Febrile seizures
Article information
Abstract
Febrile seizure (FS) is the most common seizure disorder of childhood, and occurs in an age-related manner. FS are classified into simple and complex. FS has a multifactorial inheritance, suggesting that both genetic and environmental factors are causative. Various animal models have elucidated the pathophysiological mechanisms of FS. Risk factors for a first FS are a family history of the disorder and a developmental delay. Risk factors for recurrent FS are a family history, age below 18 months at seizure onset, maximum temperature, and duration of fever. Risk factors for subsequent development of epilepsy are neurodevelopmental abnormality and complex FS. Clinicians evaluating children after a simple FS should concentrate on identifying the cause of the child's fever. Meningitis should be considered in the differential diagnosis for any febrile child. A simple FS does not usually require further evaluation such as ordering electroencephalography, neuroimaging, or other studies. Treatment is acute rescue therapy for prolonged FS. Antipyretics are not proven to reduce the recurrence risk for FS. Some evidence shows that both intermittent therapy with oral/rectal diazepam and continuous prophylaxis with oral phenobarbital or valproate are effective in reducing the risk of recurrence, but there is no evidence that these medications reduce the risk of subsequent epilepsy. Vaccine-induced FS is a rare event that does not lead to deleterious outcomes, but could affect patient and physician attitudes toward the safety of vaccination.
Introduction
Febrile seizure (FS) is the most common type of childhood seizure disorder, which occurs in an age-specific manner, is associated with a fever of 38.0℃ or higher, and presents without evidence of any definite causative diseases, such as central nervous system (CNS) infection or metabolic abnormality1,2). Most cases of FS are benign and self-limiting, and in general, treatment is not recommend3). FS has been defined differently by the National Institutes of Health (NIH), the International League against Epilepsy (ILAE), and the American Academy of Pediatrics (AAP). The NIH (1980) defined FS as follows: an abnormal, sudden, excessive electrical discharge of neurons (gray matter) that propagates down the neuronal processes (white matter) to affect an end organ in a clinically measurable fashion, occurring in infancy or childhood, usually between 3 months and 5 years of age, associated with fever, but without evidence of intracranial infection or defined cause4). The ILAE (1993) defined FS as a seizure occurring in childhood after age 1 month, associated with a febrile illness not caused by infection of the CNS, without previous neonatal seizures or a previous unprovoked seizure, and not meeting the criteria of other acute symptomatic seizures5). Most recently, the AAP (2008) defined FS as a seizure occurring in febrile children between the ages of 6 and 60 months who do not have an intracranial infection, metabolic disturbance, or history of afebrile seizure1). FS is considered a "syndrome" because it fulfills several characteristics that are similar among affected children: (1) FS generally occurs within a restricted age range; (2) the majority of children with FS show normal neurological and structural development after the episode; and (3) FS is not associated with structural or developmental anomalies in the brain, although the existence of such pathology may enhance susceptibility to FS6). Genetics, comorbidities (premature birth, fetal growth retardation), and environmental risk factors (exposure to nicotine in utero, or antihistamine use) may increase risk of FS in addition to the age factor7,8).
Simple and complex FS-definition/terminology
Livingston et al.9) first introduced the term "simple febrile convulsion" and "epileptic seizures precipitated by fever" to designate two groups on the basis of age of onset, seizure characteristics, electroencephalography (EEG) findings, frequency of seizures, and genetic factors. "Febrile seizures" can rationally be distinguished from "seizures with fever." The latter includes any convulsion in any child with a fever of any cause. Thus, children with seizures and fever, both with definite causes such as CNS infections or overt neurological disorders, have "seizures with fever" rather than "febrile seizures"10).
Recently, clinicians have begun classifying FS as either simple or complex. Simple FS is defined as generalized, lasting less than 15 minutes, comprised of generalized tonic and clonic activity without a focal component, and without recurrence within 24 hours or within the same febrile illness1). Complex or complicated FS is defined as exhibiting one or more of the following features: (1) partial onset or focal features; (2) prolonged duration of more than 15 minutes; (3) recurrent febrile seizure within 24 hours of the first episode; and (4) association with postictal neurological abnormalities, as exemplified by Todd paresis10,11).
Incidence and prevalence
FSs occur in 2% to 5% of children 6 months to 5 years of age1). The peak incidence occurs at approximately 18 months of age and is low before 6 months or after 3 years of age2). Generally, the incidence of FS decreases markedly after 4 years of age (and the condition rarely occurs in children older than 7 years of age2,12). FS occurs more frequently in the Asian population, affecting 3.4%-9.3% of Japanese children2) and 5%-10% of Indian children, but only 2%-5% of children in the United States (US) and Western Europe. The highest prevalence is 14% in Guam13). Unfortunately, there is no epidemiological study in Korean children.
Males have consistently emerged as having a higher frequency of FS (male to female ratio, 1.1:1 to 2:1). However, some large studies have shown no significant gender difference14).
There are two seasonal peaks in FS incidence: November-January, corresponding to the peak of viral upper respiratory infection, and June-August, when common viral gastrointestinal illnesses occur14). Variation in prevalence is related to differences in case definitions, ascertainment methods, geography, and cultural factors15).
In a study of children with a first FS, most seizures were simple, and at least one complex feature was noted in approximately 35% of cases, including features of focality (16.1%), multiple seizures (13.8%), prolonged duration (>15 minutes, 9.3%) and recurrent febrile seizure within 24 hours (16.2%); 6.5% showed two complex features, and 0.7% showed three complex features11). Febrile status epilepticus, that is, seizures that last more than 30 minutes, represents only 5% of FS16), and represents about 25% of all episodes of childhood status epilepticus with more than two thirds of cases occurring at 2 years of age17). Only 21% of children experience seizures either prior to or within 1 hour of the onset of fever; 57% have seizure after 1 to 24 hours of fever, and 22% experience febrile seizure more than 24 hours after the onset of fever18,19).
FS is mostly generalized and convulsive in character, but approximately 5% of FS cases have nonconvulsive features presenting with unconsciousness, staring, eye deviation, atonia, or cyanosis2).
Genetics
These seizures have a familial tendency in some cases and are sporadic in others, suggesting that both genetic and environmental elements contribute to their generation20). The importance of genetic factors in FS has long been recognized. Population studies have demonstrated that FS occurs at a much higher-than-expected incidence in first- and second-degree relatives of children with FS21). Family history also has a role in determining whether children have FS recurrences and subsequently develop afebrile seizures2,21). Twenty-five to 40% of patients showed a positive family history for FS; the incidence of FS being 20.7% among siblings, 10.9% among parents, and 14.1% among first-degree relatives of probands21). In a study done to compare these rates with those of control subjects, the incidence was 8.4% in siblings, 1.6% in parents, and 3.8% among first-degree relatives of the controls21). Overall, there appears to be a multifactorial mode of inheritance for febrile convulsions, but there may be a subset of children with an autosomal-dominant mode of inheritance22).
Five areas of the genome have shown to be linked to FS in some way. Two of them, FEB1 and FEB2 , found on chromosomes 8 and 19p, are only involved in FS. Three others involve "generalized epilepsy with FS+" (GEFS+) syndrome21,22).
GEFS+ patients present with complex febrile seizures, which are often seen beyond 5 years of age, and develop afebrile seizures later in childhood23).
These children are reported to have a variety of mutations in the α-subunit of the sodium channel (SCN1A and B)22,23), and in the γ2-subunit of the γ-aminobutyric acid type A (GABAA) receptor (GABRG2)22,24).
Pathophysiology
Although the mechanism of FS remains unclear, animal models are informative25). First, elevated brain temperature alters many neuronal functions, including several temperature-sensitive ion channels26). This influences neuronal firing and increases the probability of generating massive neuronal activity, i.e., seizures. Also, an inflammatory process including secretion of cytokine in the periphery and in the brain is known to be a part of the mechanism27). Second, it was discovered that fever and hyperthermia share common mechanisms in provoking seizures: the fever-promoting pyrogen interleukin-1β contributes to fever generation and conversely, fever leads to the synthesis of this cytokine in the hippocampus28,29). In addition, interleukin-1β has been shown to increase neuronal excitability, acting via both glutamate and GABA30). In vivo, these actions of interleukin-1β enhance the actions of seizure-provoking agents31). The importance of endogenous interleukin-1β in the occurrence of FS was supported by studies in mice that lacked the receptor for this cytokine31). Fever of specific infectious etiologies, specifically human herpes virus 6 (HHV6), might influence the probability of generation of FS31,32). Third, hyperthermia-induced hyperventilation and alkalosis have been proposed as a pivotal element of FS generation in that alkalosis of the brain provokes neuronal excitability33) and contributes to seizure pathophysiology. However, human conditions associated with severe alkalosis, including prolonged crying and pyloric stenosis of infants, are not associated with the generation of seizures25).
Risk factors for recurrence and subsequent epilepsy
1. Risk factors for first FS
Two studies have examined the risk factors associated with FS34,35). In one study, four factors were associated with an increased risk of FS: (1) a first- or second-degree relative with a history of FS; (2) a neonatal nursery stay of more than 30 days; (3) developmental delay; and (4) day-care attendance. Children with more than two risk factors have a chance of developing FS in approximately 28%34). In another multivariable analysis, significant independent risk factors were peak temperature and history of FS in a first- or higher-degree relative35). Gastroenteritis as the underlying illness appeared to have a significant inverse (i.e., protective) association with FS12,35,36).
2. Risk factors for recurrent FS
Overall, approximately one-third of children with a first FS experience one or more recurrent FSs11) and 10% have three or more FSs18,19,37,38,39,40,41).
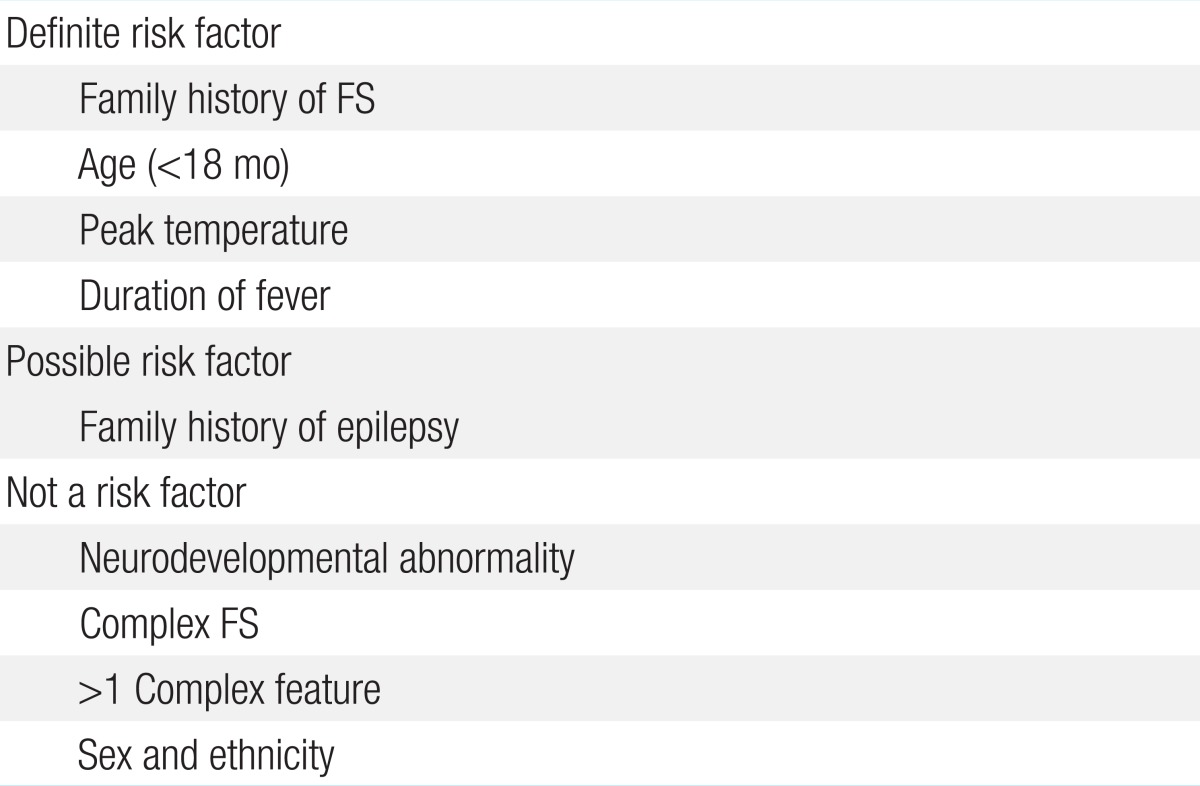
The risk factors potentially associated with FS recurrence are summarized in Table 1. The most consistent risk factors reported are a family history of FS and onset of first FS at less than 18 months of age12,18,19,37,38). Two other definite risk factors for recurrence of FS are peak temperature and the duration of fever prior to seizure18,19,38). The higher the peak temperature, the lower the chance of recurrence; children with a peak temperature of 101℉ had a 42% recurrence risk at 1 year, compared with 29% for those with a peak temperature of 103℉, and only 12% for those with a peak temperature >105℉
The shorter the duration of recognized fever, the higher the chance of recurrence12); the recurrence risk at 1 year was 46% in children who experienced FS within an hour of recognized onset of fever, compared to 25% in children with prior fever lasting 1 to 24 hours, and 15% in children with more than 24 hours of recognized fever prior to the FS12). Children with multiple risk factors have the highest risk of recurrence18). A child with two or more of the risk factors listed in Table 1 has a recurrence rate greater than 30% at 2 years; a child with three or more risk factors has a recurrence rate greater than 60%18). In contrast, recurrence risk is less than 15% for a 2-year-old child with none of the risk factors mentioned in Table 112,18). A recurrent FS is also more likely to be prolonged if the initial FS was prolonged11,41). The existence of any relationship between a family history of unprovoked seizures or epilepsy and the overall risk of FS recurrence appears to be doubtful. Some studies report a modest increase in the risk of FS recurrence in children with a family history of unprovoked seizures, but a large study in Rochester, Minnesota, found no difference in recurrence risk between children with a family history of epilepsy (25%) and those with no such family history (23%)37). The presence of a neurodevelopmental abnormality in the child or a history of complex FS have not been shown to be significantly associated with an increased risk of subsequent FSs18,19,37,38,41). Moreover, neither ethnicity nor sex associates with an increased risk of recurrent FSs12).
3. Risk factors for subsequent epilepsy
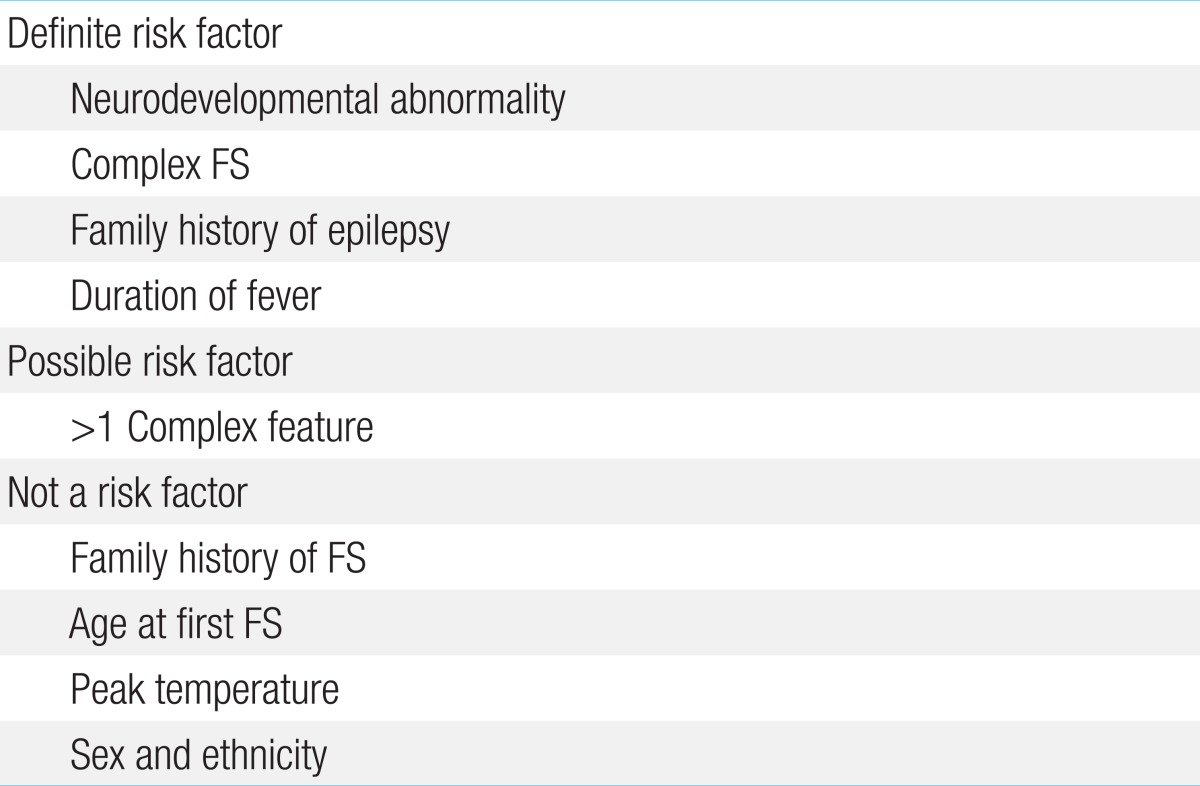
The risk factors for developing subsequent epilepsy after FS are summarized in Table 2. Following a simple FS, the risk of developing epilepsy is no different from that in the general population16,40). Some studies on children with FS indicate that 2% to 10% of children who have FS will subsequently develop epilepsy. However, a family history of epilepsy and the occurrence of a complex FS were found to be associated with increased risk of subsequent epilepsy16,40). Repeated bouts of simple FS under the age of 12 months lead to a slight increase in epilepsy risk42). The occurrence of multiple FSs was also associated with a slight but statistically significant increase in the risk of subsequent epilepsy in two additional studies11,16,43). One study found that children with a FS that occurred within 1 hour of a recognized fever (i.e., at onset) had a higher risk for subsequent epilepsy than children with FS associated with longer duration of fever40). Two studies have found that prolonged FSs (i.e., febrile status epilepticus) were associated with an increased risk of subsequent epilepsy compared to a complex FS that was less prolonged16,43). The number of complex features in a FS may possibly affect the risk of recurrence. Although one study found that patients with two complex features (e.g., prolonged and focal) had an elevated risk of subsequent epilepsy, another study did not detect this association16). A family history of FS, age at first FS, and the height of fever at first seizure are not associated with a differential risk of developing epilepsy16,40,43). The only common risk factor for both recurrent FSs and subsequent epilepsy was duration of fever prior to the FS16,18,19). The types of subsequent epilepsy that develop are variable; however, the types of epilepsy that occur in children with prior FS are not significantly different from those that occur in children without such a history12,20,43). It is controversial whether FS is simply an age-specific marker of future seizure susceptibility or if it has a causal relationship with subsequent epilepsy12). One study showed a 13% incidence of epilepsy caused by the presence of at least two of the following risk factors: (1) a family history of non-FSs; (2) abnormal neurologic or developmental status prior to FS; and (3) complex FS, such as a prolonged or focal seizure. Only 2 to 3% of children who have none or one of the above risk factors subsequently develop non-FSs4).
Evaluation
Children should be promptly evaluated after an initial seizure. Most parents of patients with FS seek medical care within an hour of the seizure, but after resolution of the seizure and return of the patient to full alertness44).
The initial evaluation should focus on determining the source of the fever44,45). History taking should include documentation of any family history of FSs or epilepsy, status of immunizations, recent antibiotic use, duration of the seizure, any prolonged postictal phase, and any focal symptoms. On physical examination, attention should be given to the presence of meningeal signs and the child's level of consciousness39).
To begin, one must consider whether there is an infection of the CNS in the form of meningitis or encephalitis, particularly in younger infants in whom the signs can be more subtle. Therefore, the important issue for evaluation is whether a lumbar puncture is necessary to exclude meningitis. If meningitis is excluded, the next step is to consider what tests are needed to determine the cause of the febrile illness. Finally, consider whether there is a structural CNS abnormality that predisposed the child to having a seizure13,46).
1. Lumbar puncture
Recommendations by the AAP (1966) for lumbar puncture (LP) in children with first simple FS were summarized as follows47): (1) In infants younger than 12 months, performance of an LP is strongly advised, because the clinical signs and symptoms associated with meningitis may be minimal or absent in this age group; (2) In a child between 12 and 18 months of age, an LP should be considered, because clinical signs and symptoms of meningitis may be subtle; (3) In a child older than 18 months, although an LP is not routinely warranted, it is recommended in the presence of meningeal signs and symptoms (i.e., neck stiffness and positive Kernig and Brudzinski signs); (4) In infants and children who have had FS and have received prior antibiotic treatment, clinicians should be aware that treatment might mask the signs and symptoms of meningitis. Therefore, an LP should be seriously considered. However, with the advent of Haemophilus influenzae type b (Hib) and Streptococcus pneumoniae conjugate vaccines in many countries, bacterial meningitis in children 6 months and older has become very rare42,48,49,50). In a retrospective review, children aged 6 to 18 months underwent an LP as part of the evaluation for a first simple FS48). Cerebrospinal fluid (CSF) pleocytosis was present in 10%, but no child had bacterial meningitis48). Another study assessed the rate of acute bacterial meningitis among 526 children aged 6 to 60 months, who presented with a first episode of complex FS. Only three of 14 children with CSF pleocytosis had acute bacterial meningitis (all S. pneumoniae), which would be a prevalence of 0.9%. Two of the three patients presented seizure prior to introduction of pneumococcal conjugated vaccine49). Investigators in India conducted a retrospective study of children aged 6 to 18 months admitted to a tertiary-care hospital for a first febrile seizure. The prevalence of meningitis in a first FS was 0.86%, compared with 4.8% in a complex FS49). Based on published evidence and consensus45,48,49,50), the recommendation for LP has recently changed to "strongly consider" in infants under 12 months.
The updated AAP guidelines for neurodiagnostic evaluation in children with simple FS are supported by evidence from some reviews45). Guidelines for LP in children with simple FS is summarized as follows: (1) LP should be performed in any child who presents with a seizure and fever and has meningeal signs and symptoms (e.g., neck stiffness, positive Kernig and Brudzinski signs), or in any child whose history or examination suggests the presence of meningitis or intracranial infection; (2) LP is an option in any infant between 6 and 12 months of age who presents with a seizure and fever when the child has not received scheduled immunization, if the child is considered deficient in Hib or S. pneumoniae immunizations or when immunization status cannot be determined because of an increased risk of bacterial meningitis; (3) LP is an option in a child who presents with a seizure and fever and was pretreated with antibiotics, because antibiotic treatment can mask the signs and symptoms of meningitis.
As a practical consequence, LP should not be performed routinely. As stated in the guidelines, current data no longer support routine LP in well-appearing, fully immunized children who present with a simple FS.
On the basis of published evidence48,49,50,51), CSF is more likely to be abnormal in children initially seen with fevers and seizures who have had the following: (1) suspicious findings on physical and/or neurologic examinations, particularly meningeal signs; (2) complex FS; (3) previous physician visit within 48 hours before the seizure; (4) seizures on arrival to Emergency Departments; (5) prolonged postictal states; and (6) initial seizures after 3 years of age.
2. Electroencephalography
EEG is of limited value in the evaluation of children with FS12,45). EEG is more likely to be abnormal in older children with FS, children with a family history of FS, children with complex FS, or children with pre-existing neurodevelopmental abnormalities52,53). Although EEG abnormalities may be present in these children, their clinical significance is unclear.
There is no consistent evidence that routine EEG and/or abnormal EEGs after the first FS are predictive of either the risk of recurrence of FS or of the development of epilepsy45). Even studies that included children with complex FS and/or those with preexisting neurologic diseases (a group of children at higher risk of developing epilepsy) have failed to show EEG to be predictive of the development of epilepsy47). However, epileptiform discharges on the EEGs of patients with FS are important predictive risk factors for the development of epilepsy because the febrile illness lowers the seizure threshold, and patients with FS presenting with frontal paroxysmal EEG abnormalities may be at higher risk54).
Performing EEG within 24 hours of presentation can show generalized background slowing, which could make identifying possible epileptiform abnormalities difficult. Generalized slowing on EEG can be present up to 7 days after a child presents with febrile status epilepticus24,55). The reported incidence of EEG abnormalities in children with FS varies from 2% to 86%52,53,54). This wide range may be due to variable ages of the patients, variable criteria for selection of cases, differences in the definition of abnormalities, and variations in the time of EEG recording after seizures4,45). However, epileptic discharges do not correlate with recurrence and cannot predict the later development of epilepsy2).
The AAP stated that EEG should not be a part of the routine evaluation in neurologically healthy children with a simple FS. However, this statement did not include patients with complex FS1,45,47).
3. Neuroimaging
Based on available evidence and consensus, the AAP recommended that neuroimaging not be included in the routine evaluation of a child with a first simple FS in both 1966 and 201145,47). There is no evidence to support the use of skull films in the evaluation of a child with a first FS45).
No data have been published that either support or negate the need for computed tomography (CT) or magnetic resonance imaging (MRI) in the evaluation of children with simple FS. However, some data show that CT scanning is associated with radiation exposure that may escalate future cancer risk. MRI is a burden because it requires sedation and is costly56). Neuroimaging has provided evidence that hippocampal injury (hippocampal edema and subsequent mesial temporal sclerosis) can occasionally occur during prolonged and focal FSs in infants who otherwise appear normal. However, it is not clear whether focality and long duration are independent factors57).
A pre-existing lesion can increase the propensity for further focally prolonged seizures and thus cause further hippocampal damage24). A recent study found MRI abnormalities in 14.8% of children with complex FS, while only 11.4% of 159 children with simple FS had imaging abnormalities; however, this was not statistically significant58). The most common abnormalities in MRI were subcortical focal hyperintensity, abnormal white matter signal, and focal cortical dysplasia.
As with EEG, neuroimaging may be considered in children with neurologic abnormalities on examination and in those with recurrent FS.
4. Other investigations
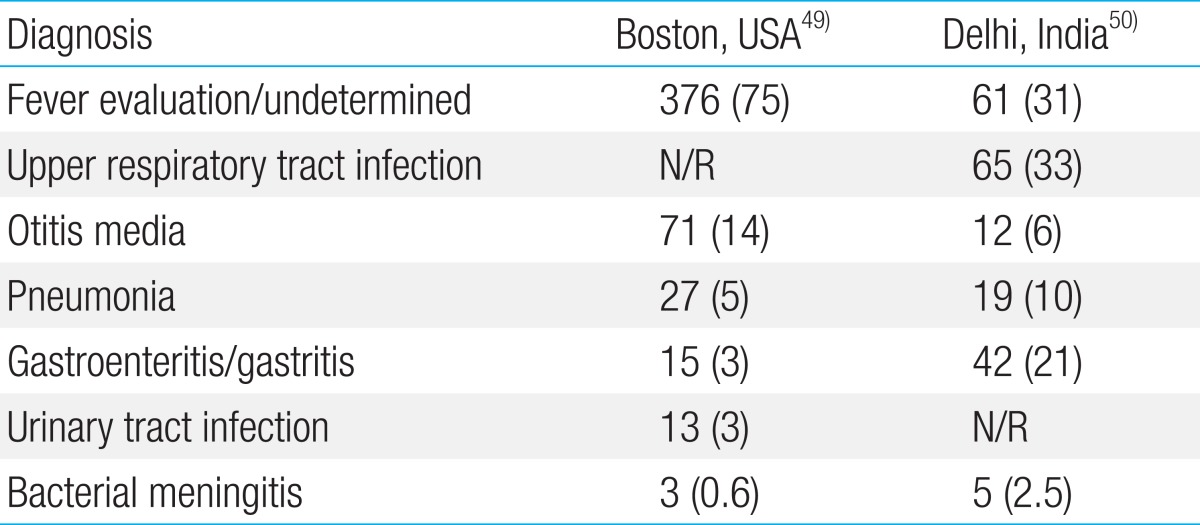
Based on available evidence and consensus, the AAP recommends that the following tests should not be performed routinely for the sole purpose of identifying the cause of a simple FS: measurement of serum electrolytes (calcium, phosphorus, or magnesium), blood glucose, or complete blood count. However, some children initially seen with FS are dehydrated and have low serum sodium concentration; therefore, they should be treated with overhydration with hypotonic fluid45,59). Complete blood counts may be useful in the evaluation of fever, particularly in young children, because the incidence of bacteremia in children younger than 2 years of age with or without FS is not different11). Therefore, laboratory testing in children with FS should be directed toward identifying the source of the fever rather than as a routine evaluation of the seizure itself. Causes of febrile illness in children with first FS, including the complex type, were the subject of two recent studies (one from a developed country and the other from a developing country) and are listed in Table 349,50). However, these studies did not consider the socioeconomic status, geographic differences, and immunization status of the population46). It can be seen that the majority of febrile illnesses are of undetermined etiology, a viral infection, or a bacterial respiratory tract infection. Occult bacteremia was a major reason for visits to the Emergency Department, but this has almost been eliminated by routine vaccination against S. pneumoniae and Hib in the US over the last two decades60). Moreover, even in an under immunized population, the likelihood of occult bacteremia is not higher in a child with a simple FS than in one with fever alone. Based on published evidence about the causes of fever in FS, it is evident that routine blood tests do not influence the management of the FS patient with no comorbidity45). There is also evidence that HHV 6 and 7 are one of the major causes of FS32).
In summary, most febrile illnesses in the FS-prone age group are of viral etiology, predominantly respiratory infections. Results of rapid viral diagnostic tests, notably those commonly available for HHV, respiratory syncytial virus and influenza, may aid in the management of a child during seasons when viruses are circulating in the population. However, there is no treatment for most viral infections46).
Treatments and prophylaxis
Parents may become extremely anxious when their child has FS, and concerned about the child's future because it can further interfere with the child's daily life15). It is important that physicians play a vital role in reassuring families about the prognosis, including risks of seizure recurrence, neurologic morbidity, and mortality after FS, alleviate their anxiety, and let them return to normal life15). Approaches to the treatment of FS are based on (1) the immediate treatment of prolonged or cluster seizures, (2) intermittent treatment at the time of illness, and (3) continuous anticonvulsant therapy for prophylaxis of FS12,24).
1. Immediate management
Treatment options for FS should include the use of a rescue seizure medication when the FS lasts longer than 5 minutes and when intravenous administration is not possible61). Acute medications such as rectal diazepam (0.5 mg/kg) or buccal (0.4-0.5 mg/kg) or intranasal (0.2 mg/kg) midazolam administration are effective in stopping an ongoing seizure when intravenous access is not available, and can also be provided for home use in patients with initial prolonged febrile seizure and a high risk of recurrence62,63). Randomized, controlled trials have shown that midazolam has an efficacy superior to that of diazepam62,63). The choice of acute treatment depends on the formulations available in different countries44). In the acute setting, intravenous diazepam and lorazepam are the drugs of choice for aborting seizures or terminating continuous febrile or afebrile seizures10). Diazepam is the fastest-acting benzodiazepine and rapidly crosses biological membranes, including the rectal mucosa and blood-brain barrier. A noteworthy disadvantage of diazepam is its short duration of action, the drug disappearing rapidly from the brain10). Lorazepam has a more prolonged anticonvulsant action64). Lorazepam is widely used in many countries including Korea, probably because of the longer duration of action and fewer adverse effects, but its acute anticonvulsant action is less rapid than that of diazepam65). Intravenous lorazepam (Ativan) in a dose of 0.1 mg per kg is the treatment of choice for acute tonic-clonic pediatric seizures10,42,64).
2. Intermittent therapy at time of fever
By the evidence of many reported studies, intermittent use of antipyretics such as ibuprofen or acetaminophen at the onset of fever is not recommended for ongoing FS or for prevention of recurrent FSs46). However, antipyretics are usually administered for the purpose of making a child feel more comfortable. There are small relative risks of hepatotoxicity, metabolic acidosis, renal failure, or respiratory failure with acetaminophen; and coma with ibuprofen, when given in over-dose or with other risk factors present66,67). Diazepam, given orally or rectally at the onset of fever, has been demonstrated statistically to be effective in reducing the recurrence of simple and complex FS; however, the seizure could begin before the detection of fever, resulting in "failure" of the preemptive therapy68,69,70). Although intermittent use of oral diazepam at the onset of fever is effective at reducing recurrence of simple FS, the AAP does not recommend it because of potential adverse effects and because many cases of recurrent FS occur before the recognition of fever68,70). Intermittent, rather than regular, prophylaxis with phenobarbital or valproate at onset of fever is not proven effective in reducing the incidence of subsequent FS71).
3. Continuous anticonvulsant therapy
In most studies, phenobarbital and valproate have been proven effective in preventing recurrent FSs70,71,72,73,74,75,76). However, the NIH Consensus Statement concluded that febrile seizures are benign events and, in general, treatment is not recommended45,47). For children at higher risk for epilepsy (i.e., those with abnormal neurologic development, complex FS, or a family history of afebrile seizures), treatment with phenobarbital or valproic acid "might be considered." It also might be considered for children whose first FS occurred earlier than 12 months of age and who had multiple FSs3). Controversy regarding the appropriate medical treatment of children with FS has been ongoing3). According to a study comparing phenobarbital and a placebo group in children with FS, there is a significant benefit of phenobarbital; five percent of the treated children had a recurrence compared with 25% in the placebo group72). However, it must be given daily, and blood levels must be within the therapeutic range (serum concentration of 15 µg/mL or higher) for the effective use of this drug12). Children receiving either intermittent or no phenobarbital showed no significant difference in recurrence; conversely, children receiving continuous phenobarbital on a daily basis showed a significant reduction in seizures. Therefore, if continuous phenobarbital shows no effect, it is likely to reflect noncompliance46,71). In contrast to most studies, one study did not observe a significant advantage of phenobarbital even in children treated within the therapeutic range. Recurrence occurred in 19% of the control children, in 11% of those prescribed phenobarbital, and in 8% of those who completed treatment. Whether the inclusion of a larger number of subjects might have identified a significant benefit of phenobarbital is unclear75).
Daily treatment with valproic acid is effective in reducing the risk of recurrent FS. In randomized, controlled studies, only 4% of children taking valproic acid, as opposed to 35% of control subjects, had a subsequent FS. Therefore, valproic acid seems to be as effective in preventing recurrent simple FS as phenobarbital, and significantly more effective than placebo75,76). Several studies have compared valproic acid with phenobarbital in the prevention of recurrent FSs. In evaluation of three groups of children treated with phenobarbital, valproic acid, and no therapy77), valproic acid provided significantly better outcomes than the control group with no therapy. Of interest, significant differences were not identified in seizure prevention between those patients treated with phenobarbital and the control children77). However, a randomized study with children who had previous FS and were treated with valproic acid, phenobarbital, or placebo found a statistically significant difference among the three groups, with recurrence rates of 4%, 19%, and 35%, respectively. Therefore, significant differences were evident between the treatment groups (valproic acid versus phenobarbital) and between children treated with valproic acid and those treated with placebo72,78).
Carbamazepine and phenytoin are not effective in preventing recurrent FSs. Therefore, these medications should be avoided when considering treatment for simple or complex FS3).
Although newer antiepileptic agents may prove to be safer and more effective in the treatment of recurrent or prolonged FSs, these medications have not yet been adequately studied in children with recurrent or complex FS23).
Levetiracetam can be an effective medication in preventing the recurrence of complex FS79).
A major disadvantage of continuous administration is the wide spectrum of adverse effects. Side effects and toxic reactions of phenobarbital are reported in up to 40% of infants or children receiving phenobarbital and are the cause of discontinuation of therapy in up to 25% of patients. Valproic acid causes few serious side effects or toxic reactions; however, gastrointestinal upset, toxic hepatitis, pancreatitis, and other side effects have been reported. Liver function should be monitored periodically in patients undergoing prolonged valproic acid therapy, particularly in infants 2 years and younger80). In another study, adverse effects were reported less often with valproate (24%) than with phenobarbital (61%), but the authors concluded that the risk-benefit ratio was insufficient to recommend either of these antiepileptic drugs for secondary prophylaxis80).
In summary, oral/intravenous diazepam and lorazepam are the drugs of choice for aborting a prolonged seizure in an acute setting. Although antipyretics may improve the children's comfort from fever, they should not be used to prevent FS prophylactically.
Although there is evidence that both continuous antiepileptic therapy with phenobarbital or valproic acid, and intermittent therapy with oral/rectal diazepam are effective in reducing the risk of recurrence, the AAP does not recommend that intermittent or continuous anticonvulsants be used to prevent recurrence of FS.
In situations in which parental anxiety associated with FS is severe, intermittent oral/rectal diazepam therapy at the onset of febrile illness to prevent recurrence may be advisable1,3,12,46,80).
Prognosis and outcome
Four potential adverse outcomes of FS that theoretically may be altered by an effective therapeutic agent are: (1) decline in IQ (intelligence quotient); (2) increased risk of epilepsy; (3) risk of recurrent FSs; and (4) death1). The first concern, a decline in IQ, low academic performance, neurocognitive inattention, or behavioral abnormalities, has not been shown to be a consequence of recurrent simple FSs81). Children who experienced FS observed no significant difference in their learning compared with sibling controls. In a study of children with FSs compared with control children, no difference in learning was identified, except in those children who had neurologic abnormalities before their first seizure81). The second concern, increased risk of epilepsy, is more complex. Children with simple FS have approximately the same risk (i.e., 1%) of developing epilepsy by the age of 7 as the general population39). However, children with a history of multiple simple FS, younger than 12 months at the time of their first FS, and a family history of epilepsy, are at higher risk, with generalized afebrile seizures developing by 25 years of age in 2.4%43). There currently is no evidence that simple FS causes structural damage to the brain1). The third concern, in contrast to the rare risk of developing epilepsy, is that children with simple FS have a high rate of recurrence. The risk varies with age. Children younger than 12 months at the time of their first simple FS have an approximately 50% probability of having recurrent FSs. Children older than 12 months at the time of their first event have an approximately 30% probability of a second FS18). Finally, there is a theoretical risk of a child dying during a simple FS, but no case of this has yet been reported1).
Vaccination and FS
Although vaccine-induced FS is a rare event that does not lead to deleterious outcomes, it could affect patient/parent and physician attitudes toward the safety of vaccination. Vaccine administration is the second most common medical event associated with FS82,83). Immunization has been associated with FS and an event occurring within 72 hours of immunization is commonly accepted as being associated with vaccine. Exceptions to this are the live attenuated vaccines for which events may be delayed until 7-14 days after vaccination84). Vaccines cause fever and may induce FS, but it is not clear whether a vaccine-induced fever is more epileptogenic than a fever due to other causes, such as a viral infection. Estimates of relative risk of seizure are dependent on vaccine type and components84). Seizure is more likely to occur after administration of certain vaccines, particularly live attenuated vaccines such as the measles, mumps, and rubella (MMR) vaccine, and toxin-containing or whole-cell preparations such as diphtheria-tetanus-acelluar pertussis (DTaP)85,86,87). Rates of all adverse events with acellular pertussis vaccines are estimated to be about one third of those of whole-cell preparations88).
As reported for simultaneous administration of influenza and pneumococcal vaccines, the associated risks could be increased when vaccines are administered in combination83). The recent multicomponent, recombinant meningococcal serogroup B (4CMenB) vaccine is an example. A recent study showed that increased reactogenicity is associated with this vaccine when it is administered concomitantly with routine vaccines (diphtheriatetanus-acellular pertussis, inactivated poliovirus, and hepatitis B plus Haemophilus influenzae type b [DTaP-IPV-HBV/Hib] and seven-valent pneumococcal vaccine [PCV7])89). Seventy-seven percent of infants had a fever of 38.5℃ or higher after 4CMenB injection, compared with only 45% after routine vaccinations alone and 47% with Neisseria meningitidis group C covaccination83,89). It needs to be kept in mind that children who experience FSs after immunization do not appear to be at higher risk for subsequent seizures or neurodevelopmental disability43,90). Although they are frequently reported, the adverse events following immunization such as FS are, nevertheless, rare events that may be attributable to DTaP and MMR vaccinations90). In comparison, measles disease itself results in 1 in 1000 infected children developing encephalitis, and 1 in 50 and 1 in 250 children with pertussis disease experience convulsions and encephalopathy, respectively82).
Therefore, despite the occurrence of this transient complication from fever after immunization, it needs to be emphasized that vaccination against measles and pertussis91,92), as well as invasive pneumococcus93) and Hib type b disease94,95), has significantly reduced not only the overall incidence of neurologic disorders associated with the diseases themselves, but also that of the associated serious and often permanent neurodevelopmental disabilities due to these diseases81).
Administration of acetaminophen at the time of primary immunization with an inactivated-component vaccine (e.g., DTwP-polio) has been shown to significantly reduce or prevent the appearance of fever and has found wide acceptance96,97).
Ibuprofen and acetaminophen have been equally recommended for administration at the time of DTaP immunization, both prior to vaccination, and every 4 hours for 24 hours thereafter for children with a history of FS, to reduce the possibility of postvaccination fever82).
In summary2): (1) None of the standard vaccinations is currently contraindicated for children with FS. All vaccinations of children with FS, and especially of children with a history of prolonged FS lasting >15 minutes, should be given individually under the supervision of the pediatrician or child neurologist who is responsible for providing information regarding the usefulness and potential side effects of any vaccination. (2) Children can be given a vaccination at least 2-3 months after the last episode of FS. This period may be shortened in light of the child's condition and the type of vaccine to be administered. (3) When a temperature of 37.5℃ or higher develops during the risk period for fever after vaccination, a suppository or oral diazepam can be administered prophylactically.
Guidelines for referral
It is important to refer every child to a hospital after an episode at home, as there will be significant parental anxiety afterwards. A thorough and detailed history of the episode should be written down. Guidance for referral to a secondary or tertiary hospital is summarized in Table 413,17).
Information for parents/guardians
To reduce anxiety and fear and to enable parents/guardians to cope with a child with FS, the following explanations must be given: (1) the natural history of FS, including incidence, age dependency, natural course, recurrence rate, incidence in siblings, difference between epilepsy and FS, the probability of onset of later epilepsy, and the prognosis for mental/behavioral development; (2) the possible measures to cope with fever and seizure episodes; and (3) a full explanation of the appropriate choice of antiepileptic drugs and potential side effects, and the need to avoid over-reliance on drug therapy2,44,98).
Acknowledgments
The author gratefully acknowledges data collection and language assistance rendered by Dr. Lee Eun Hye and medical student Lee You Jin.
Notes
No potential conflict of interest relevant to this article was reported.