All issues > Volume 69(5); 2026
Association between impaired renal function based on decline in serum creatinine level and brain injury in neonates with hypoxic-ischemic encephalopathy
- Corresponding author: Farid Boubred. APHM, Hospital University La Conception, Neonatal Unit, Service de Médecine néonatale, 147 Boulevard Baille, 13005 Marseille, France Email: farid.boubred@ap-hm.fr
- Received November 9, 2025 Revised February 14, 2026 Accepted March 3, 2026
- Abstract
-
- Background
- Background
- Impaired renal function (IRF), defined as the rate of decline in serum creatinine levels during the week after birth, frequently affects neonates with moderate or severe hypoxic-ischemic encephalopathy (HIE). However, its clinical relevance in this vulnerable population requires further investigation.
- Purpose
- Purpose
- This study aimed to evaluate the association between IRF and brain injury severity in neonates with HIE.
- Methods
- Methods
- This retrospective single-center study included neonates treated with therapeutic hypothermia for moderate or severe HIE. A multivariable logistic regression analysis evaluated the association between IRF and the combined outcomes of early death or severe brain injury (ED/SevereBI).
- Results
- Results
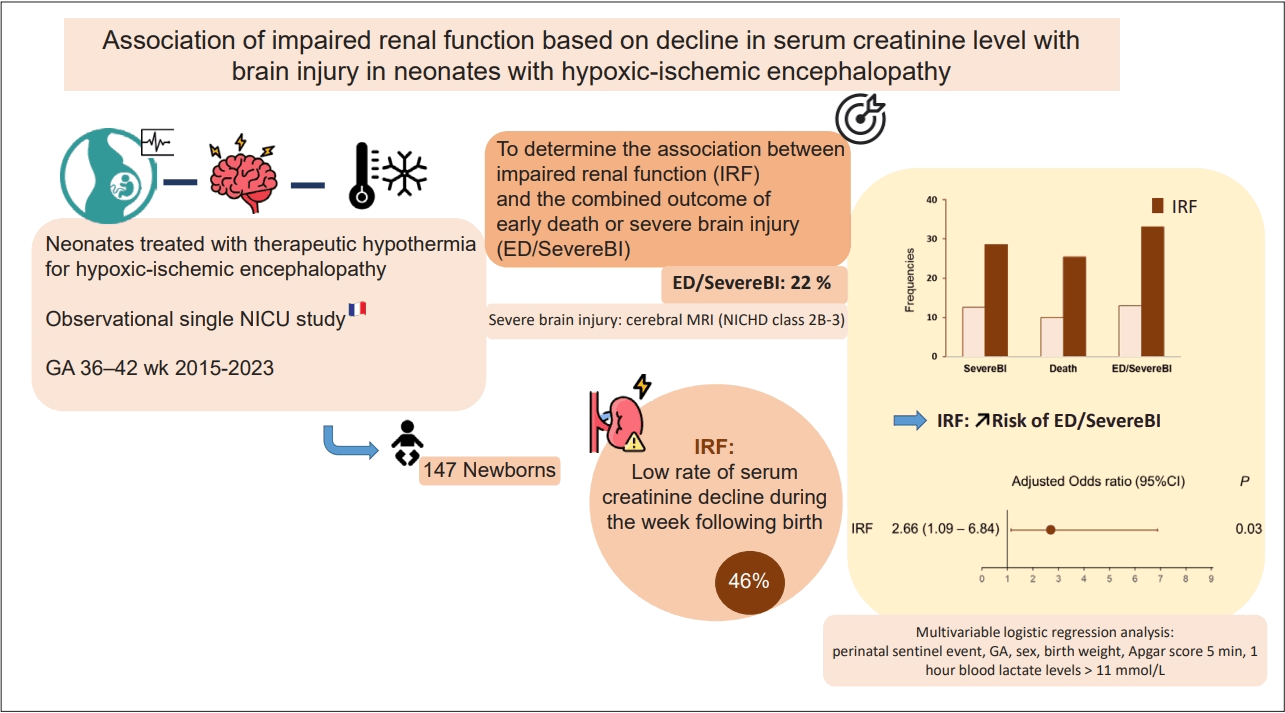
- Of the 147 included neonates, 67 (45.6%) had IRF and 32 (22%) had ED/SevereBI. Those with ED/SevereBI were more likely to have a lower 5-min Apgar score (median [interquartile range]: 4 [2–5] vs. 2 [1–3], P<0.01), have a higher initial blood lactate level (mean cord blood lactate level, +34%, P<0.05), be intubated in the delivery room (50% vs. 75%, P=0.01), and have IRF (39% vs. 69%, P<0.01). After the adjustment for neonatal characteristics and perinatal asphyxia parameters, neonates with IRF had a 2- to 3-fold higher odds of ED/SevereBI than those without IRF (adjusted odds ratio [95% confidence interval]: 2.66 [1.09–6.84], P=0.03).
- Conclusion
- Conclusion
- In neonates treated with therapeutic hypothermia for HIE, IRF can be used as a marker of adverse outcomes. Further studies are required to evaluate its long-term prognostic value.
- Introduction
- Introduction
Neonatal hypoxic-ischemic encephalopathy (HIE) affects 1–8 neonates per 1000 live births in both high- and low-income countries and is a leading cause of death and long-term neurological disabilities in neonates, worldwide [1-4]. Neonatal HIE results from cerebral dysfunction and injury due to hypoxia and hypoperfusion caused by perinatal asphyxia. Treatment options for this condition are limited and are essentially based on therapeutic hypothermia (TH) which has been shown to modestly reduced long-term neurological disabilities [5,6].In addition to impairing the central nervous system, perinatal asphyxia often leads to multiorgan dysfunction, with the kidneys being among the most vulnerable organs affected. Acute kidney injury (AKI) develops in 30%–40% of neonates with moderate or severe HIE and associates with mortality and neurological disorders [7-9]. Its effects on the severity of brain injury are debated [10,11]. Neonatal AKI is mostly defined using the Kidney Disease: Improving Global Outcome (KDIGO) criteria, which include an increase in serum creatinine (SCr) levels (SCr ≥0.3 mg/dL within 48 hours) and low urinary output (oliguria ≤1 mL/kg/day) [12]. However, the AKI-KDIGO definition is not sufficiently sensitive to detect alterations in renal function in the days following birth due to the physiological decrease in SCr levels and the difficulties in rigorously assessing urine output which is often low in the postnatal period. An alternative definition based on the rate of SCr decline during the 7 days after birth has been proposed to identify neonates with impaired renal function (IRF) [13-15]. IRF may reflect haemodynamic disorders and hypovolemia, resulting in renal hypoperfusion, drug-induced nephrotoxicity, and kidney injury. Some studies have demonstrated that the IRF and AKI-KDIGO definitions are equally associated with adverse neonatal outcomes [14,15]. However, the prognostic value of IRF based on the rate of SCr level decline in neonatal morbidity and mortality, especially in neonates with HIE, requires further investigations.This study aimed to investigate the association between IRF and brain injury severity in neonates treated with TH for HIE.
- Methods
- Methods
- 1. Study design
- 1. Study design
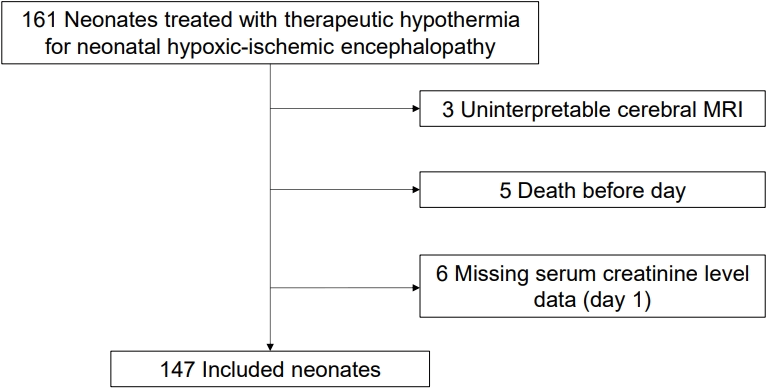
This retrospective, observational study included neonates born with gestational age (GA) ≥36 weeks between 1 January 2015 and 31 December 2023 who required TH for moderate or severe HIE at the Neonatal Intensive Care Unit, Hospital University La Conception, Marseille, France. TH was initiated within the first 6 hours of life, for a duration of 72 hours, according to the French National Guidelines [16]. Parents were informed of the study and they could decline to participate to the study. This study was approved by the research and ethics committee of the French Pediatric Society (CERSFP_23_153-2). In accordance with the design of the study and the French Law, parents were informed and they can decline participation to the study. Written informed consent from the parents was not required. The ethics committee approved a waiver of informed consent.We excluded neonates with congenital anomalies or genetic disorders, those whose parents refused to grant consent to collect and analyse the data, and those with uninterpretable cerebral magnetic resonance imaging (MRI) results. According to the IRF criteria, we also excluded neonates with missing SCr level data on day 1 of life and those who died before day 3, as evaluating the rate of SCr level decline was not possible.- 2. IRF definition
- 2. IRF definition
IRF was defined based on the rate of SCr decline during the first week of life. Neonates with a rate of SCr level decline ≤33%, ≤40%, or ≤46% on days 3, 5, or 7, respectively, were considered to have IRF 13.- 3. Principal outcome
- 3. Principal outcome
The principal outcome was defined as the composite outcome of early death (ED) before performing the cerebral MRI, or severe brain injury (SevereBI) on cerebral MRI (ED/SevereBI). The cerebral MRI protocol included 3-dimensional conventional T1- and T2-weighted sequences, diffusion-weighted sequences, and magnetic resonance spectrometry (3T MRI and Siemens software; Symphony Maestro Class, Siemens, Germany). The brain injuries were classified using the National Institute of Child Health and Human Development classification [17,18]. Severe brain injury was defined as bilateral basal ganglia and thalamic injuries (with internal capsule involvement), watershed infarction with additional cerebral involvement (stage 2B), or diffuse cerebral necrosis (stage 3).- 4. Statistical analysis
- 4. Statistical analysis
Data were collected from the neonates’ medical records and included obstetrical data, mode of delivery, perinatal sentinel events (fetal dystocia, uterine rupture, placental abruption, clinical chorioamnionitis, fetal hemorrhagic anemia, and umbilical cord anomalies), anthropometric neonatal characteristics (GA, birth weight, sex), and neonatal complications (duration of mechanical ventilation, cerebral MRI findings, late-onset sepsis, and death). Small for GA was defined according to sex and GA using the Fenton growth charts [19]. Parameters of perinatal asphyxia, including cord blood pH and lactate, blood pH and lactate levels within the first hour of life, Apgar score (1, 5, and 10 minutes after birth), and cardiopulmonary resuscitation at birth (chest compression, need for tracheal intubation, and adrenalin administration) were collected.We used univariate statistical tests using the chi-square (or Fischer exact test) and Student t-tests (or Mann-Whitney test), as appropriate, to compare groups of neonates with and without ED/SevereBI for categorical and quantitative variables, respectively. An additional multivariable logistic regression analysis was performed to assess the association between IRF and the composite outcome of ED/SevereBI. The model was adjusted for GA, sex, birth weight z score, Apgar score at 5 minutes, blood lactate levels within the 1st hour and perinatal sentinel events (R-Software). A P value <0.05 was considered statistically significant.
- Results
- Results
During the study period, 161 neonates were treated with TH for moderate or severe HIE. Fourteen neonates were excluded because of missing SCr level data (n=6), death before the third day of life (n=5), or not interpretable cerebral MRI (n=3) (Fig. 1). Among the 147 included neonates, 4 (2.7%) died early without cerebral MRI assessment (ED) and 28 (19%) had severe brain injury (SevereBI).During the study period, 67 neonates (46%) had IRF and 22 of them (32.8%) had ED/SevereBI. In the subset of neonates meeting the KDIGO-AKI criteria (n=15), the composite outcome of ED/SevereBI was observed in 8 neonates (53.3%). But all of them were also classified as having IRF.Compared to neonates without severe brain injury, those with ED/SevereBI were more likely to have lower Apgar scores (P<0.01), higher blood lactate levels at birth (P=0.02) and within the first hour after birth (+34%, P<0.001), and to require delivery room intubation (50% vs. 75%, P=0.01) (shown in Table 1). GA, sex, birth weight z score, mode of delivery, and perinatal sentinel events did not differ between the 2 groups of neonates. Nevertheless, the rate of IRF was higher in neonates with ED/SevereBI (39% vs. 69%, P<0.01). Fig. 2 illustrates the evolution of SCr in the 2 groups during the week following birth.Neonates with IRF had more frequent severe brain injury on MRI (28.6% vs. 12.5%, P=0.01) compared to neonates without IRF. The mortality rate was also higher (25.4% vs. 10%, P=0.01). Characteristics of neonates with IRF are shown in Supplementary Table 1 and Supplementary Fig. 1.The multivariable logistic regression analysis showed that neonates with IRF had 2- to 3-fold higher odds of ED/SevereBI than their counterparts (adjusted odds ratio [aOR] (95% confidence interval [CI]): 2.66 [1.09-6.84]; P=0.03).
- Discussion
- Discussion
In this study of neonates treated with TH for HIE, those who had IRF defined by a low rate of SCr level decline during the first week of life were at a high risk of ED/SevereBI.This definition has been proposed as a surrogate marker of IRF in neonates [13,14]. The rate of SCr level decline considers the maturational process of the glomerular filtration rate (GFR) and the high susceptibility of the immature kidney to an adverse environment [20,21]. Following birth, there is a rapid increase in GFR that results in an exponential decrease in SCr levels, by more than 50% during the first week of life [13]. However, GFR is low, making newborns highly susceptible to various factors, including hemodynamic and oxygenation changes, nephrotoxic drug exposure, and fluid depletion. The neonatal AKI-KDIGO definition which is based on the rise in the SCr level (≥0.3 mg/dL within any 48-hour period) and/or low urine output (<1 mL/kg/hr), does not integrate this evolution and can underestimate newborns with mild kidney injury [14]. In addition, urine output is usually low in the first few days after birth, and oliguria is not always observed in infants with AKI. In a population of 106 neonates treated with TH for HIE, Gupta et al. [13] showed that neonates diagnosed with IRF based on SCr level decline or with the neonatal AKI-KDIGO definition have similar adverse outcomes, including high use of vasopressors, prolonged mechanical ventilation, fluid overload, longer hospital stay, high blood gentamycin levels, and high urinary markers of kidney injury compared to control newborns. In summary, during the postnatal period, the rate of SCr level decline may be considered a reliable marker of renal injury, with greater sensitivity than that of the neonatal AKI-KDIGO criteria.The prognostic significance of IRF, as indicated by a low rate of SCr level decline during the first week after birth, in mortality and brain injury severity in newborns with HIE, has not yet been thoroughly investigated. In this study, we found an association between IRF and the composite outcome of ED/SevereBI (aOR [95% CI]: 2.66 [1.09–6.84]; P=0.03). Similar findings have been observed in a recent retrospective study of neonates treated with TH for HIE in a neonatal intensive care unit in the USA. The authors found that neonates with IRF based on SCr level decline had increased odds of the same composite outcome compared to the control group (aOR [95% CI]: 2.72 [1.18–6.26]; P=0.02) [15]. This study also showed that this definition captured more neonates with early-stage AKI than the KDIGO criteria: 51% of neonates had IFR (SCr level decline definition) versus 28.4% using the neonatal AKI-KDIGO criteria. The mechanism by which IRF associates with severe brain injury is unknown and complex. IRF can be considered a marker of the severity of perinatal asphyxia with renal injury due to a profound decrease in fetal systemic blood pressure and renal blood flow [22,23]. IRF may also act as a contributing factor for brain injury through diverse pathways. IRF induces systemic inflammation by stimulating proinflammatory cytokines release and reducing their clearance [24,25]. Metabolic changes associated with renal injury including electrolyte disturbances (hyponatremia), the accumulation of inorganic molecules and reduced drug clearance, in conjunction with a proinflammatory state, can affect the integrity of the blood brain barrier. These changes may perpetuate brain injury through enhancing cerebral edema, oxidative stress, microglial activation and accumulation of inflammatory cells, excitatory metabolism and endothelial dysfunction [26,27]. These findings, including those of the present study, indicate that IRF, based on the rate of SCr decline during the first week of life, could be considered a reliable marker of adverse neonatal outcomes in neonates with HIE.Our study had some limitations. This was a retrospective, single-center study, and potential confounders were not considered. GFR measurements using creatinine clearance, urine output, fluid intake and electrolyte balance were lacking. Electrolyte imbalance, particularly hyponatremia, should also be considered; given its potential link to IRF. Long-term neurological and kidney outcomes were not collected; also, we could not evaluate the long-term consequences of IRF on neurodevelopment and kidney function [9,28,29].Although the period of inclusion extended from 2015 to 2023, no substantial changes in healthcare management occurred during this period that might have significantly affected the incidence of IRF. In addition, the contemporary characteristics of the studied population managed with current recommendations strengthen the relevance of our findings. We believe that the results of this study may help clinicians identify newborns at high risk of neonatal morbidity and rapidly adapt clinical practices, including fluid therapy, electrolyte balance, and therapeutic options, all of which can improve neonatal healthcare.In conclusion, in this study of neonates treated with TH for moderate or severe HIE, IRF based on SCr level decline was significantly associated with adverse neonatal outcomes. These findings underscore the clinical relevance of IRF during the days following birth. Further studies are required to investigate the long-term neurological and renal consequences of IRF.
Supplementary materials
Supplementary materials
Supplementary Table 1 and Supplementary Fig. 1 are available at https://doi.org/10.3345/cep.2025.02670.Supplementary Table 1.
cep-2025-02670-Supplementary-Table-1.pdfCharacteristics of neonates with impaired renal function (IRF)Supplementary Fig.1.
cep-2025-02670-Supplementary-Fig-1.pdfSerum creatinine levels during the week after birth in neonates with impaired renal function. Neonates with impaired renal function are in black square. The serum creatinine values are expressed as the mean with the corresponding standard deviation. **P<0.001.
- Footnotes
-
Conflicts of interest No potential conflict of interest relevant to this article was reported.
Funding This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contribution Conceptualization: FB, IG; Data curation: MBoget, WR, JF; Formal analysis: MBoget, WR, DG, MBoucekine, IG; Methodology: FB, JF, DG, MBoucekine, IG; Visualization: FB, MBoget, WR, JF, DG, MBoucekine, IG; Writing - original draft: FB, MBoget, WR, JF; Writing - review & editing: FB, MB, WR, JF, DG, MBoucekine, IG
-
Fig. 2.
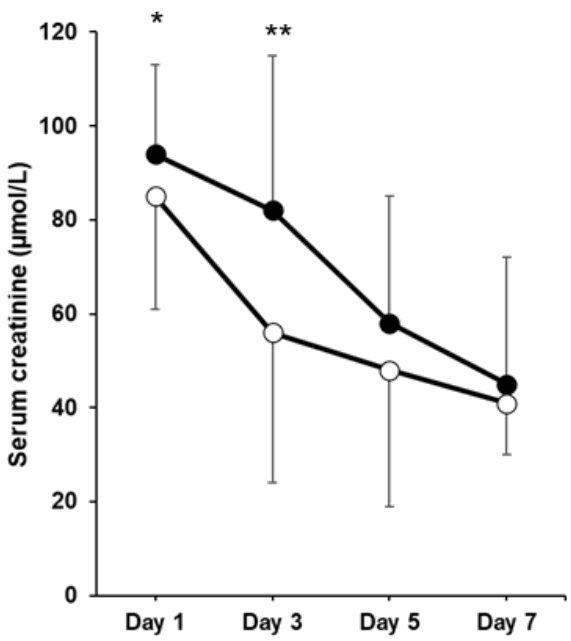
Table 1.
Values are presented as number (%), mean±standard deviation, or median (interquartile range).
ED/SevereBI, early death or severe brain injury; GA, gestational age; BW, birth weight; SGA, small for gestational age; DR, delivery room; IMV, invasive mechanical ventilation; IRF, impaired renal function.
Boldface indicates a statistically significant difference with P<0.05.
- References
- 1. Douglas-Escobar M, Weiss MD. Hypoxic-ischemic encephalopathy: a review for the clinician. JAMA Pediatr 2015;169:397–403.
[Article] [PubMed] [PMC]2. Pappas A, Milano G, Chalak LF. Hypoxic-ischemic encephalopathy: changing outcomes across the spectrum. Clin Perinatol 2023;50:31–52.
[PubMed]3. Acun C, Karnati S, Padiyar S, Puthuraya S, Aly H, Mohamed M. Trends of neonatal hypoxic-ischemic encephalopathy prevalence and associated risk factors in the United States, 2010 to 2018. Am J Obstet Gynecol 2022;227:751.e1–751.e10.
[Article] [PubMed]4. Beck J, Bednarek N, Pierrat V, Vilotitch A, Loron G, Alison M, et al. Cerebral injuries in neonatal encephalopathy treated with hypothermia: French LyTONEPAL cohort. Pediatr Res 2022;92:880–7.
[Article] [PubMed]5. Shankaran S, Laptook AR, Ehrenkranz RA, Tyson JE, McDonald SA, Donovan EF, et al. Whole-body hypothermia for neonates with hypoxic-ischemic encephalopathy. N Engl J Med 2005;353:1574–84.
[Article] [PubMed] [PMC]6. Jacobs S, Hunt R, Tarnow-Mordi W, Inder T, Davis P. Cooling for newborns with hypoxic ischaemic encephalopathy. Cochrane Database Syst Rev 2003;(4):CD003311.
[Article] [PubMed] [PMC]7. Jetton JG, Boohaker LJ, Sethi SK, Wazir S, Rohatgi S, Soranno DE, et al. Incidence and outcomes of neonatal acute kidney injury (AWAKEN): a multicentre, multinational, observational cohort study. Lancet Child Adolesc Health 2017;1:184–94.
[PubMed] [PMC]8. Selewski DT, Jordan BK, Askenazi DJ, Dechert RE, Sarkar S. Acute kidney injury in asphyxiated newborns treated with therapeutic hypothermia. J Pediatr 2013;162:725–9.e1.
[Article] [PubMed]9. Cavallin F, Rubin G, Vidal E, Cainelli E, Bonadies L, Suppiej A, et al. Prognostic role of acute kidney injury on long-term outcome in infants with hypoxic-ischemic encephalopathy. Pediatr Nephrol 2020;35:477–83.
[Article] [PubMed]10. Sarkar S, Askenazi DJ, Jordan BK, Bhagat I, Bapuraj JR, Dechert RE, et al. Relationship between acute kidney injury and brain MRI findings in asphyxiated newborns after therapeutic hypothermia. Pediatr Res 2014;75:431–5.
[Article] [PubMed]11. Elgendy MM, Cortez J, Saker F, Acun C, Matar RB, Mohamed MA, et al. Acute kidney injury in infants with hypoxicischemic encephalopathy. Pediatr Nephrol 2024;39:1271–7.
[Article] [PubMed]12. Zappitelli M, Ambalavanan N, Askenazi DJ, Moxey-Mims MM, Kimmel PL, Star RA, et al. Developing a neonatal acute kidney injury research definition: a report from the NIDDK neonatal AKI workshop. Pediatr Res 2017;82:569–73.
[Article] [PubMed] [PMC]13. Gupta C, Massaro AN, Ray PE. A new approach to define acute kidney injury in term newborns with hypoxic ischemic encephalopathy. Pediatr Nephrol 2016;31:1167–78.
[Article] [PubMed] [PMC]14. Perazzo S, Revenis M, Massaro A, Short BL, Ray PE. A new approach to recognize neonatal impaired kidney function. Kidney Int Rep 2020;5:2301–12.
[Article] [PubMed] [PMC]15. Ahn HC, Frymoyer A, Boothroyd DB, Bonifacio S, Sutherland SM, Chock VY. Acute kidney injury in neonates with hypoxic ischemic encephalopathy based on serum creatinine decline compared to KDIGO criteria. Pediatr Nephrol 2024;39:2789–96.
[Article] [PubMed]16. Saliba E, Norbert K, Cantagrel S. Neuroprotection par hypothermie de l’encéphalopathie hypoxique-ischémique du nouveau-né à terme. Réanimation 2010;19:655–64.
[Article]17. Wisnowski JL, Wintermark P, Bonifacio SL, Smyser CD, Barkovich AJ, Edwards AD, et al. Neuroimaging in the term newborn with neonatal encephalopathy. Semin Fetal Neonatal Med 2021;26:101304.
[Article] [PubMed] [PMC]18. Shankaran S, Barnes PD, Hintz SR, Laptook AR, Zaterka-Baxter KM, McDonald SA, et al. Brain injury following trial of hypothermia for neonatal hypoxic-ischaemic encephalopathy. Arch Dis Child Fetal Neonatal Ed 2012;97:F398–404.
[Article] [PubMed] [PMC]19. PediTools. Growth Fenton 2013. Available from: https://peditools.org/fenton2013/.20. Saint-Faust M, Boubred F, Simeoni U. Renal development and neonatal adaptation. Am J Perinatol 2014;31:773–80.
[Article] [PubMed]21. Iacobelli S, Guignard JP. Maturation of glomerular filtration rate in neonates and infants: an overview. Pediatr Nephrol 2021;36:1439–46.
[Article] [PubMed]22. Badurdeen S, Roberts C, Blank D, Miller S, Stojanovska V, Davis P, et al. Haemodynamic instability and brain injury in neonates exposed to hypoxia⁻ischaemia. Brain Sci 2019;9:49.
[Article] [PubMed] [PMC]23. Turner MJ, Rumpel JA, Spray BJ, Stence N, Neuberger I, Frymoyer A, et al. Urine biomarkers of acute kidney injury and association with brain MRI abnormalities in neonatal hypoxic-ischemic encephalopathy. J Perinatol 2024;44:1203–7.
[Article] [PubMed]24. Chen J, Hartono JR, John R, Bennett M, Zhou XJ, Wang Y, et al. Early interleukin 6 production by leukocytes during ischemic acute kidney injury is regulated by TLR4. Kidney Int 2011;80:504–15.
[Article] [PubMed] [PMC]25. Liu M, Liang Y, Chigurupati S, Lathia JD, Pletnikov M, Sun Z, et al. Acute kidney injury leads to inflammation and functional changes in the brain. J Am Soc Nephrol 2008;19:1360–70.
[Article] [PubMed] [PMC]26. Husain-Syed F, Takeuchi T, Neyra JA, Ramírez-Guerrero G, Rosner MH, Ronco C, et al. Acute kidney injury in neurocritical care. Crit Care 2023;27:341.
[Article] [PubMed] [PMC]27. Lu R, Kiernan MC, Murray A, Rosner MH, Ronco C. Kidneybrain crosstalk in the acute and chronic setting. Nat Rev Nephrol 2015;11:707–19.
[Article] [PubMed]

 About
About Browse articles
Browse articles For contributors
For contributors