Importance of neonatal screening for primary immunodeficiencies
Article information
Key message
· Early detection of asymptomatic infants with primary immunodeficiencies before the onset of infections enables effective treatment and intervention to prevent serious sequelae.
· T-cell receptor excision circles and kappa-deleting recombination excision circles have recently been used to detect T- or B-cell lymphopenia in neonates.
Primary immunodeficiencies (PIDs) are a heterogeneous group of inborn errors of immunity including T- and B-cell deficiencies, complement deficiencies, phagocyte disorders, and combined immunodeficiencies. With the novel progress in immunological and molecular techniques, including the development of diagnostic genetic tests, 430 PID have been identified and characterized [1]. PID predispose individuals to recurrent or unusual infections and are associated with other immune disorders including autoimmune diseases and lymphomas [2]. It is difficult to identify infants with PID prior to the onset of clinical symptoms. Thus, some patients may remain untreated for several years, which leads to many complications. Neonatal screening for PID enables the early identification of asymptomatic infants with a PID. Early diagnosis and intervention prevent serious sequelae.
Assays for T-cell receptor excision circles (TRECs) and kappa-deleting recombination excision circles (KRECs) are being increasingly used to screen for PID in many countries worldwide. For example, TREC-based PID screening programs were established in the majority of American states, Israel, Taiwan, Qatar, and Canada. The Netherlands, Japan, France, Spain, Norway, France, the United Kingdom, Turkey, Saudi Arabia, Iran, Iceland, Denmark, and Brazil have applications in progress to commence screening programs [3]. In Iran, between 2014 and 2016, neonatal screening for PID was performed of 2,160 newborns and identified 51 cases of severe combined immunodeficiency (SCID), agammaglobulinemia, and ataxia telangiectasia [4].
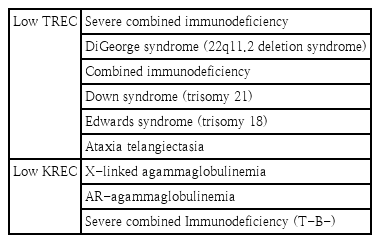
TREC are small circular pieces of episomal DNA that are formed during T-cell receptor rearrangement within the thymus. TREC is an accurate measure of thymic function because it arises in the late phase of thymocyte maturation. Approximately 70% of T cells generate TREC. Several properties were established for TREC that make them useful markers of thymic output: high stability, not prone to degradation, and not replicated when cells divide [5]. The development of a TREC assay utilizing dried blood spots enabled neonatal screening for SCID as a public health measure. SCID are caused by diverse genetic mutations that lead to the absence of all T-cell functions and in a lack of B cells and natural killer cells. SCID is the most severe PID. Affected patients present with chronic diarrhea, pneumonia, sepsis, and failure to thrive. Recurrent infections with opportunistic organisms including Pneumocystis jiroveci, Candida albicans, adenovirus, cytomegalovirus, and varicella zoster virus. The early detection of SCID should be considered a pediatric emergency because making a diagnosis prior to the onset of severe infections permits lifesaving hematopoietic stem cell transplantation (HSCT). Recent evidence suggests that patient outcomes are markedly improved if HSCT is performed before the age of 3.5 months [6].
The TREC assay can also be used to identify syndromes with T-cell impairments, such as Down syndrome (trisomy 21), Edwards syndrome (trisomy 18), and ataxia telangiectasia [7]. Like T cells, in the process of B-cell maturation, KREC is produced by kappa-deletion recombination events that determine allelic and isotypic exclusion of the gamma chain [8]. B-cell maturation defects in agammaglobulinemia occur before kappa-deleting recombination, KREC should not be produced. Therefore, KREC measurements are used to identify cases of asymptomatic agammaglobulinemia, which comprise around 30% of all antibody deficiencies. Agammaglobulinemia patients present with B-cell differentiation arrest in the bone marrow and the consequent absence of mature B cells and profound deficiency of all immunoglobulins (Ig). In these patients, recurrent infections appear after 6 months of age, whereas the mean age at diagnosis is 3 years [9]. This diagnostic delay results in frequent infections such as pneumonia, sepsis, meningitis, and other bacterial infections. Live viral vaccines such as those for measles, mumps, and rubella and chicken pox may be lethal to these children. Thus, early diagnosis and treatment including periodic Ig replacement therapy are essential to improving patient prognosis [10]. TREC/KREC-based screening enables the early identification of asymptomatic infants with cellular and antibody deficiencies (Table 1) and facilitating diagnostic precision, appropriate treatment, and improved clinical outcomes.
Notes
Conflicts of interest
No potential conflicts of interest relevant to this article are reported.