All issues > Volume 69(5); 2026
Sarcopenia in pediatric gastroenterology and hepatology: an updated review
- Corresponding author: Toshifumi Yodoshi, MD PhD. Division of Gastroenterology, Hepatology and Nutrition, Cincinnati Children’s Hospital Medical Center, 3333 Burnet Ave, Cincinnati, OH 45229, USA Email: Toshifumi.Yodoshi@cchmc.org
- Received February 10, 2026 Revised March 26, 2026 Accepted March 26, 2026
- Abstract
-
Sarcopenia—the pathologic loss of skeletal muscle mass and strength—is increasingly recognized in pediatric gastroenterology and hepatology as an important determinant of clinical outcomes. Although historically linked to aging, secondary sarcopenia in children arises from chronic inflammation, malnutrition, physical inactivity, corticosteroid exposure, endocrine disturbances, and anabolic resistance. Unlike adult medicine, where diagnostic frameworks are more established, pediatric definitions remain heterogeneous because growth and puberty substantially influence body composition and muscle function. Diagnosis therefore relies on size-adjusted muscle indices, usually normalized to height squared, interpreted against age- and sex-specific reference curves. Body mass index alone is insufficient because muscle depletion and abnormal fat distribution may be present despite normal or elevated body weight, particularly in children with sarcopenic obesity and metabolic dysfunction-associated steatotic liver disease. Accumulating evidence suggests that pediatric sarcopenia is not simply a marker of frailty but a clinically meaningful predictor of adverse outcomes. In pediatric liver transplantation and cirrhosis, sarcopenia is associated with higher waitlist mortality, longer intensive care stays, and more posttransplant infections. In pediatric inflammatory bowel disease, reduced muscle mass correlates with aggressive disease, earlier biologic escalation, and increased surgical risk. This review summarizes current evidence on the epidemiology, pathophysiology, diagnosis, and management of pediatric sarcopenia in gastrointestinal and liver diseases. We discuss available diagnostic tools, including computed tomography/magnetic resonance imaging, dual-energy x-ray absorptiometry, bioelectrical impedance analysis, and grip strength, highlighting their practical advantages and limitations. We also propose a pragmatic diagnostic algoririthm and outline management strategies that extend beyond caloric supplementation, emphasizing adequate protein intake, resistance exercise, and optimization of underlying disease. Early recognition of sarcopenia may improve risk stratification, functional outcomes, and long-term prognosis in children with chronic gastrointestinal and liver diseases.
- Introduction
- Introduction
Sarcopenia is defined as a pathologic deficit in skeletal muscle mass and often strength or function. First introduced by Rosenberg in 1989 to describe age-related muscle loss [1], the concept of sarcopenia has since evolved to encompass muscle wasting in a variety of settings, including chronic diseases in younger populations [2]. In adults, consensus definitions by the European Working Group on Sarcopenia in Older People (EWGSOP) and the Asian Working Group for Sarcopenia (AWGS) are well-established, utilizing standardized cutoffs for muscle strength and quantity [3-6]. The AWGS specifically provides cutoffs tailored to Asian body composition and anthropometry, highlighting the importance of population- specific references [5,6].In pediatrics, however, there is no universally accepted definition or diagnostic cutoff for sarcopenia [7]. Children are in a dynamic state of growth, making absolute measurements difficult to interpret. Consequently, pediatric assessment requires normalizing muscle mass for body size—typically by height squared (height2)—to generate indices comparable to adult appendicular skeletal muscle mass indices [7]. While pediatric reference curves for muscle size (e.g., total psoas muscle area, tPMA) have been generated [2], diagnostic thresholds remain inconsistent [7]. Studies utilize varying z scores, percentiles (commonly ≤5th percentile for age/sex) [7,8], or study-specific controls. A recent scoping review highlighted this heterogeneity, underscoring the urgent need for a unified pediatric consensus [7].Compounding this diagnostic challenge is the interplay between sarcopenia, malnutrition, and frailty. While sarcopenia often contributes to the physical phenotype of frailty and overlaps with malnutrition, they are distinct entities [9]. Crucially, traditional nutritional assessments like body mass index (BMI) often fail to detect muscle wasting, particularly in "sarcopenic obesity." Recent shifts in obesity nosology further emphasize that BMI alone cannot define individual adiposity burden or obesity-related illness; rather, body composition, fat distribution, and obesity-associated functional consequences should also be considered. This perspective is particularly relevant in pediatric gastrointestinal (GI)/hepatology, where a child may have normal or elevated BMI yet still harbor clinically meaningful muscle depletion [10]. A recent study in pediatric autoimmune liver disease revealed that while only 3% of patients met BMI-based malnutrition criteria, 48% had sarcopenia [11]. This discrepancy emphasizes that reliance on weight-based metrics alone risks underestimating nutritional risk [12,13].Recognizing sarcopenia in pediatric gastroenterology and hepatology is critical as it is a robust predictor of adverse clinical outcomes. In adults, sarcopenia independently predicts mortality, infection risk, and poor transplant outcomes, distinct from liver severity scores like model for end-stage liver disease (ESLD) [14]. Emerging pediatric data suggest similar risks in inflammatory bowel disease (IBD) [15,16], liver cirrhosis [8,17], and metabolic dysfunction-associated steatotic liver disease (MASLD) [18,19]. This narrative review aims to provide a comprehensive synthesis for clinicians by exploring: (1) the epidemiology and prognostic significance in pediatric GI and liver diseases, (2) pathophysiological mechanisms driving muscle loss, (3) diagnostic modalities including imaging and functional measures, and (4) practical management strategies involving nutritional and exercise interventions. Improving the recognition and management of sarcopenia is essential to optimize growth, functional outcomes, and survival in this vulnerable population.
- Definition and concepts of pediatric sarcopenia
- Definition and concepts of pediatric sarcopenia
- 1. Pediatric sarcopenia vs. adult sarcopenia
- 1. Pediatric sarcopenia vs. adult sarcopenia
The conceptual framework of sarcopenia originated in geriatric medicine, where it is defined by the age-related loss of muscle mass and function. In adults, consensus guidelines such as those from the EWGSOP and AWGS utilize fixed, absolute cutoff values for diagnosis [3-6]. For example, EWGSOP2 thresholds include grip strength <27 kg for men and <16 kg for women, and appendicular lean mass (ALM) index <7.0 and <5.5 kg/m2, respectively, in European populations [3]. The AWGS guidelines use similar parameters with slightly different cutoffs (e.g. grip <26 kg in men, <18 kg in women) appropriate for Asian body composition, and they emphasize walking speed or chair stand performance as additional criteria [5,6].Applying these adult definitions to pediatrics, however, is inappropriate due to the dynamic nature of childhood growth. In children, muscle mass and strength naturally increase with age and somatic development [20]. Therefore, a single absolute threshold cannot differentiate a pathological deficit from a developmental stage. Instead, pediatric sarcopenia must be defined relative to a healthy reference population. Current approaches rely on z scores or percentiles normalized for age, sex, and height. This need for reference-based interpretation is reinforced by the developmental biology of body composition: birth weight and subsequent growth trajectories influence later lean and fat mass, while muscle-related parameters rise nonlinearly across childhood and puberty, with more pronounced pubertal gains in boys [21,22]. A commonly cited definition for low muscle mass is a value <5th percentile or a z score < -2 relative to a healthy reference population [7].A critical divergence from adult criteria lies in the assessment of muscle function. While adult diagnoses mandate the presence of both low muscle mass and low strength, functional testing in pediatrics is limited by developmental feasibility. Handgrip dynamometry is reproducible in children aged 5–6 years and older using appropriate devices [23]. However, in infants and toddlers—a population frequently requiring liver transplantation—direct strength measures are impossible. Clinicians must resort to developmental motor scales (e.g., Alberta Infant Motor Scale [AIMS]) to infer functional impairment [24]. Consequently, the majority of pediatric studies currently rely on muscle mass quantification (via imaging or dual-energy x-ray absorptiometry [DXA]) as the primary diagnostic criterion [7], although the integration of functional metrics remains the gold standard for older children.- 2. Sarcopenic obesity
- 2. Sarcopenic obesity
A particularly relevant concept in pediatric GI/hepatology is sarcopenic obesity, defined as the coexistence of high adiposity with low muscle mass/quality [19,23,24]. This condition is “two-hit,” combining risks from obesity (e.g., metabolic and mechanical complications) and sarcopenia (frailty and functional impairment) [24]. In adults, sarcopenic obesity confers synergistically worse outcomes than either condition alone [25]. In children, obesity has become common, and notably MASLD—now the most prevalent pediatric chronic liver disease—often affects children with overweight/obesity [26]. Sarcopenic obesity in pediatrics is an emerging area of study, and there is no consensus pediatric definition yet [23]. Sarcopenic obesity is of special concern because excess fat may mask the muscle deficiency if one only looks at weight or BMI [23]. In parallel, the recent concept of “clinical obesity” highlights that obesity should not be interpreted by BMI alone, but in the context of confirmed adiposity, fat distribution, and obesity-related tissue or organ dysfunction. Within this framework, sarcopenic obesity represents a particularly high-risk phenotype because excess adiposity and muscle depletion may coexist while remaining obscured by body weight alone [10]. Children with sarcopenic obesity might appear “well-nourished” or even overweight by BMI, yet have diminished strength and metabolic issues. Indeed, low relative muscle mass in obese youth correlates with worse insulin resistance and metabolic syndrome features [27]. In pediatric MASLD, children with lower muscle mass indices have more severe liver steatosis and inflammation – suggesting that sarcopenic obesity could exacerbate liver disease progression [19]. Overall, recognizing sarcopenic obesity requires clinicians to assess body composition (not just BMI) in children with overweight, as this subgroup may benefit from targeted exercise and nutrition interventions to improve muscle mass while managing weight.- 3. Distinguishing sarcopenia, malnutrition, and frailty
- 3. Distinguishing sarcopenia, malnutrition, and frailty
In clinical practice, the terms sarcopenia, malnutrition, and frailty are often used interchangeably, yet they represent distinct pathophysiological entities with varying overlaps (Fig. 1).• Malnutrition is traditionally defined by deficits in dietary intake or uptake resulting in measurable anthropometric changes, such as low weight-for-height or BMI [28]. While chronic malnutrition often causes sarcopenia due to the catabolism of muscle protein, the two are not synonymous.• Frailty is a multidimensional syndrome of reduced physiologic reserve and increased vulnerability to stressors [29]. Sarcopenia is a key biological driver of physical frailty, but frailty also encompasses cardiovascular, immunological, and psychosocial deficits [30].These conditions frequently coexist in children with chronic GI or liver diseases [31]. However, their dissociation is clinically critical. In advanced chronic liver disease, while virtually all frail patients are sarcopenic, sarcopenia can exist without overt frailty, serving as an early warning sign of physiologic decline [8,31,32]. Crucially, traditional nutritional markers often fail to detect this decline. A study in pediatric autoimmune liver disease revealed a striking discordance: while only 3% of patients met BMI-based malnutrition criteria, 48% were sarcopenic [11]. This underscores that reliance on weight-based metrics risks underestimating nutritional risk [33]. Consequently, clinicians should assess nutritional status, muscle mass, and functional status separately to accurately capture the child’s risk profile [34,35].- 4. Epidemiology of sarcopenia in pediatric gastroenterology and hepatology
- 4. Epidemiology of sarcopenia in pediatric gastroenterology and hepatology
The recognition of muscle wasting as a critical comorbidity in pediatric gastroenterology has lagged behind adult medicine. While sarcopenia is firmly established as a predictor of mortality in adult cirrhosis—with prevalence rates ranging from 30% to 60% [36-38]—pediatric data have historically been sparse. However, accumulating evidence now demonstrates that sarcopenia is highly prevalent across a spectrum of pediatric GI and liver diseases, affecting approximately one-quarter to one-half of patients depending on the population and diagnostic criteria used [11,13,15,19,39] Crucially, this deficit is often "hidden" in children who exhibit normal weight or obesity, and it serves as a potent, independent predictor of adverse clinical outcomes.- 5. Prevalence and prognostic impact in chronic liver disease
- 5. Prevalence and prognostic impact in chronic liver disease
Muscle wasting is ubiquitous in children with ESLD. A recent review by Chen et al. [8] summarized that across various cohorts of children with chronic liver disease or those awaiting transplantation, 24% to 70% met criteria for sarcopenia. The wide variability reflects differences in measurement techniques (computed tomography [CT], magnetic resonance imaging [MRI], DXA) and the heterogeneity of study populations, which range from infants with biliary atresia to adolescents with autoimmune conditions [11,39].- 6. Pediatric liver transplant candidates
- 6. Pediatric liver transplant candidates
Cross-sectional imaging consistently reveals significant muscle deficits in children awaiting liver transplantation. Mangus et al. [40] found height-indexed psoas muscle area 23% lower than controls, and Lurz et al. [41] observed markedly reduced tPMA. Crucially, muscle mass does not correlate with Pediatric End-stage Liver Disease (PELD) scores [42]. Because PELD relies solely on biochemical markers, it fails to capture nutritional deterioration, meaning children with "low-risk" scores may harbor unrecognized frailty. Pretransplant sarcopenia is prevalent and clinically consequential. Woolfson et al. identified sarcopenia (tPMA z score < -2) in 40% of candidates [17], which correlated with concurrent malnutrition and longer posttransplant intensive care unit (ICU) stays. Furthermore, Jitwongwai et al. [43] observed that lower psoas index tracked with higher waitlist mortality in a cohort dominated by infants with biliary atresia, underscoring the severe burden of muscle loss in this population.- 7. Sarcopenia in infants with biliary Atresia
- 7. Sarcopenia in infants with biliary Atresia
Infants with biliary atresia are uniquely vulnerable to malnutrition due to cholestasis and hypermetabolism. Takeda et al. [39] identified sarcopenia (tPMA z score < -2) in 24% of 89 infants undergoing liver transplantation. Clinically, this deficit predicted significantly greater intraoperative blood loss, longer operative times, and severe postoperative complications, including portal vein stenosis and bloodstream infections. Reinforcing these findings, Verhagen et al. [44] reported 101 children (mostly biliary atresia) and found that in infants <1 year, lower skeletal muscle index correlated with longer hospital and ICU stays, while lower subcutaneous fat was associated with severe infections. These data demonstrate that even in the first year of life, sarcopenia is a major determinant of surgical resilience.- 8. Autoimmune liver diseases: the "hidden" deficit
- 8. Autoimmune liver diseases: the "hidden" deficit
Sarcopenia is prevalent in noncirrhotic conditions like autoimmune hepatitis and primary sclerosing cholangitis, driven by systemic inflammation and corticosteroids rather than malabsorption. Amevor et al. [11] evaluated 58 children using MRI and found that 48% met sarcopenia criteria (tPMA ≤ 5th percentile). Strikingly, while nearly half were sarcopenic, only 3% were classified as underweight by BMI. This profound discordance highlights that reliance on weight-based metrics masks muscle wasting in this population. Furthermore, sarcopenic patients exhibited significantly higher visceral fat volumes, indicating a "sarcopenic obesity" phenotype [21]. Clinically, low muscle mass correlated with worse parent-reported general health scores, suggesting that "hidden" sarcopenia negatively impacts quality of life [11].- 9. Sarcopenia in MASLD
- 9. Sarcopenia in MASLD
MASLD represents a paradigm of sarcopenic obesity, where low muscle mass drives disease severity independent of BMI [45]. Mirroring adult data showing a >5-fold risk of steatosis with low muscle, Yodoshi et al. [19] demonstrated in over 300 children that lower skeletal muscle mass index (SMI) on MRI was independently associated with higher steatosis grades. Multivariate analysis revealed that each 1-unit increase in skeletal muscle index reduced the odds of severe steatosis by 27%, regardless of BMI z score [19]. Subsequent BIA studies confirmed that low muscle mass—rather than body fat percentage—was the stronger predictor of histologic severity [18,19]. This relationship extends to nonobese MASLD; Kwon and Jeong [46] found that 70% of children with fatty liver and BMI <23 kg/m2 met sarcopenia criteria compared to 48% of controls. These findings indicate that relative muscle deficiency is as metabolically detrimental as excess fat, likely by exacerbating insulin resistance and reducing protective myokines [26,47].- 10. Sarcopenia in pediatric IBD
- 10. Sarcopenia in pediatric IBD
Children with IBD are at high risk for sarcopenia, which is frequently present at diagnosis [48]. A systematic review by Aljilani et al. [48,49] confirmed lower lean mass in patients versus controls. MRI studies indicate that up to 80% of pediatric Crohn’s patients have reduced muscle area [16,50]. Pedersen et al. [51] similarly found that lower muscle area predicted complications and surgical risk. Prognostically, sarcopenia marks an aggressive phenotype independent of BMI [52]. Atlan et al. [53] demonstrated in 101 children that those in the lowest muscle quartile faced strikingly higher hazards of requiring biologic therapy (hazard ratio [HR], 12) and experiencing exacerbations (HR, 9). This deficit was more prevalent in Crohn disease (68%) than ulcerative colitis (32%). Fortunately, interventions like exclusive enteral nutrition can improve lean mass [54], underscoring the importance of monitoring muscle mass to predict risk and guide therapy.- 11. Acute illness and sarcopenia
- 11. Acute illness and sarcopenia
"Acute sarcopenia" can develop rapidly during severe illness [55]. López et al. [56] demonstrated in children with perforated appendicitis that psoas muscle area decreased by a mean of 0.9% per day over a 7- to 10-day hospitalization. This rapid catabolism occurred regardless of baseline BMI, affecting even well-nourished children [55]. Mirroring "ICU-acquired weakness," this specific muscle loss is often masked by minimal declines in BMI, yet can significantly delay recovery [57]. Because episodic acute losses can accumulate into chronic depletion, muscle preservation strategies must be initiated immediately upon admission rather than waiting for chronic signs to appear [55].
- Prognostic implications of sarcopenia in pediatric GI and liver diseases
- Prognostic implications of sarcopenia in pediatric GI and liver diseases
- 1. Liver disease and transplantation outcomes
- 1. Liver disease and transplantation outcomes
Pediatric evidence mirrors adult hepatology, identifying sarcopenia as a critical determinant of survival and morbidity independent of PELD scores [17,42]. Boster et al. [42] demonstrated that reduced muscle area predicts significantly higher posttransplant mortality, capturing "biological frailty" often missed by standard labs. Regarding morbidity, sarcopenia in children with chronic liver disease and those undergoing liver transplantation is associated with longer intensive care and hospital stays, greater ventilator dependence, and higher rehospitalization rates [17,42,60]. In infants with biliary atresia, pretransplant sarcopenia correlates with increased intraoperative blood loss, a higher incidence of portal vein complications, prolonged hospitalization, and more frequent severe postoperative infections, underscoring its role as a marker of heightened perioperative vulnerability [60].- 2. Prognostic significance in MASLD and metabolic disease
- 2. Prognostic significance in MASLD and metabolic disease
Although pediatric longitudinal data are limited, sarcopenia appears to accelerate metabolic dysregulation in youth with obesity and MASLD, consistent with adult data linking low muscle mass to MASLD progression, advanced fibrosis, and mortality [61-63]. In adolescents, lower baseline relative muscle mass is associated with a higher subsequent risk of type 2 diabetes and adverse cardiometabolic profiles, suggesting that skeletal muscle loss marks an early trajectory toward metabolic decompensation [63,64]. In adults, the coexistence of sarcopenia and obesity substantially increases the odds of MASLD and MASLDrelated fibrosis [62], thereby defining a “sarcopenic obese” phenotype that likely represents a high-risk subgroup in pediatrics as well, warranting aggressive insulin sensitization and targeted resistance-based exercise to halt disease progression [61,64].- 3. Disease course and surgical risk in IBD and other conditions
- 3. Disease course and surgical risk in IBD and other conditions
In pediatric IBD, sarcopenia functions as a biomarker of more refractory disease, paralleling adult data in which low muscle mass predicts failure of steroid therapy, need for biologics or rescue therapy, and earlier progression to surgery [48,65,66]. It also signals heightened surgical vulnerability, being associated with higher rates of postoperative complications, infections, readmission, and prolonged hospitalization, thereby supporting a prehabilitation approach that leverages the preoperative window for intensive nutritional and functional optimization [48,65,66]. This coupling of low muscle mass with reduced physiological resilience appears consistent across pediatric conditions, with oncology and cardiac cohorts showing that pretreatment sarcopenia predicts treatment toxicity, postoperative complications, and inferior survival [34].
The clinical relevance of sarcopenia in pediatric GI and liver disease extends far beyond anthropometric description; it is an independent marker of higher mortality, longer intensive care utilization, and increased relapse and complication rates in this population [42,58]. Just as osteopenia signals skeletal fragility and fracture risk, sarcopenia denotes a state of physiologic frailty and reduced homeostatic reserve that predisposes children to decompensation under surgical, infectious, or inflammatory stress [17,58,59].
- Pathophysiology: why do children with GI and liver diseases develop sarcopenia?
- Pathophysiology: why do children with GI and liver diseases develop sarcopenia?
- 1. Chronic inflammation and hypermetabolism
- 1. Chronic inflammation and hypermetabolism
The development of sarcopenia in children with chronic GI and liver diseases reflects a convergence of chronic inflammation with anabolic resistance, nutrient deficiency and accelerated starvation, and liver-muscle and endocrine dysregulation, all superimposed on the critical developmental window for muscle and pubertal growth (Fig. 2) [48,67] Chronic systemic inflammation is a central driver of muscle loss in conditions such as IBD and autoimmune liver diseases. Proinflammatory cytokines, including tumor necrosis factor-α, interleukin-1β, and interleukin-6, directly promote skeletal muscle protein breakdown through activation of ubiquitin–proteasome and autophagy pathways [68]. In parallel, these cytokines exert central effects on appetite regulation, inducing anorexia and thereby compounding substrate deficiency at times when metabolic demands are increased [65,69]. Beyond direct catabolism, persistent inflammation induces a state of anabolic resistance. Proinflammatory signaling interferes with the growth hormone (GH)–insulin-like growth factor-1 (IGF-1) axis by inhibiting JAK/STAT signaling, blunting GH-mediated anabolic effects on skeletal muscle [70]. Clinically, this manifests as low circulating IGF-1 levels in children with active Crohn disease despite normal or elevated GH concentrations, resulting in impaired linear growth and failure of muscle accretion during a period when muscle mass should normally increase rapidly [69]. Severe inflammation and ESLD may further exacerbate muscle loss by increasing resting energy expenditure by approximately 10%–20%, such that failure to proportionally increase caloric intake leads to catabolism of both adipose tissue and skeletal muscle to meet energy requirements [69,70].- 2. Nutritional deficits and "accelerated starvation"
- 2. Nutritional deficits and "accelerated starvation"
Inadequate substrate availability compromises muscle protein synthesis and accelerates catabolism, particularly in the background of cirrhosis-related anabolic resistance [67,71,72]. Reduced oral intake is common and multifactorial, driven by cytokine-mediated anorexia, organomegaly-related early satiety, and dysgeusia in chronic liver disease [72]. In cholestatic disorders, fat malabsorption further compounds nutritional compromise by leading to deficiencies in lipid-soluble vitamins. Among these, vitamin D deficiency is especially relevant, as impaired vitamin D signaling has been associated with muscle weakness and myopathy, potentially amplifying functional consequences of muscle mass loss [31]. In advanced liver disease, nutrient deficiency is compounded by a distinct metabolic phenomenon often referred to as “accelerated starvation.” Because the liver is the primary glycogen reservoir, reduced glycogen storage capacity in cirrhosis precipitates an early transition to fasting physiology, even after short periods without intake. Children therefore shift rapidly toward amino acid mobilization from skeletal muscle to support hepatic gluconeogenesis [73]. This pathophysiology provides a strong physiological rationale—supported by clinical trials in cirrhosis—for late-evening nutritional supplementation as a strategy to attenuate overnight muscle protein breakdown and preserve lean mass [67,74].- 3. The liver-muscle axis and hormonal dysregulation
- 3. The liver-muscle axis and hormonal dysregulation
Chronic liver disease creates a biochemical and endocrine environment that actively suppresses muscle health. A prominent example is hyperammonemia. When hepatic ammonia clearance is impaired, skeletal muscle becomes an alternative site for ammonia disposal via conversion to glutamine; however, this adaptive mechanism carries metabolic costs, including consumption of branched-chain amino acids that would otherwise support muscle protein synthesis [75]. Hyperammonemia is also directly toxic to myocytes: it impairs mitochondrial function, reduces ATP production, and increases reactive oxygen species generation [75]. Concomitantly, hyperammonemia upregulates myostatin—an inhibitor of muscle growth—and inhibits mechanistic target of rapamycin (mTOR) signaling, a central regulator of muscle protein synthesis [75]. Endocrine perturbations further contribute to impaired muscle accrual. Cirrhosis can disrupt the gonadal axis, contributing to low testosterone in adolescent males via increased aromatization to estrogen [76]. Given the central role of sex steroids in pubertal gains in muscle mass, pubertal delay and hypogonadism can meaningfully worsen failure to accrue muscle. Metabolic dysregulation also plays a key role, particularly in MASLD and cirrhosis. Systemic insulin resistance reduces insulin’s anabolic suppression of proteolysis and impairs glucose uptake in skeletal muscle, contributing to metabolic inflexibility and promoting fat infiltration into muscle (myosteatosis), which further impairs muscle quality and physical function [77].- 4. Iatrogenic and environmental factors
- 4. Iatrogenic and environmental factors
Iatrogenic exposures and reduced mechanical loading frequently amplify disease-driven muscle loss. Glucocorticoids are potently catabolic, suppressing muscle protein synthesis, increasing proteolysis, and worsening insulin resistance [78]. With prolonged exposure, type II fiber atrophy and proximal weakness become clinically relevant, particularly in steroid-dependent IBD and autoimmune hepatitis. In parallel, fatigue, pain, and hospitalization reduce habitual physical activity and mechanical loading. Because mechanical loading is required to stimulate muscle anabolism (“use it or lose it”), disuse accelerates atrophy and worsens insulin resistance [79]. Consistent with this framework, in pediatric liver transplant recipients, lower moderate-to-vigorous physical activity has been associated with sarcopenia [80], suggesting that physical inactivity is not merely a consequence but also a reinforcing driver of muscle depletion.- 5. Pediatric specificity: impaired muscle accrual during growth
- 5. Pediatric specificity: impaired muscle accrual during growth
A defining pediatric vulnerability is that chronic disease blunts the normal trajectory of muscle gain, especially during puberty. Thus, a teenager with IBD and delayed puberty may not only lose muscle during catabolic episodes but also miss the developmental window for peak muscle acquisition [70,81]. Even after disease control, incomplete “catch-up” in muscle accrual can leave a persistent deficit—distinct from adult atrophy and tightly linked to growth failure and delayed sexual maturation [70].
- Diagnosis and assessment of pediatric sarcopenia
- Diagnosis and assessment of pediatric sarcopenia
- 1. Clinical assessment and screening
- 1. Clinical assessment and screening
Assessment begins with a high index of suspicion. Physical signs such as temporal wasting, prominent interosseous muscles, or “flabby” skin folds may suggest muscle depletion, but examination is relatively insensitive for mild sarcopenia, particularly in children with overweight or obesity in whom muscle loss can be masked by adiposity [21]. Longitudinal growth trajectories remain essential: declines in weight-for-height or BMI percentiles may indicate loss of somatic tissue that often includes muscle. mid-upper arm circumference (MUAC), triceps skinfold, and mid-upper arm muscle area can serve as bedside adjuncts; however, their specificity for muscle depletion is limited. For example, in infants with ESLD, MUAC did not correlate with imaging-based psoas area index [41], indicating that a “normal” MUAC cannot reliably exclude sarcopenia. In addition, fluid-retaining states (edema/ascites) can confound anthropometric interpretation, further reinforcing the need for objective measures.- 2. Objective muscle mass quantification
- 2. Objective muscle mass quantification
1) Dual-energy x-ray absorptiometry
1) Dual-energy x-ray absorptiometry
DXA is widely used modality in pediatric practice because it is rapid, widely available, and low radiation [7]. DXA estimates ALM, typically normalized by height squared (ALM/height2), and interpreted using pediatric reference populations. In most pediatric studies, low muscle mass is operationalized using z scores or percentiles (e.g., z score < -2 or <5th percentile) [7].2) Bioelectrical impedance analysis
2) Bioelectrical impedance analysis
BIA is a practical, bedside alternative that is radiation-free and scalable, particularly with modern multifrequency devices. However, BIA is highly hydration-sensitive; in decompensated liver disease with ascites/edema, lean mass may be overestimated because excess fluid is captured as fat-free mass, whereas dehydration can bias estimates downward. Thus, BIA is best suited for euvolemic patients and for longitudinal trend monitoring in stable settings. In pediatric studies, BIA-derived indices such as SMI and muscle-to-fat ratio (MFR) have been used. Recent age- and sex-specific BIA reference data provide a more explicit interim framework: low muscle mass can be defined as SMI < -2 standard deviation (SD), and MFR-based sarcopenia as an MFR below the mean -2 SD of individuals in the 3rd BMI quintile, stratified by age and sex [22]. These criteria may improve standardization of BIA-based assessment in pediatric practice, although further validation in disease-specific GI/hepatology cohorts remains needed.3) Cross-sectional imaging (CT and MRI)
3) Cross-sectional imaging (CT and MRI)
These imaging provide the most precise assessment of muscle quantity and, in some settings, quality. CT and MRI allow direct visualization of muscle compartments and can identify myosteatosis (fat infiltration) [7]. In pediatrics, psoas muscle area or total abdominal muscle area is commonly measured at L3–4 or L4–5.7) MRI is preferred when available because it avoids ionizing radiation and is often already obtained for clinical indications (e.g., MRI-PDFF [proton density fat fraction] for MASLD or MR enterography for Crohn), enabling “opportunistic” body composition assessment without additional burden [11,19]. Diagnostic thresholds vary, but many pediatric studies use age/sex-referenced z scores or percentiles, with psoas muscle index commonly calculated as (right+left psoas area)/height2 and low muscle mass defined as <5th percentile or >2 SD below reference [7,19].4) Emerging modalities (ultrasound)
4) Emerging modalities (ultrasound)
Muscle ultrasound is an emerging, radiation-free modality. By measuring the thickness of muscle (like quadriceps femoris) or cross-sectional area via ultrasound, one can estimate muscle mass. Pediatric applicability is promising (it’s used in neonatal intensive care units to track preemie nutrition status via thigh muscle thickness) [84]. Preliminary pediatric studies have used ultrasound of rectus femoris as a quick gauge of muscle mass during hospital stays (e.g., in pediatric intensive care unit patients, decreasing quad thickness predicted worse outcomes) [85,86]. However, it is highly operator-dependent and lacks standardization. No universal pediatric cutoffs exist, and pediatric radiologists are still validating it. It may become more popular for monitoring trends (e.g., measure muscle thickness weekly to see if it’s declining), but as of now, ultrasound is not a primary modality for diagnosing sarcopenia in the pediatric GI/hepatology context – it’s more in critical care or experimental use.5) Functional assessment
5) Functional assessment
While muscle mass quantification is essential, adult definitions emphasize that sarcopenia is a failure of both mass and function [3,5].• Handgrip strength (children >5–6 years): Handgrip dynamometry is the most validated functional test in older children [87]. It is simple, reproducible, and correlates with overall total muscle strength. Low grip strength is typically defined as < -2 SD or <10th percentile for age and sex [88]. Ooi et al. [80] successfully applied a cutoff of grip strength < -2 SD to identify sarcopenia in children following liver transplantation [81]. While valuable, results can be confounded by hand pain (e.g., arthritis) or lack of motivation, and reliability is limited in children under 6 years [87)].• Physical performance measures: Performance tests assess the real-world impact of muscle loss. Common tools include the 6-minute walk test (6MWT) and stair-climb power. In the same posttransplant cohort by Ooi et al. [80], sarcopenic children exhibited significantly slower stair-climb times compared to nonsarcopenic peers, highlighting a functional deficit relevant to daily life. In a busy clinic, simple bedside tests—such as counting chairrise repetitions or timed single-leg standing—can provide qualitative data, though standardized cutoffs for these are lacking [89]. It is important to note that tests like the 6MWT are nonspecific; a poor result may reflect cardiopulmonary deconditioning rather than isolated muscle weakness [80].• Infants and toddlers: In children too young for dynamometry, direct strength testing is not feasible. Clinicians must infer functional status from developmental milestones. Validated tools like the AIMS can identify gross motor delays (e.g., failure to sit unsupported or crawl on time), which in the context of chronic liver disease, often serve as the functional surrogate for sarcopenia [90].
Laboratory markers are occasionally discussed as indirect surrogates of muscle mass, but their clinical utility remains limited [82]. Low serum creatinine in the setting of stable renal function may suggest reduced muscle mass, and the creatinine-to-cystatin C ratio has emerged as a promising pediatric adjunct associated with impaired physical performance. However, these markers remain nonspecific and are influenced by renal function, inflammation, and liver disease; therefore, they should be viewed as practical screening adjuncts rather than stand-alone diagnostic tests for pediatric sarcopenia. Mechanistically relevant markers such as IGF-1 or myostatin are also of interest, but they are not yet validated for routine clinical use [83].- 3. Clinical application: a proposed diagnostic algorithm
- 3. Clinical application: a proposed diagnostic algorithm
Step 1: Risk stratification and screening
Step 1: Risk stratification and screening
The first step is identifying the "at-risk" child. Clinicians should maintain a high index of suspicion in patients with:· Chronic liver disease (especially biliary atresia or cirrhosis).· Active or steroid-dependent IBD.· Persistent growth failure or weight stagnation. While simple screening tools like BMI or MUAC alone lack sensitivity for isolated muscle loss, values <5th percentile should trigger immediate further evaluation.Step 2: Objective quantification
Step 2: Objective quantification
If suspicion is raised, objective measurement is required. The choice of modality depends on availability (Table 1):Step 3: Functional assessment (The "severity" step)
Step 3: Functional assessment (The "severity" step)
In children capable of cooperation (typically >6 years), measuring muscle function distinguishes simple muscle wasting from true sarcopenia (Table 1). Table 1 summarizes the major diagnostic modalities, their pediatric applications, and pragmatic interim criteria. Representative pediatric GI/hepatology conditions associated with sarcopenia, along with commonly used definitions, approximate prevalence, and major outcome signals, are summarized in Table 2.· Handgrip dynamometry: A grip strength z score < -2 serves as a rapid, validated marker of functional decline [87].· Performance: In rehabilitation settings, the 6MWT can quantify endurance deficits [80].Step 4: Diagnosis
Step 4: Diagnosis
In cooperative children, a pragmatic diagnosis of pediatric sarcopenia should be made when low muscle mass-defined as a z score < -2 or <5th percentile relative to age-and sex-specific reference populations-is accompanied by low muscle function based on age-appropriate reference standards [7,87,91,92]. When validated functional testing is not feasible, particularly in infants, toddlers, or critically ill children, low muscle mass alone may support an operational diagnosis (or probable sarcopenia) that should prompt intervention and follow-up reassessment [7,90]. For BIA-based assessment, recent age- and sex-specific pediatric criteria include SMI < -2 SD and MFR below the mean -2 SD of individuals in the 3rd BMI quintile, stratified by age and sex [22]. Where available, ethnicity-specific pediatric references should be used, particularly for BIA-based assessment in Asian populations [7,22].
Given the heterogeneity of current methods and the lack of a universal consensus, we propose a pragmatic, step-by-step diagnostic algorithm (Fig. 3) to standardize sarcopenia assessment in pediatric gastroenterology and hepatology.- 4. The importance of reference data
- 4. The importance of reference data
A major barrier to standardized diagnosis has historically been the lack of robust pediatric normative data [7]. Recent advances, however, have provided clinically useful benchmarks. Notably, Lurz et al. [2] published sex-specific percentiles for tPMA derived from healthy children aged 1–16 years, allowing muscle measurements to be interpreted analogously to growth charts. More recently, BIA-derived age- and sex-specific reference curves and sarcopenia-related cutoffs have been reported in Asian youth, including SMI < -2 SD and MFR below the mean -2 SD of the age- and sex-stratified 3rd BMI quintile as practical interim cutoffs [22]. Because these thresholds are modality- and population-specific, they should complement rather than replace broader pediatric reference standards, but they represent an important step toward ethnicity-sensitive assessment [22]. Similarly, normative reference curves for handgrip strength have been established by the CDC and other cohorts [91,92]. The use of such population-specific references is essential; applying fixed adult cutoffs to growing children will inevitably result in misclassification and diagnostic error.- 5. Differentiation from secondary causes
- 5. Differentiation from secondary causes
Clinicians must distinguish sarcopenia related to systemic GI/liver disease from "secondary" sarcopenia caused by primary neuromuscular disorders (e.g., cerebral palsy) or severe disuse (e.g., casting). The algorithm presented here focuses on metabolic and nutritional etiologies, where intervention can reverse the underlying deficit.
Diagnosing sarcopenia in children requires a shift from subjective observation to objective quantification. Unlike in adults, where sarcopenia is defined by fixed thresholds, pediatric assessment must account for growth and pubertal stage, requiring integration of body composition analysis and, when feasible, functional testing [7]. Table 1 summarizes the major diagnostic modalities, their pediatric applications, and pragmatic interim criteria. Table 1 summarizes the major diagnostic modalities, their pediatric applications, and pragmatic interim criteria. Representative pediatric GI/hepatology conditions associated with sarcopenia, along with commonly used definitions, approximate prevalence, and major outcome signals, are summarized in Table 2.
- Management of pediatric sarcopenia
- Management of pediatric sarcopenia
- 1. Nutritional rehabilitation
- 1. Nutritional rehabilitation
1) Optimizing protein and caloric intake
1) Optimizing protein and caloric intake
Children with sarcopenia require protein intakes at the higher end of physiological recommendations to overcome anabolic resistance.• Protein targets: Current guidelines suggest 1.5–2.0 g/kg/day for children with chronic diseases [93]. Infants with cholestasis may require up to 2–3 g/kg/day to support catch-up growth [94,95]. Even in obesity-related sarcopenia (MASLD), protein restriction should be avoided; high-protein diets (20%–25% of energy) help preserve lean mass during weight management.• Caloric needs: Caloric goals must account for hypermetabolic states. For example, a child with cystic fibrosis may require 120%–130% of estimated energy needs to reverse catabolism [96].2) Specific micronutrients and supplements
2) Specific micronutrients and supplements
The foundation of management is ensuring sufficient substrate for muscle protein synthesis. Malnutrition and hypermetabolism frequently coexist in this population, necessitating aggressive support.- 2. Exercise and physical activity: the anabolic stimulus
- 2. Exercise and physical activity: the anabolic stimulus
Exercise is the primary physiologic stimulus for muscle hypertrophy [99]. While nutritional support provides the amino acids, mechanical loading provides the signal (mTOR activation) required to build muscle [99,100]. Without this stimulus, nutritional supplementation alone may lead to fat gain rather than muscle accretion.- 4. Strategy 1: In-hospital mobilization
- 4. Strategy 1: In-hospital mobilization
The rapid muscle catabolism observed in pediatric acute appendicitis (~1% loss of psoas area per day) serves as a stark warning: muscle loss begins immediately upon immobilization [56].• Goal: Mitigate "acute sarcopenia" during hospitalization for flares or surgery.• Intervention: Mobilization should be viewed as a "vital sign." For bedbound patients (e.g., severe acute colitis), physical therapy (PT) should initiate low-intensity in-bed resistance exercises (e.g., bridging, straight leg raises) and assisted transfers [105,106]. Ambulation in the hallway should occur as soon as hemodynamic stability allows [106].- 5. Strategy 2: Outpatient resistance training (rebuilding the reserve)
- 5. Strategy 2: Outpatient resistance training (rebuilding the reserve)
For the stable outpatient, the focus shifts to hypertrophy. The prescription follows the FITT(frequency, intensity, time, type) principle [107]:• Type (modality): Resistance training is superior to aerobic exercise for reversing sarcopenia [107, 108]. Effective modalities include body-weight exercises (squats, lunges, push-ups), resistance bands (Therabands), or light free weights. These induce the mechanical tension necessary for muscle growth.• Frequency & time: Aim for 2–3 sessions per week, with each session lasting 20–30 minutes [107,108]. Consistency is more critical than duration.- 6. Strategy 3: Developmental considerations
- 6. Strategy 3: Developmental considerations
Management should be individualized to developmental stage. In infants and younger children, the priority is preservation of growth and muscle accrual through energy-and protein-adequate feeding, correction of micronutrient deficiencies, and play-based or PT-guided activity that promotes gross motor development. In school-aged prepubertal children, structured habit-building, supervised body-weight or resistance-band exercise, and close monitoring of growth velocity are central. In adolescents, pubertal muscle accretion, delayed puberty, autonomy, and body image become increasingly relevant; accordingly, higher absolute protein delivery, progressive resistance training, and evaluation for endocrine contributors such as delayed puberty or hypogonadism should be considered when recovery is suboptimal. Although pediatric sarcopenia-specific trials are limited, tailoring interventions to growth stage is physiologically justified and clinically pragmatic.• Engaging Adolescents [110]: Teenagers are often motivated by functional goals (e.g., "returning to sports") or aesthetics ("looking fit") rather than "disease management." Framing resistance training as "sports conditioning" can improve adherence.• School Reintegration [111]: Returning to physical education class is a key milestone. Clinicians should provide clear clearance letters that allow for "activity as tolerated" or adaptive physical education (e.g., shorter duration, no contact) to prevent social isolation while ensuring safety.- 7. Emerging and adjunctive therapies
- 7. Emerging and adjunctive therapies
While standard care focuses on nutrition and exercise, several adjunctive therapies are under investigation or used in refractory cases:• Hormonal agents: In adolescents with severe growth failure or delayed puberty, GH or low-dose anabolic steroids (e.g., oxandrolone) have been used off-label to promote anabolism (e.g., in burn victims or Turner syndrome) [112,113]. While effective for muscle gain, they carry risks of virilization and liver enzyme elevation. Their use is currently reserved for exceptional, refractory cases under expert endocrine guidance.• Neuromuscular electrical stimulation (NMES): For critically ill children in the ICU who are too sedated or weak for active PT, NMES can passively stimulate muscle contraction. Small trials in adults suggest it may attenuate atrophy during prolonged immobilization; pediatric applications are exploring this as a bridge to active rehab [114,115].- 8. Integrated management and monitoring
- 8. Integrated management and monitoring
1) Monitoring progress: a dynamic loop
1) Monitoring progress: a dynamic loop
- (1) Surveillance frequency
- (1) Surveillance frequency
· Every clinic visit (3–6 months): Assess functional metrics such as handgrip strength. This acts as a "vital sign" for muscle health, providing immediate feedback on functional recovery.· Annual/biannual: Reassess objective body composition (DXA or BIA). A 6- to 12-month interval is typically required to detect measurable changes in lean mass z scores.- (2) Red flags (Troubleshooting failure): If muscle mass fails to improve despite reported adherence to nutrition and exercise, the clinician must investigate for "silent" barriers:
- (2) Red flags (Troubleshooting failure): If muscle mass fails to improve despite reported adherence to nutrition and exercise, the clinician must investigate for "silent" barriers:
· Subclinical disease activity: Is there ongoing microscopic inflammation (e.g., elevated fecal calprotectin) driving anabolic resistance?· Endocrine comorbidities: Has the child developed hypothyroidism, hypogonadism (delayed puberty), or Vitamin D deficiency?· Social/behavioral barriers: Are there financial constraints to accessing high-protein food, or is depression preventing exercise adherence?
Sarcopenia management is not a "set and forget" intervention. It requires dynamic monitoring to ensure efficacy.2) Defining success: the window of opportunity
2) Defining success: the window of opportunity
- (1) Functional restoration
- (1) Functional restoration
The ultimate goal is functional capability. A teenager reporting, "I can now finish a full day of school without needing a nap" or "I rejoined the soccer team," represents a clinical victory far more meaningful than a z score change [116].- (2) Reversibility
- (2) Reversibility
Pediatric sarcopenia is a reversible lesion, provided it is identified and treated during the critical window of growth [3]. The goal is to intervene before the epiphyseal plates close, securing not just the child's current health, but their lifelong metabolic resilience.
Success in pediatric sarcopenia differs from geriatrics. While success in the elderly often means "slowing decline," children possess immense regenerative capacity.
Successful treatment of pediatric sarcopenia requires a shift from a siloed "organ-specific" approach to a comprehensive "whole-body" strategy. This demands a coordinated multidisciplinary team to orchestrate the complex interplay between disease control, nutrition, and rehabilitation.
Managing sarcopenia in children requires a multipronged approach addressing its root causes: inadequate nutrition, physical inactivity, and uncontrolled underlying disease [34]. Unlike in adults, there are no approved pharmacologic agents specifically for sarcopenia. Therefore, therapy rests on three pillars: Nutritional Rehabilitation, Resistance Exercise, and Disease Optimization, followed by structured monitoring.
- Conclusion
- Conclusion
Sarcopenia in pediatric gastroenterology and hepatology has emerged from an underrecognized research concept to a clinically relevant condition affecting a substantial proportion of children with chronic liver disease, IBD, and obesity-related disorders such as MASLD. Importantly, pediatric sarcopenia is often occult, masked by normal growth parameters or excess adiposity, yet it confers independent risk for adverse outcomes including perioperative morbidity, prolonged recovery, and impaired quality of life.This review highlights the need for a conceptual shift in clinical assessment from reliance on body weight and BMI toward deliberate evaluation of muscle mass and, when feasible, muscle function. A body-composition–based approach that also considers growth, puberty, adiposity distribution, and obesity-related clinical consequences is likely to identify high-risk children more accurately than weight-based metrics alone. Although a unified pediatric definition remains to be established, pragmatic tools—including DXA, opportunistic cross-sectional imaging, and handgrip dynamometry—are already available and can be incorporated into routine care to identify vulnerable patients before overt decompensation. From a management perspective, the multifactorial pathophysiology of pediatric sarcopenia underscores the limitations of isolated interventions. Effective care requires an integrated strategy combining optimization of underlying disease activity with protein-adequate nutritional support and resistance-based physical activity to counteract anabolic resistance.Notably, pediatric sarcopenia differs fundamentally from its geriatric counterpart in that muscle loss in children is potentially reversible. Early identification during critical periods of growth and pubertal development provides a window of opportunity to restore muscle mass and functional reserve. Accordingly, routine attention to muscle health should be considered a core component of pediatric gastroenterology and hepatology practice. By prioritizing muscle assessment and preservation alongside traditional disease-specific outcomes, clinicians may favorably modify the long-term trajectory of chronic GI and liver diseases in children.
- Footnotes
-
Conflicts of interest No potential conflict of interest relevant to this article was reported.
Funding This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contribution All contributions to this study were made by the single author (TY).
-
Fig. 1.

Fig. 2.
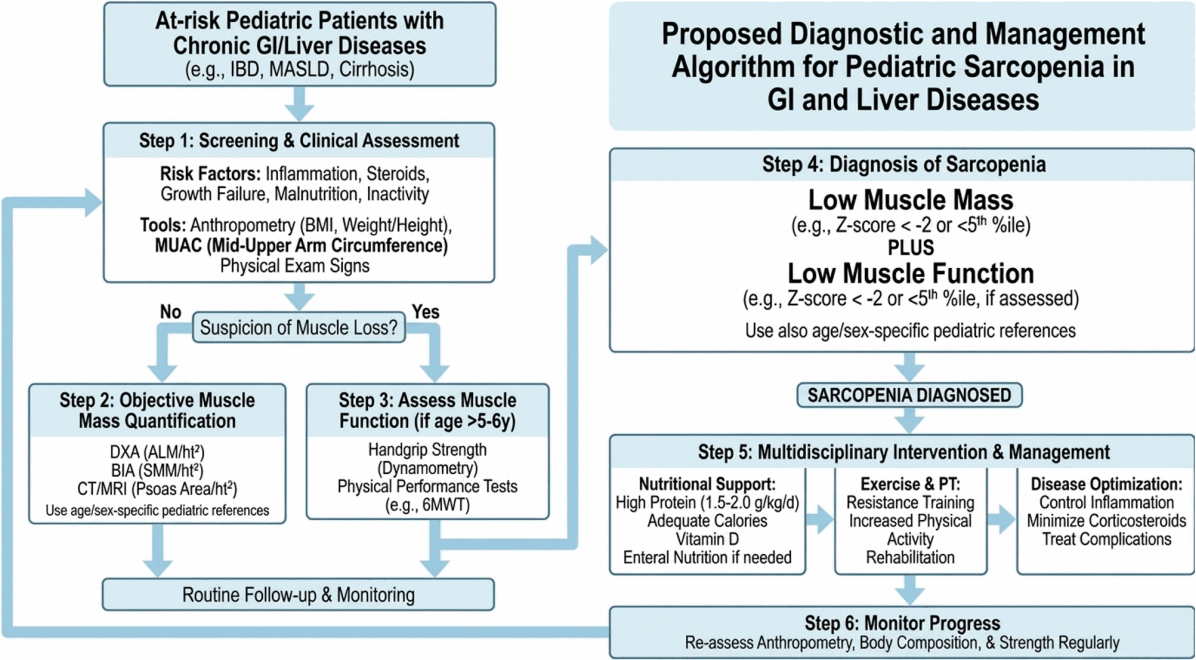
Fig. 3.

Table 1.
BMI, body mass index; MUAC, mid-upper arm circumference; DXA, dual-energy x-ray absorptiometry; ALM, appendicular lean mass; BIA, bioelectrical impedance analysis; SMI, skeletal muscle mass index; SD, standard deviation; MFR, muscle-to-fat ratio; MRI, magnetic resonance imaging; CT, computed tomography; tPMA, total psoas muscle area; NICU, neonatal intensive care unit; 6MWT, 6-minute walk test; BMC, bone mineral content; EWGSOP, European Working Group on Sarcopenia in Older People; HU, Hounsfield unit; PICU, pediatric intensive care unit; TUG, Timed Up and Go test.
Table 2.
| Condition | Definition/measurement (examples) | Reported prevalence (approximately) | Outcome signals (examples) |
|---|---|---|---|
| Autoimmune liver diseases (AIH/PSC/ASC) | MRI tPMA ≤5th percentile | 48% in one cohort | Higher visceral fat; worse parent-perceived general health [8] |
| End-stage liver disease/transplant candidates | CT/MRI psoas z score <−2 or low PMI | 24%–40% | Longer ICU stay; infections; perioperative morbidity [40-42] |
| Biliary atresia (infants at transplant) | CT/MRI tPMA z score <−2 | 24% in infant cohort | More perioperative complications [39,44] |
| Pediatric MASLD | MRI/BIA muscle indices | prevalence varies by definition | Lower muscle linked to greater steatosis severity [18,19] |
| Pediatric IBD (Crohn/UC) | MRI/DXA lean mass indices | Variable; low muscle common | Exacerbations; biologic escalation; surgical risk [48-53] |
| Acute complicated appendicitis | CT serial psoas area change | Not prevalence; acute loss observed | 0.8%–1% psoas area loss per day during admission [56] |
GI, gastrointestinal; AIH, autoimmune hepatitis; PSC, primary sclerosing cholangitis; ASC, autoimmune sclerosing cholangitis; MRI, magnetic resonance imaging; tPMA, total psoas muscle area; CT, computed tomography; PMI, psoas muscle index; ICU, intensive care unit; MASLD, metabolic dysfunction-associated steatotic liver disease; BIA, bioelectrical impedance analysis; IBD, inflammatory bowel disease; UC, ulcerative colitis; DXA, dual-energy x-ray absorptiometry.
- References
- 1. Rosenberg IH. Sarcopenia: origins and clinical relevance. J Nutr 1997;127:990S–991S.
[Article] [PubMed]2. Lurz E, Patel H, Lebovic G, Quammie C, Woolfson JP, Perez M, et al. Paediatric reference values for total psoas muscle area. J Cachexia Sarcopenia Muscle 2020;11:405–14.
[Article] [PubMed] [PMC]3. Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 2019;48:16–31.
[Article] [PubMed] [PMC]4. Xu J, Wan CS, Ktoris K, Reijnierse EM, Maier AB. Sarcopenia is associated with mortality in adults: a systematic review and meta-analysis. Gerontology 2022;68:361–76.
[Article] [PubMed]5. Chen LK, Hsiao FY, Akishita M, Assantachai P, Lee WJ, Lim WS, et al. A focus shift from sarcopenia to muscle health in the Asian Working Group for Sarcopenia 2025 Consensus Update. Nat Aging 2025;5:2164–75.
[Article] [PubMed]6. Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, et al. Asian Working Group for Sarcopenia: 2019 Consensus Update on Sarcopenia Diagnosis and Treatment. J Am Med Dir Assoc 2020;21:300–7.e2.
[Article] [PubMed] [PMC]7. Inoue T, Wakabayashi H, Kawase F, Kokura Y, Takamasu T, Fujiwara D, et al. Diagnostic criteria, prevalence, and clinical outcomes of pediatric sarcopenia: a scoping review. Clin Nutr 2024;43:1825–43.
[Article] [PubMed]8. Chen C, Ayers M, Squires JH, Squires JE. Perspectives on sarcopenia as a predictor for outcomes in pediatric patients with chronic liver disease. Hepat Med 2022;14:173–83.
[Article] [PubMed] [PMC]9. Ligthart-Melis GC, Luiking YC, Kakourou A, Cederholm T, Maier AB, de van der Schueren MA. Frailty, sarcopenia, and malnutrition frequently (co-)occur in hospitalized older adults: a systematic review and meta-analysis. J Am Med Dir Assoc 2020;21:1216–28.
[Article] [PubMed]10. Rubino F, Cummings DE, Eckel RH, Cohen RV, Wilding JPH, Brown WA, et al. Definition and diagnostic criteria of clinical obesity. Lancet Diabetes Endocrinol 2025;13:221–62.
[PubMed] [PMC]11. Amevor AA, Yodoshi T, Trout AT, Dillman JR, Singh R, Jarvis R, et al. Sarcopenia is highly prevalent in children with autoimmune liver diseases and is linked to visceral fat and parent-perceived general health. Liver Int 2022;42:394–401.
[Article] [PubMed] [PMC]12. Di Giorgio A, Mandato C, Lurz E. Frailty in children with chronic liver disease: growing evidence for an underexplored topic. J Pediatr Gastroenterol Nutr 2025;81:907–10.
[Article] [PubMed]13. Beer SS, Wong Vega M. Malnutrition, sarcopenia, and frailty assessment in pediatric transplantation. Nutr Clin Pract 2024;39:27–44.
[Article] [PubMed]14. Kalafateli M, Mantzoukis K, Choi Yau Y, Mohammad AO, Arora S, Rodrigues S, et al. Malnutrition and sarcopenia predict post-liver transplantation outcomes independently of the Model for End-stage Liver Disease score. J Cachexia Sarcopenia Muscle 2017;8:113–21.
[Article] [PubMed] [PMC]15. Boros KK, Veres G, Pintér HK, Richter É, Cseh Á, Dezsőfi A, et al. Novel approach to assess sarcopenia in children with inflammatory bowel disease. Front Pediatr 2024;12:1204639.
[Article] [PubMed] [PMC]16. Çakar S, Eren G, Erdur CB, Önder M, Pelek Ş, Demirtaş D, et al. Are Vitamin D levels related to sarcopenia in children with inflammatory bowel disease? J Clin Med 2025;14:1548.
[Article] [PubMed] [PMC]17. Woolfson JP, Perez M, Chavhan GB, Johara FT, Lurz E, Kamath BM, et al. Sarcopenia in children with end-stage liver disease on the transplant waiting list. Liver Transpl 2021;27:641–51.
[Article] [PubMed] [PMC]18. Yodoshi T, Orkin S, Romantic E, Hitchcock K, Clachar AA, Bramlage K, et al. Impedance-based measures of muscle mass can be used to predict severity of hepatic steatosis in pediatric nonalcoholic fatty liver disease. Nutrition 2021;Nov-Dec;91-92:111447. doi: 10.1016/j.nut.2021.111447. [Epub].
[Article] [PubMed] [PMC]19. Yodoshi T, Orkin S, Arce Clachar AC, Bramlage K, Sun Q, Fei L, et al. Muscle mass is linked to liver disease severity in pediatric nonalcoholic fatty liver disease. J Pediatr 2020;223:93–9.e2.
[Article] [PubMed] [PMC]20. Orsso CE, Tibaes JR, Oliveira CL, Rubin DA, Field CJ, Heymsfield SB, et al. Low muscle mass and strength in pediatrics patients: Why should we care? Clin Nutr 2019;38:2002–15.
[Article] [PubMed]21. Evensen E, Emaus N, Furberg AS, Kokkvoll A, Wells J, Wilsgaard T, et al. Adolescent body composition and associations with body size and growth from birth to late adolescence. The Tromsø study: Fit Futures-A Norwegian longitudinal cohort study. Pediatr Obes 2019;14:e12492.
[Article] [PubMed]22. Song K, Lee E, Lee HS, Lee H, Kim JY, Choi Y, et al. Body composition references, sarcopenia cutoffs, and prevalence in youth using bioelectrical impedance analysis. Int J Obes (Lond) 2026;50:319–28.
[Article] [PubMed]23. Zembura M, Matusik P. Sarcopenic obesity in children and adolescents: a systematic review. Front Endocrinol (Lausanne) 2022;13:914740.
[Article] [PubMed] [PMC]24. Donini LM, Busetto L, Bischoff SC, Cederholm T, Ballesteros-Pomar MD, Batsis JA, et al. Definition and diagnostic criteria for sarcopenic obesity: ESPEN and EASO consensus statement. Clin Nutr 2022;41:990–1000.
[PubMed]25. Liu C, Wong PY, Chung YL, Chow SK, Cheung WH, Law SW, et al. Deciphering the "obesity paradox" in the elderly: a systematic review and meta-analysis of sarcopenic obesity. Obes Rev 2023;24:e13534.
[Article] [PubMed]26. Xanthakos SA, Ibrahim SH, Adams K, Kohli R, Sathya P, Sundaram S, et al. AASLD Practice Statement on the evaluation and management of metabolic dysfunction-associated steatotic liver disease in children. Hepatology 2025;82:1352–94.
[Article] [PubMed]27. Hong SH, Choi KM. Sarcopenic obesity, insulin resistance, and their implications in cardiovascular and metabolic consequences. Int J Mol Sci 2020;21:494.
[Article] [PubMed] [PMC]28. Wells JCK, Briend A, Boyd EM, Berkely JA, Hall A, Isanaka S, et al. Beyond wasted and stunted-a major shift to fight child undernutrition. Lancet Child Adolesc Health 2019;3:831–4.
[Article] [PubMed]29. Hanlon P, Wightman H, Politis M, Kirkpatrick S, Jones C, Andrew MK, et al. The relationship between frailty and social vulnerability: a systematic review. Lancet Healthy Longev 2024;5:e214–26.
[Article] [PubMed]30. Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 2001;56:M146–56.
[Article] [PubMed] [PMC]31. Lai JC, Tandon P, Bernal W, Tapper EB, Ekong U, Dasarathy S, et al. Malnutrition, frailty, and sarcopenia in patients with cirrhosis: 2021 Practice Guidance by the American Association for the study of liver diseases. Hepatology 2021;74:1611–44.
[Article] [PubMed] [PMC]32. Ooi PH, Hager A, Mazurak VC, Dajani K, Bhargava R, Gilmour SM, et al. Sarcopenia in chronic liver disease: impact on outcomes. Liver Transpl 2019;25:1422–38.
[Article] [PubMed]33. Tagliafico AS, Bignotti B, Torri L, Rossi F. Sarcopenia: how to measure, when and why. Radiol Med 2022;127:228–37.
[Article] [PubMed] [PMC]34. Kiosis G, Ioannou D, Skourtsidis K, Fouskas V, Stergiou K, Kavvadas D, et al. Newer insights on the occurrence of sarcopenia in pediatric patients with cancer: a systematic review of the past 5 years of literature. Cancers (Basel) 2025;17:3188.
[Article] [PubMed] [PMC]35. Cederholm T, Barazzoni R, Austin P, Ballmer P, Biolo G, Bischoff SC, et al. ESPEN guidelines on definitions and terminology of clinical nutrition. Clin Nutr 2017;36:49–64.
[Article] [PubMed] [PMC]36. Tantai X, Liu Y, Yeo YH, Praktiknjo M, Mauro E, Hamaguchi Y, et al. Effect of sarcopenia on survival in patients with cirrhosis: a meta-analysis. J Hepatol 2022;76:588–99.
[Article] [PubMed]37. Kim G, Kang SH, Kim MY, Baik SK. Prognostic value of sarcopenia in patients with liver cirrhosis: a systematic review and meta-analysis. PLoS One 2017;12:e0186990.
[Article] [PubMed] [PMC]38. He S, Liu CH, Wang Y, Li Z, Liu Z, Zeng H, et al. The prognostic value of sarcopenia in acute-on-chronic liver failure: a systematic review and meta-analysis. BMC Gastroenterol 2025;25:300.
[Article] [PubMed] [PMC]39. Takeda M, Sakamoto S, Uchida H, Shimizu S, Yanagi Y, Fukuda A, et al. Impact of sarcopenia in infants with liver transplantation for biliary atresia. Pediatr Transplant 2021;25:e13950.
[Article] [PubMed]40. Mangus RS, Bush WJ, Miller C, Kubal CA. Severe sarcopenia and increased fat stores in pediatric patients with liver, kidney, or intestine failure. J Pediatr Gastroenterol Nutr 2017;65:579–83.
[Article] [PubMed]41. Lurz E, Patel H, Frimpong RG, Ricciuto A, Kehar M, Wales PW, et al. Sarcopenia in children with end-stage liver disease. J Pediatr Gastroenterol Nutr 2018;66:222–6.
[Article] [PubMed]42. Boster JM, Browne LP, Pan Z, Zhou W, Ehrlich PF, Sundaram SS. Higher mortality in pediatric liver transplant candidates with sarcopenia. Liver Transpl 2021;27:808–17.
[Article] [PubMed] [PMC]43. Jitwongwai S, Lertudomphonwanit C, Junhasavasdikul T, Fuangfa P, Tanpowpong P, Gesprasert G, et al. Low psoas muscle index as an unfavorable factor in children with endstage liver disease undergoing liver transplantation. Pediatr Transplant 2021;25:e13996.
[Article] [PubMed]44. Verhagen MV, Levolger S, Hulshoff JB, Werner MJ, van der Doef HP, Viddeleer AR, et al. Utility of preoperative computed tomography-based body metrics in relation to postoperative complications in pediatric liver transplantation recipients. Liver Transpl 2021;27:1779–87.
[Article] [PubMed] [PMC]45. Wang P, Liu X, Du X, Qiu L, Liu Y, Xu S, et al. Prevalence and risk of metabolic dysfunction-associated steatotic liver disease in patients with sarcopenic obesity: a systematic review and meta-analysis. Nutr Metab (Lond) 2025;22:101.
[Article] [PubMed] [PMC]46. Kwon Y, Jeong SJ. Relative skeletal muscle mass is an important factor in non-alcoholic fatty liver disease in nonobese children and adolescents. J Clin Med 2020;9:3355.
[Article] [PubMed] [PMC]47. Balakrishnan R, Thurmond DC. Mechanisms by which skeletal muscle myokines ameliorate insulin resistance. Int J Mol Sci 2022;23:4636.
[Article] [PubMed] [PMC]48. Aljilani B, Tsintzas K, Jacques M, Radford S, Moran GW. Systematic review: sarcopenia in paediatric inflammatory bowel disease. Clin Nutr ESPEN 2023;57:647–54.
[Article] [PubMed]49. Aljilani B, Tsintzas K, Siervo M, Moran GW. Association between body mass index and age of disease onset with clinical outcomes in paediatric-onset Crohn's Disease (CD): a UK nation-wide analyses using the NIHR-IBD BioResource. Eur J Clin Nutr 2024;78:534–40.
[Article] [PubMed] [PMC]50. Blagec P, Sara S, Tripalo Batoš A, Trivić Mažuranić I, Močić Pavić A, Mišak Z, et al. Magnetic resonance imaging can be used to assess sarcopenia in children with newly diagnosed Crohn's disease. Nutrients 2023;15:3838.
[Article] [PubMed] [PMC]51. Pedersen M, Cromwell J, Nau P. Sarcopenia is a predictor of surgical morbidity in inflammatory bowel disease. Inflamm Bowel Dis 2017;23:1867–72.
[Article] [PubMed]52. Thangarajah D, Hyde MJ, Konteti VK, Santhakumaran S, Frost G, Fell JM. Systematic review: body composition in children with inflammatory bowel disease. Aliment Pharmacol Ther 2015;42:142–57.
[Article] [PubMed]53. Atlan L, Cohen S, Shiran S, Sira LB, Pratt LT, Yerushalmy-Feler A. Sarcopenia is a predictor for adverse clinical outcome in pediatric inflammatory bowel disease. J Pediatr Gastroenterol Nutr 2021;72:883–8.
[Article] [PubMed]54. Cucinotta U, Romano C, Dipasquale V. Diet and nutrition in pediatric inflammatory bowel diseases. Nutrients 2021;13:655.
[Article] [PubMed] [PMC]55. Damanti S, Senini E, De Lorenzo R, Merolla A, Santoro S, Festorazzi C, et al. Acute sarcopenia: mechanisms and management. Nutrients 2024;16:3428.
[Article] [PubMed] [PMC]56. López JJ, Cooper JN, Albert B, Adler B, King D, Minneci PC. Sarcopenia in children with perforated appendicitis. J Surg Res 2017;220:1–5.
[Article] [PubMed]57. Vanhorebeek I, Latronico N, Van den Berghe G. ICU-acquired weakness. Intensive Care Med 2020;46:637–53.
[Article] [PubMed] [PMC]58. Boster JM, Goodrich NP, Spino C, Loomes KM, Alonso EM, Kamath BM, et al. Sarcopenia is associated with osteopenia and impaired quality of life in children with genetic intrahepatic cholestatic liver disease. Hepatol Commun 2023;7:e0293.
[Article] [PubMed] [PMC]59. Becchetti C, Dajti E, Su GL, Labenz C, Colecchia A, Siramolpiwat S, et al. The impact of sarcopenia and frailty on decompensation in compensated cirrhosis: a systematic review. Hepatol Commun 2025;9:e0811.
[Article] [PubMed] [PMC]60. Boster JM, Feldman AG, Mack CL, Sokol RJ, Sundaram SS. Malnutrition in biliary atresia: assessment, management, and outcomes. Liver Transpl 2022;28:483–92.
[Article] [PubMed] [PMC]61. Yanai H, Adachi H, Hakoshima M, Iida S, Katsuyama H. Metabolic-dysfunction-associated steatotic liver diseaseits pathophysiology, association with atherosclerosis and cardiovascular disease, and treatments. Int J Mol Sci 2023;24:15473.
[Article] [PubMed] [PMC]62. Li X, He J, Sun Q. The prevalence and effects of sarcopenia in patients with metabolic dysfunction-associated steatotic liver disease (MASLD): a systematic review and metaanalysis. Clin Nutr 2024;43:2005–16.
[Article] [PubMed]63. Hong S, Chang Y, Jung HS, Yun KE, Shin H, Ryu S. Relative muscle mass and the risk of incident type 2 diabetes: a cohort study. PLoS One 2017;12:e0188650.
[Article] [PubMed] [PMC]64. Srikanthan P, Hevener AL, Karlamangla AS. Sarcopenia exacerbates obesity-associated insulin resistance and dysglycemia: findings from the National Health and Nutrition Examination Survey III. PLoS One 2010;5:e10805.
[Article] [PubMed] [PMC]65. Gold SL, Raman M, Sands BE, Ungaro R, Sabino J. Review article: putting some muscle into sarcopenia-the pathogenesis, assessment and clinical impact of muscle loss in patients with inflammatory bowel disease. Aliment Pharmacol Ther 2023;57:1216–30.
[Article] [PubMed]66. Liu S, Ding X, Maggiore G, Pietrobattista A, Satapathy SK, Tian Z, et al. Sarcopenia is associated with poor clinical outcomes in patients with inflammatory bowel disease: a prospective cohort study. Ann Transl Med 2022;10:367.
[Article] [PubMed] [PMC]67. Dasarathy S, Merli M. Sarcopenia from mechanism to diagnosis and treatment in liver disease. J Hepatol 2016;65:1232–44.
[Article] [PubMed] [PMC]68. Wang J, Maldonado MA. The ubiquitin-proteasome system and its role in inflammatory and autoimmune diseases. Cell Mol Immunol 2006;3:255–61.
[PubMed]69. Ferreira PVALS, Cavalcanti AS, Silva GAPD. Linear growth and bone metabolism in pediatric patients with inflammatory bowel disease. J Pediatr (Rio J) 2019;95 Suppl 1:59–65.
[Article] [PubMed]70. Wong SC, Dobie R, Altowati MA, Werther GA, Farquharson C, Ahmed SF. Growth and the growth hormone-insulin like growth factor 1 axis in children with chronic inflammation: current evidence, gaps in knowledge, and future directions. Endocr Rev 2016;37:62–110.
[Article] [PubMed]71. Su Y, Yuan D, Chen DG, Ng RH, Wang K, Choi J, et al. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell 2022;185:881. –95. e20.
[PubMed] [PMC]72. European Association for the Study of the Liver. EASL Clinical Practice Guidelines on nutrition in chronic liver disease. J Hepatol 2019;70:172–93.
[Article] [PubMed] [PMC]73. Tessitore M, Sorrentino E, Schiano Di Cola G, Colucci A, Vajro P, Mandato C. Malnutrition in pediatric chronic cholestatic disease: an up-to-date overview. Nutrients 2021;13:2785.
[Article] [PubMed] [PMC]74. Plank LD, Gane EJ, Peng S, Muthu C, Mathur S, Gillanders L, et al. Nocturnal nutritional supplementation improves total body protein status of patients with liver cirrhosis: a randomized 12-month trial. Hepatology 2008;48:557–66.
[PubMed]75. Dasarathy S, Hatzoglou M. Hyperammonemia and proteostasis in cirrhosis. Curr Opin Clin Nutr Metab Care 2018;21:30–6.
[Article] [PubMed] [PMC]76. Deng N, Mallepally N, Peng FB, Kanji A, Marcelli M, Hernaez R. Serum testosterone levels and testosterone supplementation in cirrhosis: a systematic review. Liver Int 2021;41:2358–70.
[Article] [PubMed]77. Henin G, Loumaye A, Deldicque L, Leclercq IA, Lanthier N. Unlocking liver health: can tackling myosteatosis spark remission in metabolic dysfunction-associated steatotic liver disease? Liver Int 2024;44:1781–96.
[Article] [PubMed]78. Permpoon U, Moon J, Kim CY, Nam TG. Glucocorticoid-mediated skeletal muscle atrophy: molecular mechanisms and potential therapeutic targets. Int J Mol Sci 2025;26:7616.
[Article] [PubMed] [PMC]79. Dirks ML, Wall BT, van de Valk B, Holloway TM, Holloway GP, Chabowski A, et al. One week of bed rest leads to substantial muscle atrophy and induces whole-body insulin resistance in the absence of skeletal muscle lipid accumulation. Diabetes 2016;65:2862–75.
[Article] [PubMed]80. Ooi PH, Mazurak VC, Siminoski K, Bhargava R, Yap JYK, Gilmour SM, et al. Deficits in muscle strength and physical performance influence physical activity in sarcopenic children after liver transplantation. Liver Transpl 2020;26:537–48.
[Article] [PubMed]81. Shimizu H, Nambu R, Tachibana N, Kunisaki R, Kudo T, Kato S, et al. Growth impairment in Japanese children with inflammatory bowel disease: a multicenter prospective cohort study. J Pediatr Gastroenterol Nutr 2025;81:1216–26.
[Article] [PubMed]82. Scott J, Yates M, Tanaka T, Ferrucci L, Cameron D, Welch AA. Cross-sectional associations between clinical biochemistry and nutritional biomarkers and sarcopenic indices of skeletal muscle in the baltimore longitudinal study of aging. J Nutr 2025;155:1535–48.
[Article] [PubMed] [PMC]83. Ota H, Fujimura T, Kunikata A, Yamagata T, Nozawa H, Ebishima H, et al. Clinical significance and normal reference value of serum creatinine to cystatin C ratio in children. Clin Nutr ESPEN 2024;62:278–84.
[Article] [PubMed]84. Martins MF, de Santa Maria NN, Dos Santos Camargo CC, Anjos DNP, de Carvalho EFT, de Freitas Dantas Gomes EL. Lower limit of normal of cross-sectional area of peripheral muscles and diaphragm measurements performed with ultrasound in full-term and preterm infants. Eur J Pediatr 2024;183:4799–806.
[Article] [PubMed]85. Valverde Montoro D, Rosa Camacho V, Artacho González L, Camacho Alonso JM. Thigh ultrasound monitoring identifies muscle atrophy in mechanically ventilated pediatric patients. Eur J Pediatr 2023;182:5543–51.
[Article] [PubMed]86. Formenti P, Umbrello M, Coppola S, Froio S, Chiumello D. Clinical review: peripheral muscular ultrasound in the ICU. Ann Intensive Care 2019;9:57.
[Article] [PubMed] [PMC]87. Wen Z, Gu J, Chen R, Wang Q, Ding N, Meng L, et al. Handgrip strength and muscle quality: results from the national health and nutrition examination survey database. J Clin Med 2023;12:3184.
[Article] [PubMed] [PMC]88. Marmol-Perez A, Ubago-Guisado E, Gil-Cosano JJ, Llorente-Cantarero FJ, Pascual-Gázquez JF, Muñoz-Torres M, et al. Co-morbid sarcopenia and low bone mineral density in young paediatric cancer survivors. J Cachexia Sarcopenia Muscle 2024;15:2156–63.
[Article] [PubMed] [PMC]89. Beaudart C, Rolland Y, Cruz-Jentoft AJ, Bauer JM, Sieber C, Cooper C, et al. Assessment of Muscle function and physical performance in daily clinical practice : a position paper endorsed by the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO). Calcif Tissue Int 2019;105:1–14.
[Article] [PubMed]90. Almeida KM, Dutra MV, Mello RR, Reis AB, Martins PS. Concurrent validity and reliability of the Alberta Infant Motor Scale in premature infants. J Pediatr (Rio J) 2008;84:442–8.
[Article] [PubMed]91. Nevill AM, Lang JJ, Niemz M, Tomkinson GR. How should youth handgrip strength be normalized? New insights using 3-D allometry with "generalizable" norm-referenced values, data from NHANES. Sports Med 2025;55:2303–12.
[Article] [PubMed] [PMC]92. Ortega FB, Leskošek B, Blagus R, Gil-Cosano JJ, Mäestu J, Tomkinson GR, et al. European fitness landscape for children and adolescents: updated reference values, fitness maps and country rankings based on nearly 8 million test results from 34 countries gathered by the FitBack network. Br J Sports Med 2023;57:299–310.
[Article] [PubMed] [PMC]93. Mehta NM, Skillman HE, Irving SY, Coss-Bu JA, Vermilyea S, Farrington EA, et al. Guidelines for the provision and assessment of nutrition support therapy in the Pediatric Critically Ill Patient: Society of Critical Care Medicine and American Society for Parenteral and Enteral Nutrition. JPEN J Parenter Enteral Nutr 2017;41:706–42.
[Article] [PubMed]94. Mehta NM, Bechard LJ, Zurakowski D, Duggan CP, Heyland DK. Adequate enteral protein intake is inversely associated with 60-d mortality in critically ill children: a multicenter, prospective, cohort study. Am J Clin Nutr 2015;102:199–206.
[Article] [PubMed] [PMC]95. Robinson DT, Calkins KL, Chen Y, Cober MP, Falciglia GH, Church DD, et al. Guidelines for parenteral nutrition in preterm infants: The American Society for Parenteral and Enteral Nutrition. JPEN J Parenter Enteral Nutr 2023;47:830–58.
[Article] [PubMed]96. Mariotti Zani E, Grandinetti R, Cunico D, Torelli L, Fainardi V, Pisi G, et al. Nutritional care in children with cystic fibrosis. Nutrients 2023;15:479.
[Article] [PubMed] [PMC]97. Yodoshi T, Orkin S, Arce-Clachar AC, Bramlage K, Liu C, Fei L, et al. Vitamin D deficiency: prevalence and association with liver disease severity in pediatric nonalcoholic fatty liver disease. Eur J Clin Nutr 2020;74:427–35.
[Article] [PubMed] [PMC]98. Pludowski P, Takacs I, Boyanov M, Belaya Z, Diaconu CC, Mokhort T, et al. Clinical practice in the prevention, diagnosis and treatment of vitamin D deficiency: a Central and Eastern European Expert Consensus Statement. Nutrients 2022;14:1483.
[Article] [PubMed] [PMC]99. Wackerhage H, Schoenfeld BJ, Hamilton DL, Lehti M, Hulmi JJ. Stimuli and sensors that initiate skeletal muscle hypertrophy following resistance exercise. J Appl Physiol (1985) 2019;126:30–43.
[Article] [PubMed]100. Goodman CA. Role of mTORC1 in mechanically induced increases in translation and skeletal muscle mass. J Appl Physiol (1985) 2019;127:581–90.
[Article] [PubMed]101. Janaudis-Ferreira T, Mathur S, Deliva R, Howes N, Patterson C, Räkel A, et al. Exercise for solid organ transplant candidates and recipients: a joint position statement of the Canadian Society of Transplantation and CAN-RESTORE. Transplantation 2019;103:e220–38.
[Article] [PubMed]102. Morales JS, Valenzuela PL, Velázquez-Díaz D, Castillo-García A, Jiménez-Pavón D, Lucia A, et al. Exercise and childhood cancer-a historical review. Cancers (Basel) 2021;14:82.
[Article] [PubMed] [PMC]103. Billany RE, Macdonald JH, Burns S, Chowdhury R, Ford EC, Mubaarak Z, et al. A structured, home-based exercise programme in kidney transplant recipients (ECSERT): a randomised controlled feasibility study. PLoS One 2025;20:e0316031.
[Article] [PubMed] [PMC]104. Ma DD, Liu Z, Au K, Tran M, Artuz CM, Greenwood M, et al. Randomized controlled trial of a virtually delivered exercise and stress management program to improve physical performance of hematopoietic cell transplant survivors. J Clin Oncol 2025;43:949–59.
[Article] [PubMed] [PMC]105. Hickmann CE, Castanares-Zapatero D, Bialais E, Dugernier J, Tordeur A, Colmant L, et al. Teamwork enables high level of early mobilization in critically ill patients. Ann Intensive Care 2016;6:80.
[Article] [PubMed] [PMC]106. Cortes OL, Delgado S, Esparza M. Systematic review and meta-analysis of experimental studies: In-hospital mobilization for patients admitted for medical treatment. J Adv Nurs 2019;75:1823–37.
[Article] [PubMed]107. Hurst C, Robinson SM, Witham MD, Dodds RM, Granic A, Buckland C, et al. Resistance exercise as a treatment for sarcopenia: prescription and delivery. Age Ageing 2022;51:afac003.
[Article] [PubMed] [PMC]108. Chen N, He X, Feng Y, Ainsworth BE, Liu Y. Effects of resistance training in healthy older people with sarcopenia: a systematic review and meta-analysis of randomized controlled trials. Eur Rev Aging Phys Act 2021;18:23.
[Article] [PubMed] [PMC]109. Liao CD, Huang SW, Chen HC, Huang MH, Liou TH, Lin CL. Comparative efficacy of different protein supplements on muscle mass, strength, and physical indices of sarcopenia among community-dwelling, hospitalized or institutionalized older adults undergoing resistance training: a network meta-analysis of randomized controlled trials. Nutrients 2024;16:941.
[Article] [PubMed] [PMC]110. Sañudo B, Sánchez-Oliver AJ, Fernández-Gavira J, Gaser D, Stöcker N, Peralta M, et al. Physical and psychosocial benefits of sports participation among children and adolescents with chronic diseases: a systematic review. Sports Med Open 2024;10:54.
[PubMed] [PMC]111. Stöcker N, Gaser D, Oberhoffer-Fritz R, Sitzberger C. KidsTUMove-A holistic program for children with chronic diseases, increasing physical activity and mental health. J Clin Med 2024;13:3791.
[Article] [PubMed] [PMC]112. Albanese A, Kewley GD, Long A, Pearl KN, Robins DG, Stanhope R. Oral treatment for constitutional delay of growth and puberty in boys: a randomised trial of an anabolic steroid or testosterone undecanoate. Arch Dis Child 1994;71:315–7.
[Article] [PubMed] [PMC]113. Grimberg A, DiVall SA, Polychronakos C, Allen DB, Cohen LE, Quintos JB, et al. Guidelines for growth hormone and insulin-like growth factor-i treatment in children and adolescents: growth hormone deficiency, idiopathic short stature, and primary insulin-like growth factor-I deficiency. Horm Res Paediatr 2016;86:361–97.
[Article] [PubMed]114. Liu M, Luo J, Zhou J, Zhu X. Intervention effect of neuromuscular electrical stimulation on ICU acquired weakness: a meta-analysis. Int J Nurs Sci 2020;7:228–37.
[Article] [PubMed] [PMC]115. Maffiuletti NA, Roig M, Karatzanos E, Nanas S. Neuromuscular electrical stimulation for preventing skeletalmuscle weakness and wasting in critically ill patients: a systematic review. BMC Med 2013;11:137.
[Article] [PubMed] [PMC]116. Elnaggar RK, Mahmoud WS, Abdrabo MS, Elfakharany MS. Effect of adaptive variable-resistance training on chemotherapy-induced sarcopenia, fatigue, and functional restriction in pediatric survivors of acute lymphoblastic leukemia: a prospective randomized controlled trial. Support Care Cancer 2025;33:214.
[Article] [PubMed]

 About
About Browse articles
Browse articles For contributors
For contributors


