A novel association between cerebral sinovenous thrombosis and nonketotic hyperglycinemia in a neonate
Article information
Abstract
Lethargy in newborns usually indicates central nervous system dysfunction, and many conditions such as cerebrovascular events, infections, and metabolic diseases should be considered in the differential diagnosis. Nonketotic hyperglycinemia is an autosomal recessive error of glycine metabolism, characterized by myoclonic jerks, hypotonia, hiccups, apnea, and progressive lethargy that may progress to encephalopathy or even death. Cerebral sinovenous thrombosis is a rare condition with various clinical presentations such as seizures, cerebral edema, lethargy, and encephalopathy. Here, we report the case of a newborn infant who presented with progressive lethargy. An initial diagnosis of cerebral venous sinus thrombosis was followed by confirmation of the presence of nonketotic hyperglycinemia.
Introduction
Sepsis, metabolic disorders, and cerebrovascular incidents are all neonatal emergencies that should be considered in the differential diagnosis of a lethargic newborn. Nonketotic hyperglycinemia (NKH) is an inherent error of glycine metabolism characterized by a deficiency in the glycine cleavage system. Generally, patients with NKH present with myoclonic jerks, hypotonia, hiccups, apnea, and progressive lethargy within the first weeks of life; this is usually followed by progression to coma or even death1).
The estimated incidence of cerebral sinovenous thrombosis (CSVT) during the neonatal period is between 1 and 12 per 100,000 live births23), with patients usually presenting with lethargy and seizures4). The most frequently involved sinuses in neonates are the superior sagittal and lateral sinuses. Here, we present the first case report of NKH with CSVT.
Case report
A male infant was admitted to the Emergency Department on the third day of life due to a weak cry and inability to feed. Physical examination revealed a lethargic and hypotonic infant. He was normothermic and pink with a heart rate of 145/min, a respiratory rate of 43/min, and normal peripheral perfusion. Despite prompt hospitalization, progressive worsening of the infant's consciousness with a diminished response to painful stimuli was observed during the following hours. Five hours after presentation, the patient's condition deteriorated acutely, and he was intubated and placed on mechanical ventilation because of impending respiratory failure. A tentative diagnosis of neonatal encephalopathy was made.
Serum levels of blood urea nitrogen, creatinine, aspartate transaminase, alanine transaminase, glucose, and electrolytes were within normal limits. No cells were observed on microscopic examination of a cerebrospinal fluid (CSF) sample obtained by lumbar puncture, and CSF levels of protein and glucose were normal. Plasma ammonium and lactate levels were normal. Cranial ultrasonography revealed the presence of severe cerebral edema. A subsequent cerebral magnetic resonance venography confirmed the diagnosis of CSVT involving the right transverse and sigmoid sinuses (Fig. 1A). Treatment with low-molecular-weight heparin (LMWH; enoxaparin 1.5 mg/kg subcutaneously twice daily) was initiated. Screening for procoagulant mutations identified the presence of a mutation in the plasminogen activator inhibitor-1 (PAI-1) gene, with no abnormalities in the factor V Leiden, prothrombin 20210 A, and methylenetetrahydrofolate reductase (MTHFR) genes. The infant's clinical condition improved during the subsequent days, and a repeat magnetic resonance venography showed that the thrombosis had resolved (Fig. 1B).
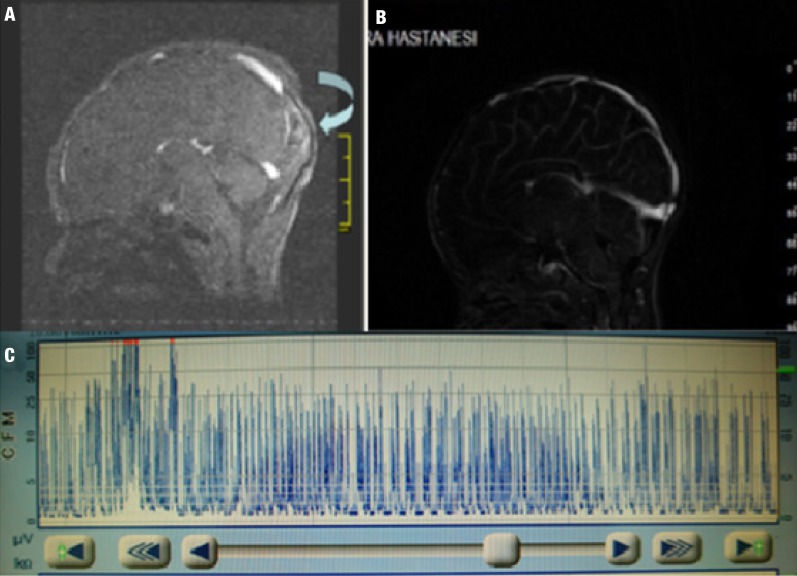
(A) An isointense filling defect is noted on two-dimensional postcontrast venography, which is consistent with a thrombus (curved arrow). (B) Normal magnetic resonance venography imaging. (C) Abnormal electro-encephalogram activity (burst suppression trace).
Two days later, the infant's condition again deteriorated, and a weak spontaneous respiratory effort prompted reintubation and mechanical ventilation. Prior to intubation, it was observed that the patient had hiccups, and a wave pattern on an electro-encephalogram was deemed abnormal (Fig. 1C). Evaluation of serum and CSF levels of amino acids revealed the presence of elevated glycine levels (serum, 1,449 µmol/L; CSF, 56 µmol/L). A CSF/serum glycine ratio of 0.176 (a value greater than 0.08 is diagnostic) led to a diagnosis of NKH. After stabilization of the patient's clinical symptoms, we performed a second lumbar puncture on the 25th day of the infant's life. The second measurement of the CSF/serum glycine ratio was 0.15. Dextromethorphan (3.5 mg/kg/day), phenobarbital (5 mg/kg/day), and vitamin B6 (pyridoxine, 100 mg/kg body weight) were added to enoxaparin treatment. Although the encephalopathic clinical symptoms were eventually resolved, the patient could not be weaned off the ventilator, and a tracheostomy was performed.
Discussion
Lethargy in newborns is usually indicative of central nervous system dysfunction, and many conditions such as CSVT, infections, and metabolic diseases should be considered in the differential diagnosis.
CSVT rarely affects children, with a reported childhood incidence of 0.67/100,000 in a large multicenter Canadian study4). In a study by the International Pediatric Stroke Study Group, more than half of the episodes of childhood CSVT occurred in newborns aged less than 28 days45), and it was concluded that neonates were at a higher risk for developing CSVT than older children6). In recent years, neonatal CSVT has become a more recognized clinical condition, mainly due to better awareness and improved neuroimaging techniques. It is widely believed that the actual incidence of neonatal CSVT is grossly underestimated, and a diagnosis is usually missed because of nonspecific clinical presentation, the need for a high index of clinical suspicion, and limited access to appropriate imaging techniques7).
The presentation of CSVT may vary in newborns. In one of the largest case series by Nwosu et al.8), respiratory problems were present in 73%; seizures, in 59%; tachycardia, in 12%; apnea, in 47%; hypotonia, in 58%; and lethargy, in 42% of affected infants. Most of the patients in the present study were brought to medical attention within 48 hours from the onset of symptoms. In our patient, breathing difficulties, hypotonia, and lethargy were the prominent findings. Maternal risk factors that have been described for CSVT include preeclampsia and gestational diabetes mellitus. Neonatal comorbidities such as anemia, cyanosis, cardiac malformations, bacterial sepsis, and dehydration have also been implicated as risk factors for CSVT. Conversely, the presence of an underlying procoagulant genetic condition (MTHFR, prothrombin 20210A, or PAI-1 gene mutations) may also predispose for CSVT9).
Our patient was homozygous for the 4G allele of the PAI-1 4G/5G promoter polymorphism. Although the role of this mutation in the pathogenesis of neonatal thromboembolism is as yet not fully explained, it has been implicated as a cause of central vein thrombosis in a premature infant10).
Neonatal CSVT commonly involves the sagittal and transverse sinuses, and multiple sinuses may be affected in up to 50%-70% of cases. The ensuing venous congestion is followed by hemorrhagic infarction. Despite having a thrombus in the sagittal sinus, our patient did not have any signs of a hemorrhagic infarct (Fig. 1). CSVT during the neonatal period is generally associated with a poor neurodevelopmental outcome, with reported neurological deficits (epilepsy, cognitive impairment, and motor retardation) in 10%-80% of affected newborns23). The treatment of CSVT in newborns remains controversial. Several studies have shown a benefit of treatment with LMWH3).
NKH is classified as neonatal, infantile, late onset, and transient based on its clinical course in the literature. Neonatal NKH generally presents within the first few days of life as poor feeding, profound lethargy and hypotonia, encephalopathy, and severe seizures. A diagnosis of NKH requires amino acid analysis of plasma and CSF samples, the hallmark of which is the presence of a CSF glycine-to-plasma glycine ratio greater than 0.08. A diagnosis of NKH also requires the exclusion of other organic acidemias as well as the absence of ketoacidosis. Our patient did not have acidosis, and evaluations for urinary organic acids were normal. The presence of elevated levels of glycine in the blood and urine plus a CSF glycine-to-plasma glycine ratio of 0.176 confirmed the diagnosis in our patient. A definitive diagnosis of NKH is based on mutation and enzymatic analysis in these cases. Many studies have shown that approximately 80% of patients with NKH are deficient in the activity of glycine decarboxylase, which is one of four enzymes of the glycine cleavage system11). Unfortunately, we could not confirm the diagnosis of NKH in the present case using enzyme measurements and associated mutation analysis because of economic and technical problems.
No definitive treatment has been described for NKH, and the main aim of the current management strategies is to decrease glycine levels and block N-methyl-D-aspartate receptors. Sodium benzoate and dextromethorphan have been used for this effect. We speculated that the presence of a procoagulant PAI-1 mutation might have facilitated the development of CSVT during an attack of NKH in this infant.
Many structural brain abnormalities are associated with NKH, including parenchymal volume loss abnormalities of white matter and agenesis or hypoplasia of the corpus callosum12). These findings are not specific for NKH. Our case did not show additional structural abnormalities. NKH often can induce abnormal functional magnetic resonance imaging findings. Cerebral glycine concentrations, detected using long echo time proton magnetic resonance spectroscopy, were reported to correlate reliably with clinical findings. This imaging study may be a useful tool for monitoring responses to treatment with sodium benzoate or dextromethorphan in cases with NKH13).
Neonatal lethargy and encephalopathy are dramatic conditions, and the differential diagnosis requires the consideration of several diseases, including cerebrovascular events, metabolic disorders, sepsis, maternal drug use, and intoxication. A detailed family history may provide clues regarding the underlying condition. In our case, both CSVT and NKH seemed to be separate etiological entities that might have contributed to the encephalopathic findings. The identification of one cause of NKH may mask the presence of another, as in our patient. NKH was only considered in the differential diagnosis after the patient developed hiccups and seizures despite resolution of the sinus thrombosis. To our knowledge, this is the first case report of NKH with CSVT.
Notes
Conflicts of interest: No potential conflict of interest relevant to this article was reported.