyngeal abscess. In these cases, all patients eventually fulfilled the diagnostic criteria; one patient was IVIG-resistant and required a second treatment of IVIG and additionally pulse steroid therapy.
The etiology of KD is unclear, but superantigens may play a part18,19). The most recently proposed etiologic candidate is a superantigen, such as toxic shock syndrome toxin-1 (TSST-1) or another bacterial toxin. The idea that a superantigen may be involved in KD was based on reports indicating that certain families of T cell receptor ╬▓ genes (V╬▓2 and V╬▓8) were preferentially expressed in the peripheral blood lymphocytes of patients with acute KD20). However, subsequent research was unable to demonstrate any such association between KD and toxin-producing strains of either S.baureus or streptococci, leading to suggestion that the initial study detected strains of toxin-producing bacteria that were part of the respective patients' normal skin flora21, 22).
The bacterial content of retropharyngeal abscess is often a mixed flora and tends to represent pathological conversion of common upper respiratory tract commensals, but common offending pathogens include Streptococcus viridans, S. aureus and epidermidis, with other gram-positive bacteria and anaerobes also seen. In the present study, the organism cultured from the pus (intra-operatively) was S. aureus. This bacteria is well known for its ability to form abscesses, and most studies recognize S. aureus as the predominant organism causing neck abscess23-25). However, it is unclear whether S. aureus had a role as a superantigen in KD or whether it was just a coincidental finding.
As in this case, failure to improve clinically, as manifested by the presence of a persistently tender lymphadenopathy despite the treatment with an appropriate antibiotic, should prompt a thorough search for the disease progression or an unrecognized coincident cause. In conclusion, if symptoms and signs of suppurative cervical lymphadenopathy in KD patients are not resolving or indeed worsen even after treatment of IVIG, coexistence of a parapharyngeal or retropharyngeal abscess needs to be considered.




 PDF Links
PDF Links PubReader
PubReader PubMed
PubMed Download Citation
Download Citation


