Clinical features of macrocephaly at birth in Korea
Article information
Abstract
Purpose
This study aimed to investigate the clinical features of macrocephaly at birth in Korea using ultrasonography.
Methods
We retrospectively investigated the medical records of full-term birth neonates in Cheil General Hospital & Women's Healthcare Center from January 2000 to June 2012. The following parameters were recorded and analyzed: gestational age, sex, birth weight, height, occipitofrontal circumference (OFC), physical examination, perinatal problems, and ultrasonography results. Macrocephaly was diagnosed when the OFC was greater than two standard deviations, based on the 2007 Korean National Growth Charts.
Results
There were 75 neonates with macrocephaly at birth (52 boys and 23 girls), with a mean OFC of 38.1±0.49 cm. A comparison of the birth weight and height with the OFC value showed that height was correlated with OFC (r=0.35) but birth weight was not correlated with OFC (r=0.06). There were no remarkable findings in 56 cases (75%). Germinal matrix hemorrhage was identified in 10 cases (13%). An enlarged cerebrospinal fluid space was found in 5 cases (6.7%). There were 3 cases of mega-cisterna magna (4%), 1 case of ventriculomegaly, and 1 case of an enlarged interhemispheric space (6 mm) among these patients. In addition, a choroid plexus cyst was seen in 1 case. Mineralizing vasculopathy in both basal ganglia with no evidence of congenital infection was found in 2 cases and an asymptomatic subarachnoid hemorrhage was found in 1 case.
Conclusion
Our results indicate that macrocephaly at birth has benign ultrasonography findings and shows a pattern of male dominance.
Introduction
Head circumference measurement is an important physical examination to evaluate childhood growth and development because deviations from normal head growth patterns can indicate an underlying congenital, genetic, or acquired neurologic problem1).
Macrocephaly is defined as an occipitofrontal circumference (OFC) of two standard deviations (SDs) or more above the mean for age, gender, and gestation (≥97th percentile)2,3). The size of the head may be large at birth or may occur with accelerated postnatal growth, and is caused by an increase in the size of any component of the cranium including the brain, cerebrospinal fluid (CSF), blood, or bone, or can be attributed to increased intracranial pressure. Statistical data about incidence rate is low, and little is known about the condition due to the rarity and heterogeneity of cases4,5). Macrocephaly is usually familial and benign, and it can be a single manifestation or appear as part of a syndrome. According to the Online Mendelian Inheritance in Man Database, 225 syndromes are related to macrocephaly.
Several reports of clinical approaches and recommendations for children with macrocephaly have been published6,7,8,9,10,11). However, there are a limited number of studies regarding macrocephaly at birth in Korea. Additionally, the radiologic features of this condition are not well-defined. This study used ultrasonography to investigate the characteristics and significance of an enlarged head at birth in Korean infants.
Materials and methods
We retrospectively investigated the medical records of neonates who were born at Cheil General Hospital & Women's Healthcare Center from January 2000 to June 2012. The following parameters were recorded and analyzed: gestational age, sex, birth weight, height, OFC, physical examination, perinatal problems, and the ultrasonography results.
All subjects were Korean and were full-term at birth. Premature neonates were excluded because: (1) there is currently inadequate information about standard head growth parameters of premature infants in Korea; (2) it is inappropriate to use intrauterine head growth charts to follow a neonate postnatally; and (3) the head growth pattern of premature infants is different from full-term infants.
All neonates received physical exmination at birth and on the day of discharge by pediatricians. Head circumference was measured with a nondistensible plastic tape and included the glabella (anteriorly) and the most prominent portion of the occiput (posteriorly)1). Cases with scalp edema, overriding sutures, or severe cephalhematoma were excluded to avoid measurement errors. Macrocephaly was diagnosed when the head circumference was greater than 2 SD for gestational age and gender, based on the 2007 Korean National Growth Charts12).
Neonates diagnosed with macrocephaly underwent ultrasonography within the second week after birth. Images were obtained using real-time ultrasonography with 8 MHz convex and 12 MHz linear transducer (LOGIQ 9 scanner, GE medical system, Milwaukee, WI, USA). Descriptive statistics were performed and correlations were determined with Pearson correlation coefficient calculations.
This study was approved by the Institutional Review Board of Cheil General Hospital & the Women's Healthcare Center.
Results
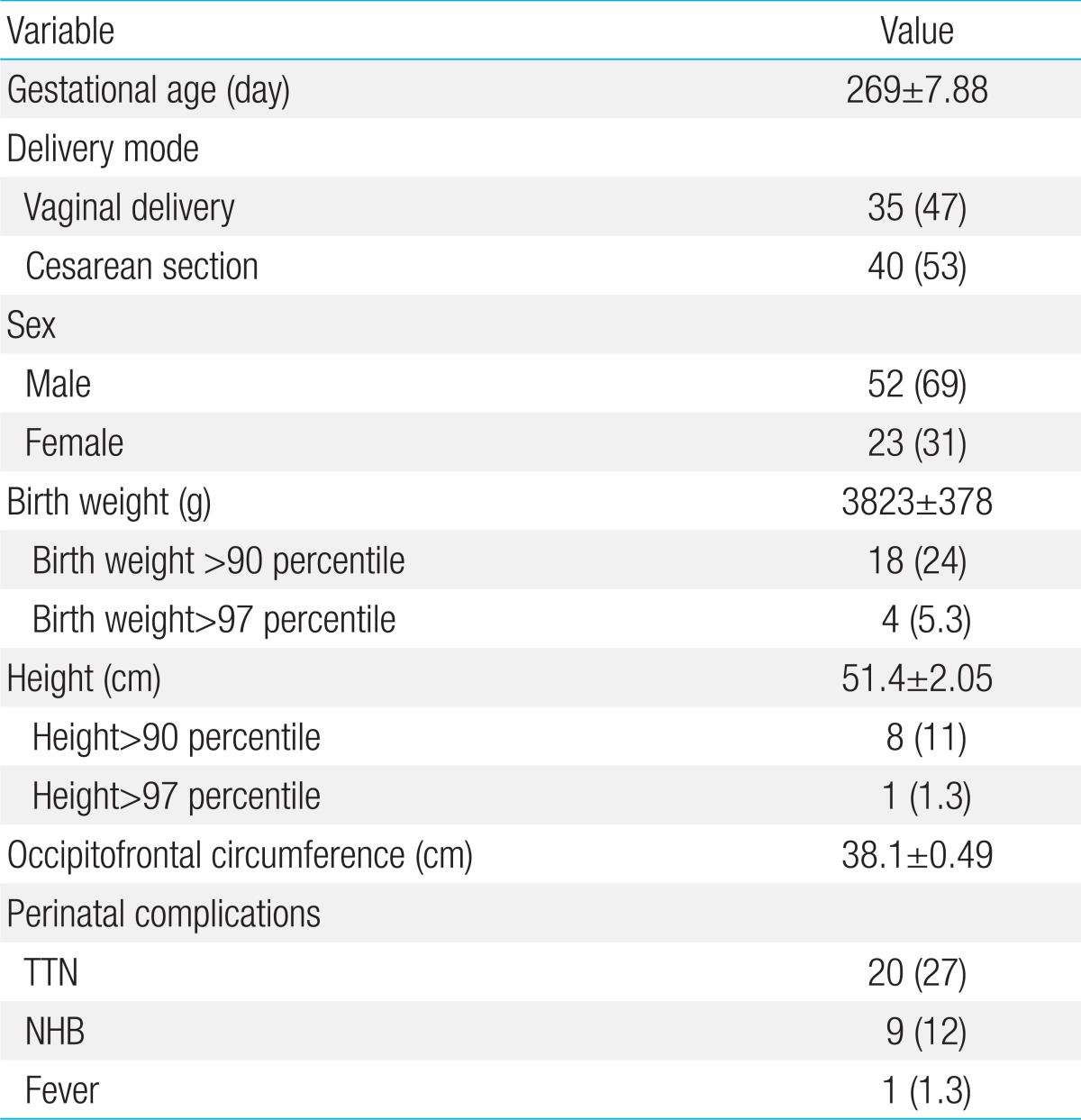
During the period of investigation, 15,598 full-term babies were born in the Cheil General Hospital and 75 of these births were included in this study (0.5%). The mean gestational age was 279±7.88 days. In regards to the mode of delivery, 35 cases had a spontaneous vaginal delivery, and 40 had a Cesarean section. There were 52 male and 23 female infants, and the ratio of males to females was approximately 2.3:1. The demographic and clinical characteristics of the subjects are shown in Table 1.
The mean birth weight was 3,923±378 g, and the mean height was 51.4±2.05 cm. Eighteen babies had a birth weight above the 90th percentile (24%) and 4 babies had a birth weight above the 97th percentile (5.3%). None of the infants were small for gestational age. Eight babies had a height above the 90th percentile (11%) and 1 baby had a height above the 97th percentile (1.3%). The mean OFC was 38.1±0.49 cm. In 14 cases (19%) the OFC exceeded the 97th percentile with more than 1 cm, and the largest OFC was 39.5 cm which was 1.7 cm greater than the 97th percentile. A comparison of the birth weight and height with the OFC showed that height was correlated with OFC (r=0.35) but birth weight was not correlated with OFC (r=0.06).
Transient tachypnea of the newborn was identified in 20 cases (27%) and neonatal hyperbilirubinemia was observed in 9 cases (12%). One patient had a fever for 1 day (1.3%), and all of the patients had a normal neonatal screening test.
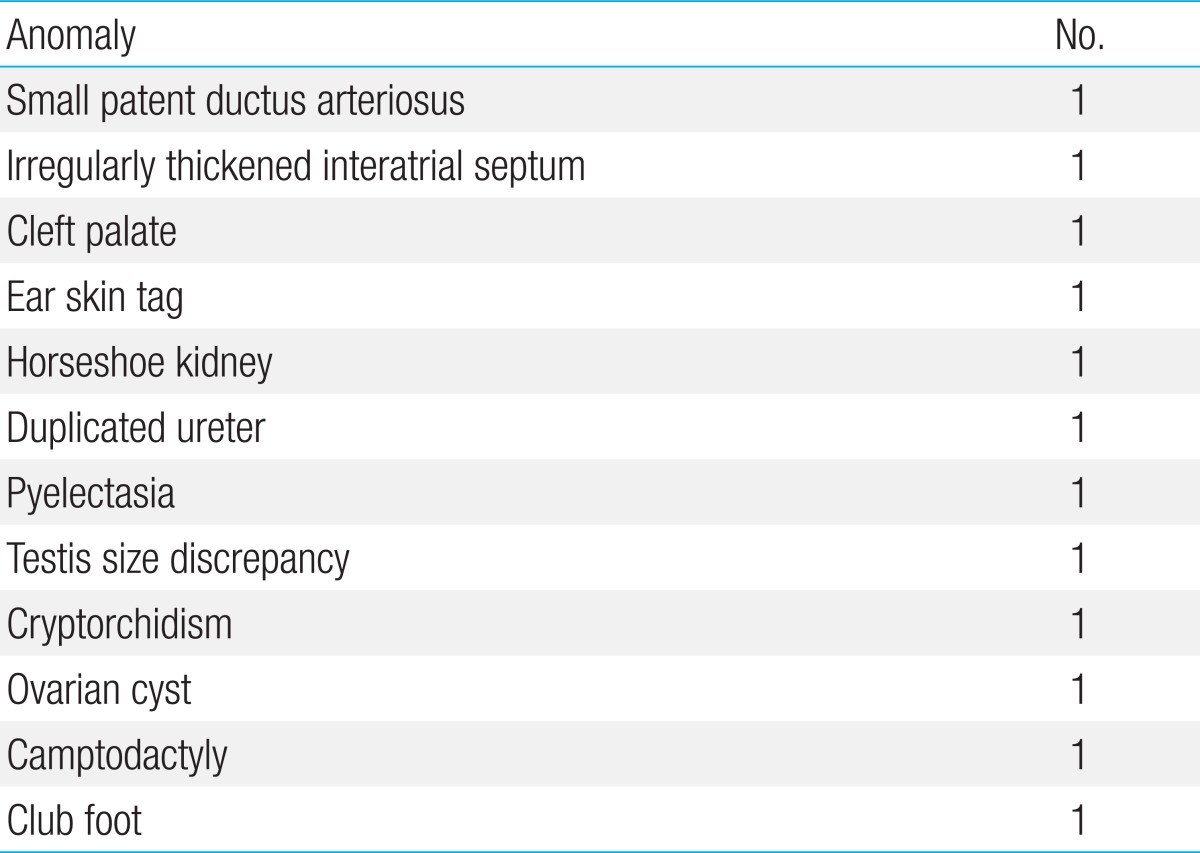
The following anomalies occurred on a single case basis in 12 patients (Table 2); small patent ductus arteriosus, irregular thickened interatrial septum, horseshoe kidney, duplicated ureter, testis size discrepancy, cryptorchidism, ovarian cyst, and club foot. None of the patients manifested abnormal findings such as hypotonia, hyperreflexia, bulging anterior fontanel, wide fontanel, or distended veins in the frontal region.
All of the patients underwent at least one ultrasonography and 1 patient also underwent magnetic resonance imaging (MRI). Table 3 shows the ultrasonography findings. There were no remarkable findings in 56 cases (75%), and these cases were diagnosed as megalencephaly. Germinal matrix hemorrhage without intraventricular hemorrhage (IVH) was found in 10 cases (13%), and 3 of these patients had subependymal cysts. An enlarged CSF space was found in 5 cases (6.7%), and 3 of these patients had a mega-cisterna magna (4%); 1 case had a ventriculomegaly, and 1 case had an enlarged interhemispheric space (6 mm). A choroid plexus cyst was identified in 1 case, and mineralizing vasculopathy in both basal ganglia with no evidence of congenital infection was found in 2 cases. Subarachnoid hemorrhage (SAH) was found in 1 case, and, initially, a small echogenic lesion was seen at the right inferoposterior temporal region along the suture line. A brain MRI was performed which detected focal SAH with adjacent parenchymal ischemia, thin subdural hemorrhage at the bilateral posterior parietal, occipital, and tentorial area without a skull fracture. This patient was asymptomatic and had no remarkable findings in the neurologic examination. A follow-up ultrasonography indicated that the SAH resolved spontaneously without sequelae.
Discussion
This study sought to identify clinical features of macrocephaly at birth. To our knowledge, this is the first study to investigate macrocephaly based on the 2007 Korean National Growth Charts. Several standardized charts are currently available to monitor OFC. These include the Centers for Disease Control and Prevention (CDC) National Center for Health Statistics head circumference charts for children 0 to 36 months of age (CDC growth charts); the World Health Organization (WHO) head circumference charts for children zero to five years of age (WHO growth standards); and the United States Head Circumference Growth Reference charts for children 0 to 21 years of age which combine growth reference data from the CDC, Nellhaus Head Growth Chart and the Fels Longitudinal Study, among others13,14,15,16). However, these growth charts, including the 2007 Korean National Growth chart, have not provided OFC references at birth based on gestational age. Similarly to normograms for preterm birth weights, an OFC reference by gestation is needed for newborn infants17,18). Additional neonates would have been included in this study if gestational age were considered.
This study showed that macrocephaly was twice as prevalent in boys as in girls. Previous studies that analyzed infants and children determined a similar male dominance pattern5,7,9,19,20,21,22). However, the inheritance pattern in this study was not analyzed due to a lack of family history information. The body measurement results indicated that birth height was correlated with OFC, which confirmed the results of another study of infants and children9).
All of the patients in this study were considered to have isolated macrocephaly. Twelve patients had several anomalies, but these were not associated with macrocephaly syndrome23,24). Although there are over 200 syndromes associated with macrocephaly, some of these syndromes are quite rare. In addition, in some cases of metabolic megalencephaly, the macrocephaly was not evident during the neonatal period8). However, a referral bias may have affected this result. Prenatal transfer to the tertiary hospital may occur after detection of severe fetal congenital anomalies.
The majority of patients had no remarkable abnormality on ultrasonography and many of the patients with anomalies were diagnosed with germinal matrix hemorrhage (GMH) and an enlarged subarachnoid space. GMH without IVH (grade I) usually disappears within 1 year. Additionally, benign enlargement of the subarachnoid space is relatively common and occurs in approximately 16 percent of infants25). Full-term infants with subarachnoid space enlargement typically have normal development, a normal neurologic examination, and usually do not require surgical intervention26,27). This study identified an enlarged subarachnoid space in 6.7% of the patients, which was lower than the results of a previous study28,29,30).
In conclusion, the results of this study indicate that benign radiologic findings are often in the macrocephaly at birth. However, a benign ultrasonography finding does not guarantee that there will be benign long-term neurological and neurophysiological outcomes. Serial OFC measurements and clinical follow-up are necessary to accurately determine the course of macrocephaly.
Notes
No potential conflict of interest relevant to this article was reported.