Prevalence, comorbidities, diagnosis, and treatment of nonallergic rhinitis: real-world comparison with allergic rhinitis
Article information
Abstract
Rhinitis is among the most common respiratory diseases in children. Nonallergic rhinitis, which involves nasal symptoms without evidence of systemic allergic inflammation or infection, is a heterogeneous entity with diverse manifestations and intensities. Nonallergic rhinitis accounts for 16%–89% of the chronic rhinitis cases, affecting 1%–50% (median 10%) of the total pediatric population. The clinical course of nonallergic rhinitis is generally rather mild and less likely to be associated with allergic comorbidities than allergic rhinitis. Here, we aimed to estimate the rate of coexisting comorbidities of nonallergic rhinitis. Nonallergic rhinitis is more prevalent during the first 2 years of life; however, its underestimation for children with atopic tendencies is likely due to low positive rates of specific allergic tests during early childhood. Local allergic rhinitis is a recently noted phenotype with rates similar to those in adults (median, 44%; range, 4%–67%), among patients previously diagnosed with nonallergic rhinitis. Idiopathic rhinitis, a subtype of nonallergic rhinitis, has been poorly studied in children, and its rates are known to be lower than those in adults. The prevalence of nonallergic rhinitis with eosinophilia syndrome is even lower. A correlation between nonallergic rhinitis and pollution has been suggested owing to the recent increase in nonallergic rhinitis rates in highly developing regions such as some Asian countries, but many aspects remain unknown. Conventional treatments include antihistamines, intranasal corticosteroids, and recent treatments include combination of intranasal corticosteroids with azelastin or decongestants. Here we review the prevalence, diagnosis, comorbidities, and treatment recommendations for nonallergic rhinitis versus allergic rhinitis in children.
Key message
• Rhinitis is among the most common respiratory diseases in children.
• Nonallergic rhinitis is a heterogeneous entity involving nasal symptoms without evidence of systemic inflammation or infection.
• The prevalence and etiology of some phenotypes of nonallergic rhinitis in children are well defined, but information about other rhinitis phenotypes remains uncertain.
• Comprehensive understanding to childhood nonallergic rhinitis might enable to provide accurate diagnostic and therapeutic approaches to childhood nonallergic rhinitis.
Graphical abstract.
Introduction
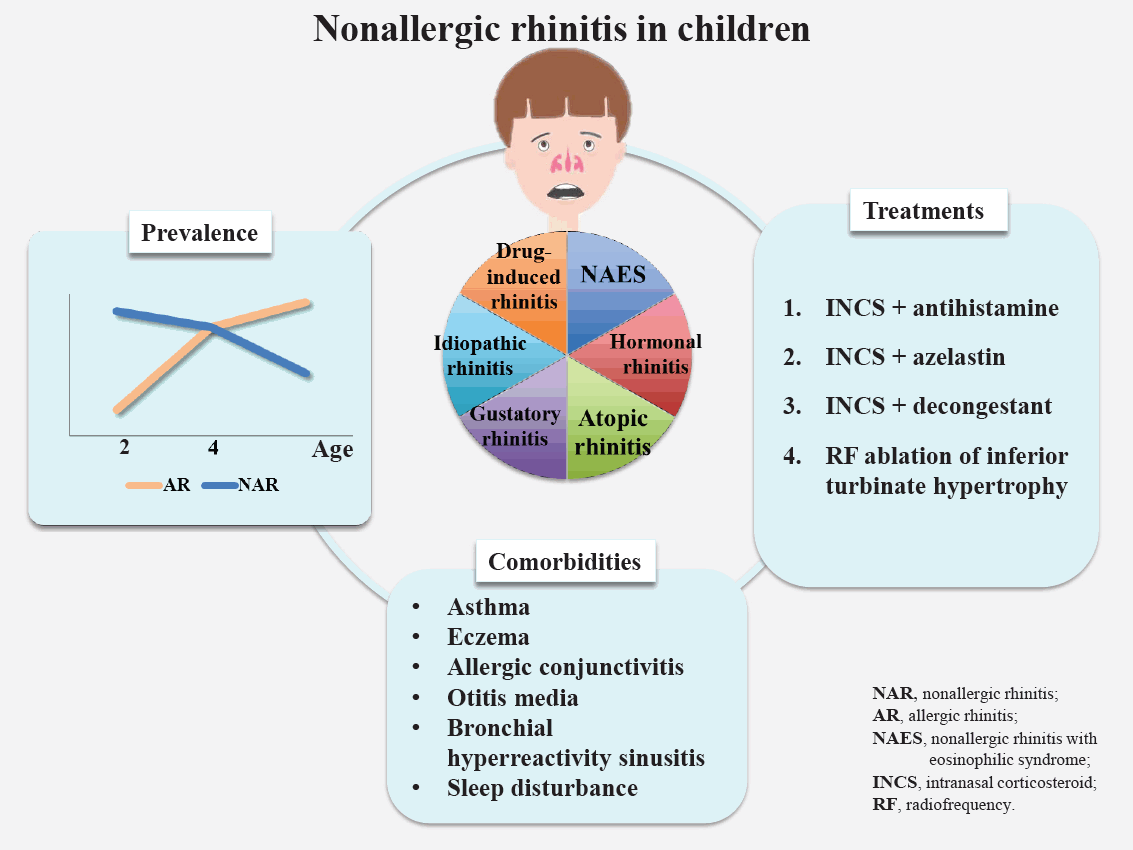
Chronic rhinitis, among the most common respiratory diseases in childhood, is characterized by nasal symptoms including rhinorrhea, nasal obstruction, sneezing, and itching [1,2]. A significant proportion of patients in clinical practice with typical rhinitis signs and/or symptoms but no evidence of allergen sensitization are classified as having nonallergic rhinitis [3,4]. Nonallergic rhinitis is a heterogeneous entity whose symptoms may be persistent or intermittent and phenotypes include nonallergic rhinitis with eosinophilia (NARES), hormonal rhinitis, atopic rhinitis, drug-induced rhinitis, gustatory rhinitis, and idiopathic rhinitis [5].
Recognition of the differentiation between nonallergic rhinitis, allergic rhinitis, and other chronic rhinitis is essential in the clinical setting considering the fact that it has various phenotypes [6,7]. There is a paucity of clinical studies on managing children with nonallergic rhinitis, as data on its diagnosis, classification, etiology, treatment, and natural history are insufficient, especially in childhood. Here we review the prevalence, clinical presentation, comorbidities, and management of nonallergic rhinitis in children.
Epidemiology of nonallergic rhinitis in children
Nonallergic rhinitis is a diagnosis of exclusion that is characterized by signs and/or symptoms similar to allergic rhinitis (e.g., rhinorrhea, nasal obstruction, nasal itching, and sneezing after allergen exposure). After the exclusion of allergic rhinitis based on the identification of allergen sensitizations, nonallergic rhinitis is suspected and efforts are made to identify associated conditions like sinusitis or adenoid hypertrophy [7,8]. Studies have reported that the prevalence of nonallergic rhinitis is 0.7%–50.0 % (median, 10.1%) in pediatric population. Nonallergic rhinitis seems to account for 15.7%–89.0% of the chronic rhinitis cases (Table 1).
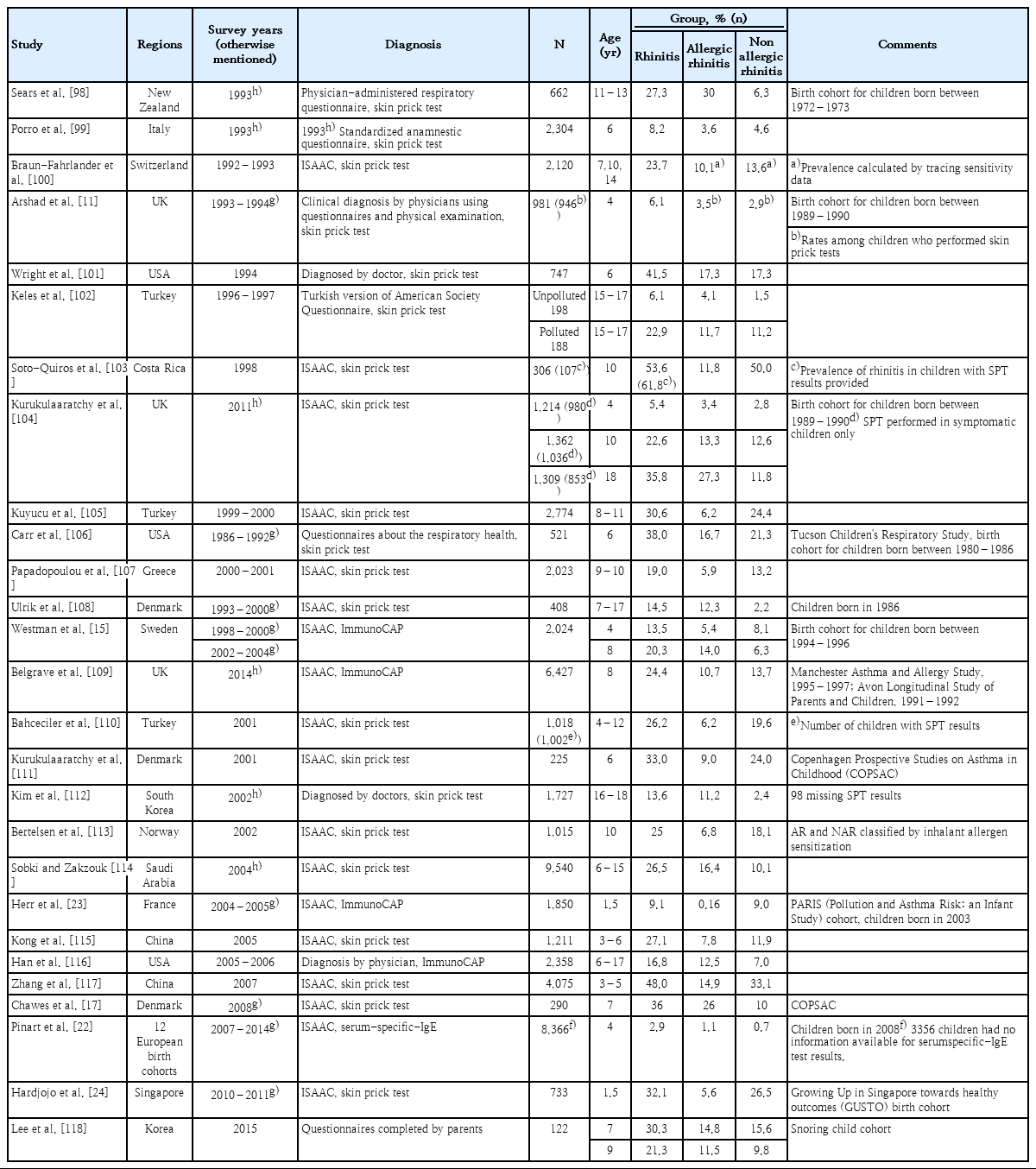
Prevalence of rhinitis among general pediatric population with and without allergic sensitization proven by skin prick tests or serum-specific immunoglobulin E levels
In epidemiologic studies, decreasing trends in the incidence of nonallergic rhinitis were observed, while the incidence of allergic rhinitis increased with age [9]. In a birth cohort from the United Kingdom, a decreasing trend in the proportion of children with symptomatic rhinitis was estimated at 79%, 45%, and 42.3%, respectively, at 2, 4, and 10 years of age [10-13]. Similar results were observed in a prospective birth cohort study (Multicenter Allergy Study) that assessed 1,314 children [14]. In another birth cohort study from Sweden, the prevalence increased from 5.4% to 14% and that of nonallergic rhinitis decreased from 8.1% to 6.3% at 4 and 8 years of age, respectively [15]. Other birth cohorts reported that the rate of rhinitis in children was similar to that of nonallergic rhinitis from 5 to 7 years of age [16,17]. A recent study of a Korean population reported that the prevalence of allergic rhinitis increased from 22.1% to 28.3% from 7 to 12 years of age, while that of nonallergic rhinitis decreased from 21.3% to 13.3% [18].
Considering the data of the studies mentioned above, nonallergic rhinitis is found to be prevalent in children younger than 2 years with chronic rhinitis, and the proportion tends to decrease with age, whereas the proportion of allergic rhinitis increases.
Clinical manifestation of nonallergic rhinitis in children
Children with nonallergic rhinitis are known to experience milder nasal signs and/or symptoms, and nonallergic rhinitis takes a less severe clinical course than allergic rhinitis [19-21]. Signs and/or symptoms of nonallergic rhinitis seem to be transient, while those of allergic rhinitis persist; this disparity increases with age [15].
Comorbidities of nonallergic rhinitis in children
Studies reported that rates of comorbid disorders were lower in children with nonallergic rhinitis than in those with allergic rhinitis [3,18]. Nevertheless, children with nonallergic rhinitis have a higher prevalence of various concurrent disorders than the general pediatric population. Various comorbidities are involved in children with nonallergic rhinitis, including asthma, eczema, allergic conjunctivitis, otitis media, bronchial hyperreactivity, sinusitis, sleep disturbances, and bronchial hyperreactivity (Table 2).
Eczema and asthma often coexist in rhinitis children with or without immunoglobulin E (IgE) sensitization, which has been proven in slightly more than one third of children with comorbidities [22]. This finding implies that IgE sensitization may not be the dominant causal mechanism of comorbidity for these diseases. Concurrent diagnosis and comorbidities among children in different rhinitis groups are shown in Table 2. Taken together, these data suggest that children with allergic rhinitis have higher rates of some IgE-mediated comorbidities (asthma, food hypersensitivity, oral allergy syndrome, and bronchial hyperactivity), whereas other comorbidity rates are similar or even higher in children with nonallergic rhinitis. Further studies are necessary to understand the comorbidities in patients of different ages and with natural courses of nonallergic rhinitis in the general population.
Actual considerations for recurrent or chronic rhinitis in children younger than 2 years of age
Despite the availability of weak data regarding infantile rhinitis, previous studies have reported that the prevalence of nonallergic rhinitis in children younger than 2 years is estimated at 1.9%–9.0%. In a study from Singapore, 66.7% of the chronic rhinitis patients under 2 years of age were diagnosed with nonallergic rhinitis, while up to 15.5% were diagnosed with nonallergic rhinitis after 6 years of age, which was higher than the reported estimated prevalence among patients younger than 2 years of age [9].
A birth cohort from France reported that 9.1% (169 of 1,850) of the enrolled children aged 18 months experienced rhinitis symptoms, which were significantly associated with a parental history of allergic rhinitis in early childhood. Among them, only 5.5% (9 of 169) were sensitized to inhalant allergens, while the remainder of the patients were categorized as having nonallergic rhinitis according to their clinical features and laboratory findings [23]. Another birth cohort from Singapore (Growing Up in Singapore Towards healthy Outcomes) of young infants aged 18 months reported that prolonged/recurrent rhinitis was significantly associated with parental atopy, but not with allergen sensitizations [24]. Rhinitis symptoms in early childhood are common, but epidemiological data are insufficient. Data derived from population-based birth cohort studies suggest that a complex association may exist between rhinitis symptoms and family history, comorbidities, allergen sensitization, and aggravating/risk factors and that children with atopic tendency may be prone to developing allergic rhinitis later in life. Since sensitization is difficult to prove using objective tools (e.g., skin prick test) in early childhood, the possibility of underestimation in children who are actually atopic must also be considered in managing children diagnosed with nonallergic rhinitis during the second year of life.
Local allergic rhinitis in children
Local allergic rhinitis (LAR), a rhinitis phenotype recognized during the last decade, is characterized by allergen-related nasal hyper-reactivity despite the absence of specific systemic atopy and is diagnosed using specific nasal provocation tests (NPTs) [5,25]. A systematic review of 46 adult studies showed positive NPT results of 86.3% and 24.7% for allergic rhinitis and nonallergic rhinitis, respectively [26]. According to these studies, LAR is a common disease that affects more than one fourth of adolescents and adults with nonallergic rhinitis.
Only a few studies on the prevalence of LAR in children have been conducted; thus, it is not well established compared to that in adults. The median prevalence of LAR in children previously diagnosed with nonallergic rhinitis was 44.4% (mean, 41.6%; range, 3.7%–66.6%) according to available studies [25,27-32].
The clinical aspects of LAR are similar to those of allergic rhinitis; thus, significant conversions to LAR and systemic atopy were reported, while no evidence of LAR and the development of systemic atopy was observed in another study [33,34]. However, LAR seems to be related to future complications, which implies the possibility of LAR evolving into systemic allergic diseases [34,35].
Idiopathic rhinitis
Idiopathic rhinitis, previously known as vasomotor rhinitis, is also known as noninfectious nonallergic rhinitis [36]. This phenotype has a multifactorial etiology that is triggered by cold air, particulate matter, and nonspecific chemicals. Links to irritant exposures including air pollution and secondary cigarette smoke were also reported in children [19,22,37]. Moreover, they induce a non–IgE-mediated inflammatory response and hyper-reactivity to mucosal components or neural innervations of the nose [36].
Autonomic nervous system imbalance is a consistent finding in idiopathic rhinitis patients. Swelling of the nasal mucosa by cold stimulation results in this dysfunction versus vasoconstriction of the nasal mucosa in healthy controls. Dry cool air stimulation causes the largest reactions of the nasal mucosa in patients with idiopathic rhinitis versus allergic rhinitis patients and normal controls [38]. Autonomic nervous system dysfunction was significant in patients with idiopathic rhinitis versus controls [39]. Meanwhile, an ultrastructural study showed similarities between allergic rhinitis and idiopathic rhinitis. In idiopathic rhinitis patients, evidence of eosinophilic nitric oxide involvement or a better response to capsaicin treatment was not seen, suggesting underlying peptide–neurogenic inflammation [40].
Idiopathic rhinitis is a diagnosis of exclusion whose mechanism is not fully understood. Since it is diagnosed in a significant portion of rhinitis patients, revealing its etiology will enable better treatment options [41].
Nonallergic rhinitis with eosinophilia syndrome
NARES (also called eosinophilic nonallergic rhinitis) is defined as chronic rhinitis with nasal eosinophil infiltration but no IgE sensitization [36]. Although there are different criteria for the diagnosis of NARES, the distribution of >20% nasal eosinophils is mandatory [42,43]. Originally described in the 1980s, NARES is estimated to affect 13%–33% of adult patients with nonallergic rhinitis [42,44].
Pediatric data on prevalence of NARES are scarce. Rupp and Friedman et al. [45] recorded 12 children with perennial rhinitis and nasal eosinophilia. Crobach et al. [46] assessed rhinitis patients aged 12 years or older by nasal smear and showed that 38 of the 363 subjects (10.5%) had nasal eosinophilia (≥10%). Subsequently, they estimated the prevalence of NARES as being approximately 2%. Thus, NARES in children seems to be rare, even more so in early childhood [46].
Hormonal rhinitis
Hormonal rhinitis related to hormonal changes accompanying pregnancy, menstruation, hypothyroidism, and acromegaly is rare in children [47]. Hormones induce vascular engorgement in the nose and cause rhinitis symptoms. Placenta growth hormones, in particular, are involved in nasal symptoms [48]. Estrogen is widely acknowledged to cause nasal congestion and engorgement [48-50]. Progesterone stimulation also reportedly causes nasal congestion, but this remains controversial [51]. However, earlier studies have suggested that changes in sex hormones that occur during the menstrual cycle affect the nasal mucosa, eventually causing nasal symptoms [52]. In contrary, testosterone inhibits eosinophil adhesion to mucosal endothelial cells, thereby leading to decreased eosinophilic activity and viability [47].
Gustatory rhinitis
Gustatory rhinitis is associated with the ingestion of hot or spicy foods. Excessive rhinorrhea occurs during food ingestion and begins within a few minutes after eating spicy food. Materials in spices such as capsaicin (8-methyl-n-vanillyl-6-nomamide) stimulate afferent sensory nerves in the oral and oropharyngeal mucosa, thereby triggering gustatory rhinitis. Its diagnosis relies on nasal symptoms reproduced while eating the causative food, and it is treated prophylactically with topical atropine [53-55].
Although observed in all age groups, gustatory rhinitis has a peak prevalence at 20–60 years [54]. However, the prevalence of gustatory rhinitis in pediatric populations remains unknown since children rarely ingest or use the causative agents (i.e., foods, smoking, or perfumes) and provocation tests for definite diagnosis are not performed as easily as in adults.
Increase in rhinitis and possible links with pollution
Ambient air pollutant levels are higher in many Asian countries than in developed Western countries [56]. The current increase in allergic and nonallergic rhinitis in developing countries may be attributable to these changes accompanied by the modernization process [56]. Attention has increased on airborne particular matters (PM) with diameters less than 10 μm (PM10) and 2.5 μm (PM2.5) because they are suspected to be related with human health such as increased risks for cardiovascular diseases, diabetes, and adverse birth outcomes. Studies have questioned these associations, however, risk assessments of the fine particles affecting rhinitis are scarce. While traffic-related air pollution (TRAP) has been widely acknowledged to increase the risk for rhinitis, studies on PM exhibited conflicting results. A cohort study of 2,598 children suggested that exposure to TRAP during pregnancy and the first year of life was related with the prevalence of allergic rhinitis later in childhood [57]. According to a crosssectional study from France, increased intranasal eosinophil levels were observed in asthma patients exposed to fine particles such as PM2.5, but not in children without asthma [58]. Large-scale nationwide surveys from Taiwan suggested that fine PM and O3 were not significantly related with an increased prevalence of rhinitis, whereas strong associations were observed between TRAP such as nitrogen oxides, carbon monoxide, and ozone [59,60]. Another study conducted in Taiwan concluded that children exposed to significantly higher levels of SO2 had a significantly higher incidence of allergic rhinitis at 12 years postexposure [61].
In a Chinese study that investigated 3,358 preschool children, gestational and early life exposure to NO2 was significantly associated with increased risks of rhinitis, whereas no significant association was found with exposure to SO2 and PM10 [62]. Likewise, no causal link between PM10 levels and allergic rhinitis was revealed by a Korean study using geographic information system–based spatial modeling [63]. Meanwhile, possible relationships between the occurrence of rhinitis and high pollutant levels were implied in a prior study, which found that high pollutant levels were likely to lead to allergic rhinitis within the following week [64]. A large-scale study of approximately 70,000 United States children suggested that children living in areas with exposure to higher levels of O3 and PM2.5 were at higher risk of developing allergic rhinitis than children with lower exposure [65]. In a study that combined daily visits of allergic rhinitis patients consulting general practitioners and daily concentration of air pollutants, higher concentrations of PM10 and other air pollutants were associated with an increased likelihood of hospital visits [66]. Korean studies yielded similar results. One study including 5,443 children reported that living near a main road was associated with an increased prevalence of rhinitis, findings that were supported by another large-scale study of nearly 3,000 children [67,68].
Meanwhile, negative results have also been reported. A Canadian study of an elderly population suggests that air pollution exerts only minor effects on the exacerbation of rhinitis [69]. Health effects to allergic and nonallergic rhinitis caused by air pollution have become a sensitive topic in Asia, including South Korea and adjacent countries that have experienced rapid urbanization over the past few decades since ambient pollutant concentrations are higher than in other Western countries. Air pollutants represented by TRAP and fine PM exert a decisive effect on rhinitis throughout life, even during pregnancy. Investigations regarding relationships between pollutants and rhinitis are being reported constantly; however, the diagnosis of rhinitis in prior studies mainly relied on questionnaires, and rhinitis phenotypes were easily overlooked [70]. Therefore, the majority of studies were unable to reveal the diverse effects of air pollutants to allergic and nonallergic rhinitis. Additionally, metabolic activation, oxidative stress, mutagenicity/genotoxicity, and inflammation/immunity disorder caused by air pollutants have great potential to cause unfavorable effects in the respiratory system, but little is known about the relationship between these respiratory morbidities and rhinitis [71]. Few studies have focused on the influence of air pollution; most studies concentrated on its effects on the lower respiratory tract rather than the upper respiratory system. Further realistic investigations may be beneficial to increase our understanding of the relationship between rhinitis and air pollution.
Diagnosis of nonallergic rhinitis
Clinicians should consider that children with chronic rhinitis who are nonatopic may have nonallergic rhinitis [7]. To differentiate among the subtypes of nonallergic rhinitis, nasal provocation with allergen, microbiologic and cytologic evaluation, and IgE measurements of the nasal cavity are available in the clinical setting [7]. Previous studies have suggested that NPT is a feasible and safe tool with high diagnostic value in children with rhinitis; in this regard, LAR in children has been diagnosed using this method [25,30,72]. Recent studies have standardized and validated NPT in children [73,74]. Furthermore, NPT using Dermatophagoides pteronyssinus extract may be used to diagnose LAR [75]. Nasal hyper-reactivity to nonspecific triggers is a typical sign in idiopathic rhinitis, but is not routinely utilized in clinical practice for children [7,76].
Treatment of nonallergic rhinitis
Management of nonallergic rhinitis is essential due to its persistent and usually lifelong course. However, the treatment strategy of nonallergic rhinitis remains imprecise since its pathogenesis is poorly understood [6,76].
1. Conventional treatment strategies with remedy used in allergic rhinitis
In LAR and nonallergic rhinitis, the combination of intranasal antihistamine and intranasal corticosteroid is the most effective treatment. Symptoms of drug-induced rhinitis caused by autonomous system abnormality induced by nonsteroidal antiinflammatory drugs may be relieved by the administration of ipratropium bromide or capsaicin [6].
In a clinical trial of 59 adults with rhinitis, intranasal corticosteroids effectively treated both allergic and nonallergic rhinitis [77]. Another clinical trial of children aged 12 years and older reported that intranasal corticosteroid was more effective in nonallergic rhinitis patients than in the control group regardless of eosinophilia [78]. Intranasal corticosteroids were reportedly effective in rhinitis patients with nasal eosinophilia [79]. NARES is also known to respond relatively well to intranasal corticosteroids [36,44]. However, other studies yielded different results regarding the effects of intranasal corticosteroid on nonallergic rhinitis. The application of intranasal corticosteroids in adolescents and adults with nonallergic rhinitis for longer than 8 weeks did not relieve rhinitis symptoms [80]. Intranasal corticosteroids as a treatment option for nonallergic rhinitis subtypes with overlapping pathophysiology of allergic rhinitis may be alluded to by these clinical trials.
2. Combination of intranasal corticosteroid and azelastin
A combination nasal spray of azelastin hydrochloride and fluticasone propionate (MP29-02; Dymista, Meda Pharmaceuticals, Somerset, NJ, USA) was recently confirmed as an effective and safe medication regimen [81,82]. Azelastin desensitizes transient receptor potential (TRP) channels, while fluticasone propionate inhibits degranulation of mast cells [83]. TRP channels are distributed broadly throughout the nasal mucosa and known to be involved in the response to multiple stimuli including temperature and osmolality changes. Furthermore, TRP channel dysfunction in respiratory disease leads to alterations in vascular permeability, inflammation, mucus secretion, and mucociliary clearance. Recently, therapeutic agents that modulate specific TRP channel function have been introduced in various respiratory diseases, while the transient receptor potential vanilloid 1 (TRPV1) channel is reportedly upregulated in idiopathic rhinitis [84]. The combination spray MP29-02, which reduces nasal hyper-reactivity, is widely acknowledged to be effective in allergic rhinitis patients [83,85]. In children, a 3-month maintenance duration of MP-AzeFlu exerted a more immediate response than fluticasone propionate alone [86]. Complete response, defined as a 100% reduction in evening reflective Total Nasal Symptom Score, was observed 9 days earlier during the first 28-day treatment period in 7 of 10 patients on the combination therapy. A previous study concluded that the long-term use of MP29-02 was superior to monotherapy with intranasal corticosteroid in patients with chronic rhinitis and that those with significant components of nonallergic rhinitis might benefit from this combination therapy considering the overlapping mechanisms [87,88].
Capsaicin TRPV1 agonist has also been tested for the treatment of nonallergic rhinitis [89]. The first controlled trial demonstrated that a 2-week regimen of intranasal capsaicin, a TRPV1 agonist, rapidly and safely improved symptoms in rhinitis subjects with significant nonallergic rhinitis components [90].
3. Combination of intranasal corticosteroid and decongestant
An alpha-adrenoceptor with a rapid onset and long duration of action is effective for nasal congestion [91]. However, rhinitis medicamentosa, tachyphylaxis, and rebound congestion are obstacles related to its long-term administration [92]. An animal study of combination xylometazoline and intranasal corticosteroid showed the benefit of minimizing long-term usage due to associated congestion, edema, inflammatory cell infiltration, epithelial degeneration, and nasociliary loss in the nasal mucosa of rabbits [93]. The 14-day administration of intranasal corticosteroid to patients with rhinitis medicamentosa resulted in significant symptom improvement [94]. Intranasal corticosteroid treatment in allergic rhinitis patients after a 3-week maintenance regimen with oxymetazoline eliminated rebound congestion within 48 hours versus 1 week in the placebo group [95]. In healthy adults, intranasal corticosteroid improved tachyphylaxis and congestion after oxymetazoline treatment [96]. The combination treatment regimen of intranasal corticosteroid and oxymetazoline also improved nasal obstruction, hyposmia, peak flow, total polyps score, and nasal mucociliary clearance time in adults with nasal polyps [97]. A recent study of intranasal corticosteroid nonrespondent children with adenoid hypertrophy and allergic rhinitis showed that the combination of intranasal corticosteroid and oxymetazoline significantly increased nasal volume [91]. Several recent studies proved that the concomitant use of intranasal decongestant with intranasal corticosteroid might prevent rhinitis medicamentosa while improving nasal congestion versus monotherapy in patients with nonallergic rhinitis [88].
Given the results of the studies mentioned above, the combined use of nasal steroids and decongestants might be more effective than nasal steroids alone.
4. Other treatments
The use of radiofrequency ablation for the treatment of inferior turbinate hypertrophy was studied in patients with allergic versus nonallergic rhinitis. Low-frequency kinetic oscillation stimulation of the nasal mucosa may be an effective short-term treatment for nonallergic rhinitis patients with nasal congestion [88].
The presence of inflammation in the pathogenesis of rhinitis is an important guide in the creation of treatment strategies. Various mediators including histamine, TAME esterase, kinins, prostaglandins, and leukotrienes released in cold stimulation may be considered in nonallergic rhinitis patients responding less to antihistamine or intranasal corticosteroid [36]. The administration of intranasal corticosteroid or antihistamine may have no effect if the main mechanism of rhinitis is not an inflammatory response.
Conclusion
Here, we reviewed the published literature regarding the prevalence, phenotypes, comorbidities, and treatment of nonallergic rhinitis in children. The prevalence and etiology of some phenotypes of nonallergic rhinitis in children are well defined, but information about other rhinitis phenotypes remains uncertain. Moreover, data of nonallergic rhinitis in early children are scarce. Prospective randomized double-blind placebo-controlled crossover studies are warranted to ensure our comprehensive understanding and enable clinicians and scientists to provide accurate diagnostic and therapeutic approaches to childhood nonallergic rhinitis.
Notes
No potential conflict of interest relevant to this article was reported.