Increased procalcitonin level is a risk factor for prolonged fever in children with Mycoplasma pneumonia
Article information
Abstract
Purpose
Macrolide-resistant Mycoplasma pneumoniae pneumonia (MPP) is characterized by prolonged fever and radiological progression despite macrolide treatment. Few studies have examined serum procalcitonin (PCT) level in children with MPP. We aimed to investigate the association of acute inflammation markers including PCT with clinical parameters in children with MPP.
Methods
A total of 147 children were recruited. The diagnosis of MPP relied on serial measurement of IgM antibody against mycoplasma and/or polymerase chain reaction. We evaluated the relationships between C-reactive protein (CRP), PCT, and lactate dehydrogenase (LDH) levels and white blood cell (WBC) counts, and clinical severity of the disease. We used multivariate logistic regression analysis to estimate the odds ratio for prolonged fever (>3 days after admission) and hospital stay (> 6 days), comparing quintiles 2–5 of the PCT levels with the lowest quintile.
Results
The serum PCT and CRP levels were higher in children with fever and hospital stay than in those with fever lasting ≤ 3 days after admission and hospital stay ≤ 6 days. CRP level was higher in segmental/lobar pneumonia than in bronchopneumonia. The LDH level and WBC counts were higher in children with fever lasting for >3 days before compared to those with fever lasting for ≤ 3 days. The highest quintile of PCT levels was associated with a significantly higher risk of prolonged fever and/or hospital stay than the lowest quintile.
Conclusion
Serum PCT and CRP levels on admission day were associated with persistent fever and longer hospitalization in children with MPP.
Introduction
Mycoplasma pneumoniae pneumonia (MPP) is one of the most common community-acquired pneumonia (CAP) in children and adolescents [1]. Even though MPP is usually mild and self-limiting, it can progress into a life-threatening, fulminant pneumonia [2-4]. Outbreaks of MPP occur in 3- to 4-year intervals worldwide [5], and the most recent epidemics in Korea happened in 2006–2007, 2011, and 2015–2016 [6].
Macrolides have been used as a first-line antibiotic treatment in children with MPP. However, since 2003, macrolide-resistant MPP (MRMP) has become more common, especially in East Asian countries such as Korea, China, and Japan [7,8]. The prevalence of MRMP in Korea reached up to 84.6% in 2015 [9-11]. Fever is considered an important indicator of antibiotic responsiveness in CAP. It has been reported that fever lasts longer in patients with MRMP than in patients with macrolide-susceptible MPP during treatment with macrolides. Some studies reported higher laboratory values, in particular for lactate dehydrogenase (LDH) and C-reactive protein (CRP), in patients with MRMP [12,13]. Even though several clinical studies aimed to identify an indicator for MRMP, no consistent results have been obtained.
Procalcitonin (PCT), a precursor of thyroid hormone, has been used as a biomarker of bacterial infection. Serum PCT level increases within 2–4 hours and peaks within 6–24 hours of systemic infection, earlier than CRP level [14]. PCT concentration parallels the severity of infection [15]. This may help distinguish a severe bacterial infection from a milder bacterial or viral infection. Many studies have investigated the relationship of serum PCT level with pneumonia, which may be instrumental in establishing diagnosis, determining prognosis, and guiding the decision to administer an empirical antibiotic therapy or to assess the effectiveness of antibiotics [16,17]. However, only a few studies have been conducted on PCT levels in children with MPP [18]. We measured the levels of acute inflammatory markers, such as PCT, LDH, and CRP, and white blood cell (WBC) counts in patients with MPP and investigated the relationships between these parameters and attributes of clinical severity, including pneumonia type, fever duration, and hospitalization.
Materials and methods
1. Study subjects
This prospective cross-sectional study was performed from August 2015 to March 2016 in the Pediatric Department, Kangbuk Samsung Hospital, Seoul, Korea. We enrolled 147 children who were admitted to the hospital because of CAP and diagnosed with MPP. The diagnosis of CAP was based on clinical symptoms and signs of lower respiratory infection, as well as on chest radiography findings. MPP was defined as follows: (1) positive serum IgM antibody against mycoplasma, with an increase in optical density (OD) or seroconversion of IgM antibody from negative to positive values after 4–7 days, and/or (2) positive results of polymerase chain reaction (PCR) for mycoplasma in samples from nasopharyngeal aspiration or sputum samples. Children who had a history of tuberculosis, chronic pulmonary diseases, congenital heart diseases, immunodeficiency, immunosuppressive conditions or medications, or congenital anomalies were excluded from the study.
All the children were treated with oral azithromycin from the first day of admission. We examined the patients’ age, sex, hospital stay, durations of fever before and after admission, chest radiographs, serum PCT, CRP, and LDH levels, and WBC counts. Prolonged fever was defined as persistent fever for more than 3 days after admission, and prolonged hospital stay was defined as hospitalization for more than 6 days. Pneumonia was classified as bronchopneumonia and segmental/lobar pneumonia by 2 radiologists.
The study protocol was approved by the Institutional Review Board of the Kangbuk Samsung Hospital (KBSMC201509017003-HE003), and informed consent was obtained from the children’s guardians.
2. Laboratory tests
Blood tests for measuring peripheral WBC counts, and CRP, LDH, and mycoplasma IgM antibody levels were performed on admission. A blood test for measuring mycoplasma IgM antibody level was performed within 7 days after admission for diagnosing MPP.
A serum sample for PCT level determination was collected on admission and stored at -20°C until analyzed. Serum PCT level was measured on a Roche Cobas e601 analyzer based on an electrochemiluminescence immunoassay using the Elecsys BRAHMS PCT kit (Roche Diagnostics Korea, Seoul, Korea). The assay detection range was 0.02–100 ng/mL.
Serum mycoplasma IgM level was assayed using an enzyme immunoassay (RIDASCREEN; R-Biopharm AG, Darmstadt, Germany). The cutoff level for mycoplasma IgM antibody level was 71 U/mL. Mycoplasma PCR of sputum or nasopharyngeal aspiration samples was performed using the Bio-Core M. pneumoniae PCR Kit (Seegene, Seoul, Korea). The mycoplasma PCR was performed in 2 patients, and those patients showed positive results in both PCR and serology tests. PCR was not performed in the remaining patients, and the diagnosis was established using the enzyme immunoassay (EIA) method.
3. Statistical analyses
Statistical analyses were performed using Stata ver. 14.2 (Stata Corp LLC, College Station, TX, USA). Mean values were compared between 2 groups using the Student t test. Significance of differences between groups in categorical variables was tested using chi-square analysis. For nonnormally distributed data, a Mann-Whitney test was performed. We used a multivariate logistic regression analysis to estimate odds ratio (OR) for prolonged fever duration and hospital stay; quintiles 2–5 of serum PCT, CRP, and LDH levels were compared to the lowest quintiles after adjusting for age and sex. All data are expressed as mean±standard deviation or median and interquartile range (IQR), and significance was defined as a P value of <0.05.
Results
1. Patient characteristics
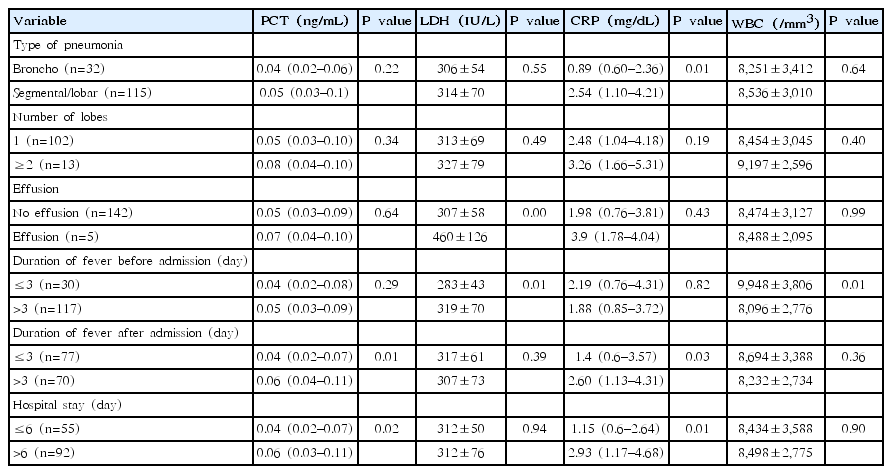
The baseline characteristics of the study subjects are presented in Table 1. We included 147 children in this study. The mean age was 5.9 years (IQR, 3–8 years), and boys comprised 52.4% of the subjects. Serum of all the 147 patients was positive for IgM antibody against mycoplasma. PCR analysis detected mycoplasma in two patients. The mean durations of fever before and after admission were 4.6 and 2.7 days, respectively. The mean hospital stay was 6.7 days (IQR, 5–8 days). The mean LDH level was 312 IU/L, the median of CRP level was 1.99 mg/dL, and the PCT level was 0.05 ng/mL. The prevalence rates of segmental/lobar pneumonia and parapneumonic effusion were 78.8% and 3.4%, respectively. The percentage of children who had fever for >7 days before admission was 24.5%. In total, 47.6% of the children had prolonged fever after admission. For patients who had persistent fever and no improvement on chest X-ray radiography, the antibiotic was changed to levofloxacin (6 cases) and corticosteroids were added (110 cases). (Data not shown in the table)
2. Serum levels of PCT, LDH, and CRP and WBC counts according to fever duration, hospital stay length, and types of pneumonia
We compared serum PCT, LDH, and CRP levels and WBC counts between patients with different types of pneumonia, durations of fever before and after admission, and lengths of hospital stay. Since serum PCT and CRP levels were not distributed normally, median values and IQRs were used instead of means and standard deviations, and the Mann-Whitney test was employed for statistical analysis. We found no significant differences in serum PCT levels according to type of pneumonia, number of lobes involved, presence of effusion, or duration of fever before admission. However, the mean serum PCT level was higher in children with prolonged fever (>3 days after admission) than in those with fever lasting for ≤3 days after admission (P=0.01). In addition, children who were admitted to the hospital for >6 days had higher serum PCT levels than those who stayed in the hospital for ≤6 days (P=0.02). Serum CRP levels were significantly increased in children with segmental/lobar pneumonia compared to those with bronchopneumonia. Patients with prolonged fever (>3 days) and longer hospitalization (>6 days) had higher CRP levels (P=0.03 and P=0.01, respectively). In contrast, serum LDH levels and WBC counts were higher in children who had fever for >3 days before admission (P=0.01). However, no significant differences were found in serum LDH levels and WBC counts in children with different fever durations after admission or lengths of hospital stay (Table 2).
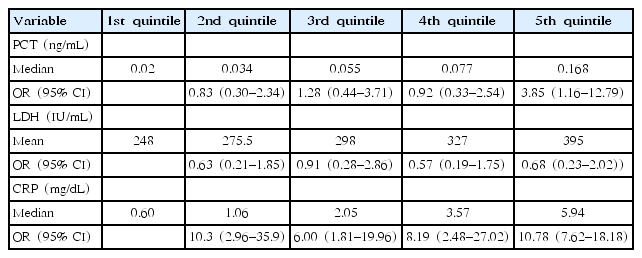
3. Logistic regression analysis of serum PCT, LDH, and CRP levels for prolonged fever and hospital stay length
We assessed serum PCT, LDH, and CRP levels for prolonged fever (Table 3) and hospital stay length (Table 4) by using a multivariate logistic regression analysis. The highest quintile of PCT and CRP levels was associated with prolonged fever (OR, 3.69; 95% confidence interval [CI], 1.27–10.7 and OR, 4.03; 95% CI, 1.21–13.34, respectively) and hospital stay length (OR, 3.85; 95% CI, 1.16–12.79 and OR, 10.78; 95% CI, 7.62–18.18, respectively) as compared with the lowest quintile. However, we found no association between serum LDH level on admission and prolonged fever or hospital stay length.

Logistic regression of serum procalcitonin and lactate dehydrogenase for prolonged fever more than 3 days after admission
Discussion
In the present study, we found that patients with prolonged fever after admission (defined as persistent fever for >3 days after initiation of macrolide therapy) and longer hospitalization (defined as hospital stay of >6 days) had higher serum PCT and CRP levels on admission. Although the increasing incidence of MRMP is believed to be a major cause of severe morbidity, refractory pneumonia is caused not only by MRMP but also excessive host immune reaction against the insults from the MP infection [19]. It has been suggested that not only pathogens but also excessive host immune reaction are responsible for acute lung injury. Thus, early administration of immunomodulators such as corticosteroids or intravenous immunoglobulin may induce a rapid improvement of clinical symptoms and radiological signs in refractory MPP [20]. We treated the majority of patients who had fever for >3 days after initiation of macrolide therapy with corticosteroids, and small percentage of patients were treated with levofloxacin, as described in the Result section.
Emerging MRMP is associated with mutations at sites 2063, 2064, 2067, and 2617 in domain V of the 23S rRNA sequence [9,21]. Some patients with MRMP present with persistent fever, have a prolonged hospital stay, and can have a high risk of complications [22,23]. Therefore, biomarkers to predict refractory MPP can be useful for making early decisions in therapeutic planning.
Serum PCT level has been studied as an early diagnostic indicator to predict the progression of bacterial infections or sepsis [24]. Among other markers such as CRP level and WBC count, serum PCT level has been considered the most useful marker for differentiation of bacterial pneumonia [25]. Among adult patients with CAP, those with high pneumonia severity indices have high PCT levels. A recent study of serum PCT levels in hospitalized children with CAP suggested that low PCT concentrations (<0.1 ng/mL) were not associated with a typical bacterial pneumonia, which may help reduce the need for antibiotic treatments [26]. However, studies of PCT levels in pneumonia mostly investigate associations with typical bacterial pathogens such as Streptococcus pneumoniae and Haemophilus influenzae [27], while PCT levels in MPP, especially in refractory cases, have not been thoroughly studied.
In this study, the median levels of PCT and CRP were 0.05 ng/mL and 1.99 mg/dL, respectively. This finding suggests that M. pneumoniae infection is more likely to be a viral infection than a bacterial infection. Furthermore, in PCT-guided antibiotic therapy, the use of antibiotics is discouraged in patients with PCT levels <0.1 ng/mL [15]. In this study, the serum PCT concentration was low, but all the samples were within the detectable range. Although low PCT levels can distinguish atypical pneumonia from typical bacterial pneumonia, MPP treatment requires the use of antibiotics.
MPP is mild and sometimes self-limiting disease, although fatal and life-threatening cases were reported [28]. Clinical and laboratory characteristics of fulminant mycoplasma pneumonia include severe cough, high fever, dyspnea, diffuse abnormal findings on chest X-ray radiography, and elevated serum CRP levels and WBC counts [29]. In this series, there were no patients with severe progressive pneumonia, but we found that the levels of PCT and CRP were higher in patients with pneumonia characterized by prolonged fever and a severe pneumonia pattern. Even though serum PCT and CRP levels were lower than the values typical of a bacterial infection, they may be useful for predicting severe cases of MPP.
A key determining factor of refractory MPP is persistent fever and radiological progression after antibiotic therapy [22]. In our study, serum PCT level on admission was associated with persistent fever and longer hospital stay. Patients with antibiotic-resistant infections require significantly longer hospital stay; data show that the duration of hospital stay for patients with antibiotic-resistant infections was prolonged by 6.4 to 12.7 days [29]. Studies on refractory MPP have shown an increase in serum levels of LDH, CRP, IL-6, or IL-18 [12,13,30]. A recent study showed that excessive serum levels of LDH, CRP, and IL-6 might be early predictors of refractory pneumonia in children with MPP [31]. We also found that the serum LDH level was elevated in children with pleural effusion or those with persistent fever for >3 days before admission, suggesting that severity of lung injury was associated with LDH level. However, serum LDH level was not significantly elevated in the children with prolonged fever after admission or hospital stay. According to our study, serum LDH level on admission is probably not a relevant indicator of refractory pneumonia, in contrast to serum PCT level. There were few patients with refractory pneumonia in this series because of good response to second-line treatments.
The main strength of our study was the assessment of serum PCT levels in children with MPP and the analysis of the association with clinical responsiveness to macrolides. To our knowledge, no study has assessed serum PCT levels in MPP based on fever or hospitalization duration. Though low, the serum PCT concentrations in all the samples were within the detectable range. Low PCT levels can also distinguish atypical pneumonia from typical bacterial pneumonia and help choose suitable antibiotic treatments in children hospitalized for CAP.
Our study has limitations. First, we did not identify the point mutations characteristic of MRMP. Thus, the results cannot be generalized to all patients with MRMP. However, we analyzed serum PCT levels based on clinical responsiveness to antibiotics. Second, disease severity was not assessed, and, while measurement of serum PCT level may have been useful for detecting patients with severe MPP, it may have been less valuable in those with mild MPP. Third, the serum PCT levels were low, mainly because of the association of low PCT levels with atypical bacterial or viral pneumonia. However, the low PCT levels shown in this study could be useful for choosing adequate antibiotics or second-line treatments for children with antibiotic-unresponsive CAP.
In conclusion, laboratory parameters associated with inflammation, including PCT, CRP, and LDH levels and WBC counts, were elevated in MPP, with individual variations. Serum PCT level was not markedly increased, but repeated measurements of serum PCT level may be incorporated into treatment guidelines for children with refractory pneumonia since PCT level might reflect the severity of the disease.
Notes
This work used reagents from Roche Diagnostics for the measurement of PCT levels. The funding source had no role in the design, collection, analysis, interpretation of data, or writing of this manuscript.