Pollen-food allergy syndrome in children
Article information
Abstract
Pollen-food allergy syndrome (PFAS) is an immunoglobulin E-mediated immediate allergic reaction caused by cross-reactivity between pollen and the antigens of foods—such as fruits, vegetables, or nuts—in patients with pollen allergy. A 42.7% prevalence of PFAS in Korean pediatric patients with pollinosis was recently reported. PFAS is often called oral allergy syndrome because of mild symptoms such as itching, urticaria, and edema mainly in the lips, mouth, and pharynx that appear after food ingestion. However, reports of systemic reactions such as anaphylaxis have been increasing recently. This diversity in the degree of symptoms is related to the types of trigger foods and the characteristics of allergens, such as heat stability. When pediatric patients with pollen allergy are treated, attention should be paid to PFAS and an active effort should be made to diagnose it.
Key message
The prevalence of pollen-food allergy syndrome (PFAS) in Korean children with pollen allergy was recently reported to be 42.7%. PFAS can cause a wide range of symptoms from mild allergy to severe anaphylaxis depending on the nature of food allergens that share the epitopes with pollen. Cases of anaphylaxis caused by PFAS have recently increased. Treatments for PFAS should be individualized for patients according to the severity of symptoms.
Introduction
Pollen-food allergy syndrome (PFAS) is an immunoglobulin E (IgE)-mediated immediate allergic reaction that occurs after ingesting fruits, vegetables, or nuts that have cross-reactivity with pollens in patients with pollinosis. PFAS is often called oral allergy syndrome because symptoms appear mainly in the lips, mouth, and pharynx. However, PFAS is a more appropriate term because the symptoms appear around the mouth as well as in the gastrointestinal tract and respiratory organs; in rare cases, it can even cause anaphylaxis [1,2]. Pollen sensitization is uncommon in young children, and PFAS occurs more frequently in older children, adolescents, and adults. However, as pollen sensitization has recently increased in Korean children [3], an interest in and examination of PFAS has become necessary in children. This article analyzes and summarizes the existing literature regarding the epidemiology, clinical patterns, causes, mechanisms, diagnosis, and treatment of PFAS.
Epidemiology
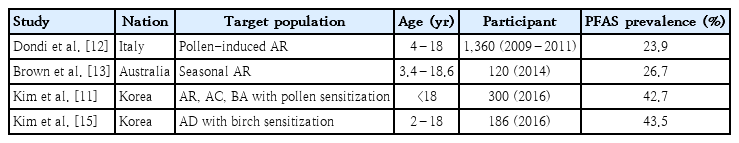
Tuft and Blumstein [4] first reported on the existence of a labile antigen in fresh fruits in 1942, while Amlot et al. [5] first used the term “oral allergy syndrome” in 1987. The prevalence of PFAS varies among regions and pollen type. In Europe, its prevalence is reportedly 40%–50% [5-7] in patients with pollen allergy and approximately 20% in the Mediterranean region, a birch- and ragweed-free area [8]. The prevalence of PFAS depends on the characteristics of the target population. The prevalence of PFAS is usually determined in populations with pollen allergy or sensitization. A survey of patients with pollen allergy in Sweden reported that PFAS was present in 70% of individuals with a birch pollen allergy and 19% of those with other pollen allergies [9]. According to Korean data, the total prevalence of PFAS was 34.6%, while the prevalence of PFAS in cases of tree pollen sensitization accounted for 48% and that in patients with grass or weed pollen sensitization accounted for 13% [10]. A recent Korean multicenter study reported that the prevalence of PFAS was 41.7% in patients with pollen allergy, 20%–30% in cases of sensitization to summer or autumn pollen alone, and higher than 50% in simultaneous sensitization to spring and autumn pollen or sensitization to all spring, summer, and autumn pollens [11].
Data on the prevalence of PFAS in children are limited. The prevalence of PFAS has been reported differently depending on the country, target group, and reporter. A 2014 study in Italy reported a 24% prevalence in children with allergic rhinitis by pollen [12]. In Australia in 2014, it was 14.7% in 163 patients aged 4–17 years [13]. In Mexico in 2016, it was 8.9% in 267 children aged 6–14 years with atopic diseases [14]. In a recent national-level multicenter study in South Korea, the prevalence of PFAS was 42.7% in pediatric patients with pollen allergy [11]. A recent Korean study of children with atopic dermatitis and birch pollinosis reported that the prevalence of PFAS was 43.5% in children aged 2–18 years with atopic dermatitis sensitized to birch pollens and as high as 36.6% in preschoolers aged 2–7 years [15]. Studies on the prevalence of PFAS in children are summarized in Table 1.
Clinical manifestation
Many patients with PFAS first experience rhinoconjunctivitis because the specific IgE antibody to pollen becomes the source of cross-reactivity with the fruit or vegetable allergen. Most symptoms of PFAS are limited to the lips and oral mucosa— areas that directly touch the food —but in some patients, they can progress to systemic symptoms, such as systemic urticaria, angioedema, airway edema, cough, dyspnea, vomiting, and abdominal pain (Fig. 1). One study reported that systemic symptoms other than the digestive system were noted in 8.7% of pati ents with PFAS, while anaphylaxis occurred in 1.7% [16]. A recent Korean multicenter study reported that PFAS caused respiratory symptoms such as coughing and dyspnea in 20.2% of patients, digestive symptoms such as vomiting and abdominal pain in 10.7%, and anaphylaxis in 8.9% [2]. In 2003, a survey of PFAS for allergists was reported in the United States, and the doctors estimated a much lower prevalence than what was actually published. The authors argued that it reflects a low index of suspicion and underdiagnosis of PFAS [16]. Cases of severe systemic symptoms caused by PFAS in Korea have recently increased. Therefore, physicians must pay special attention to this issue.
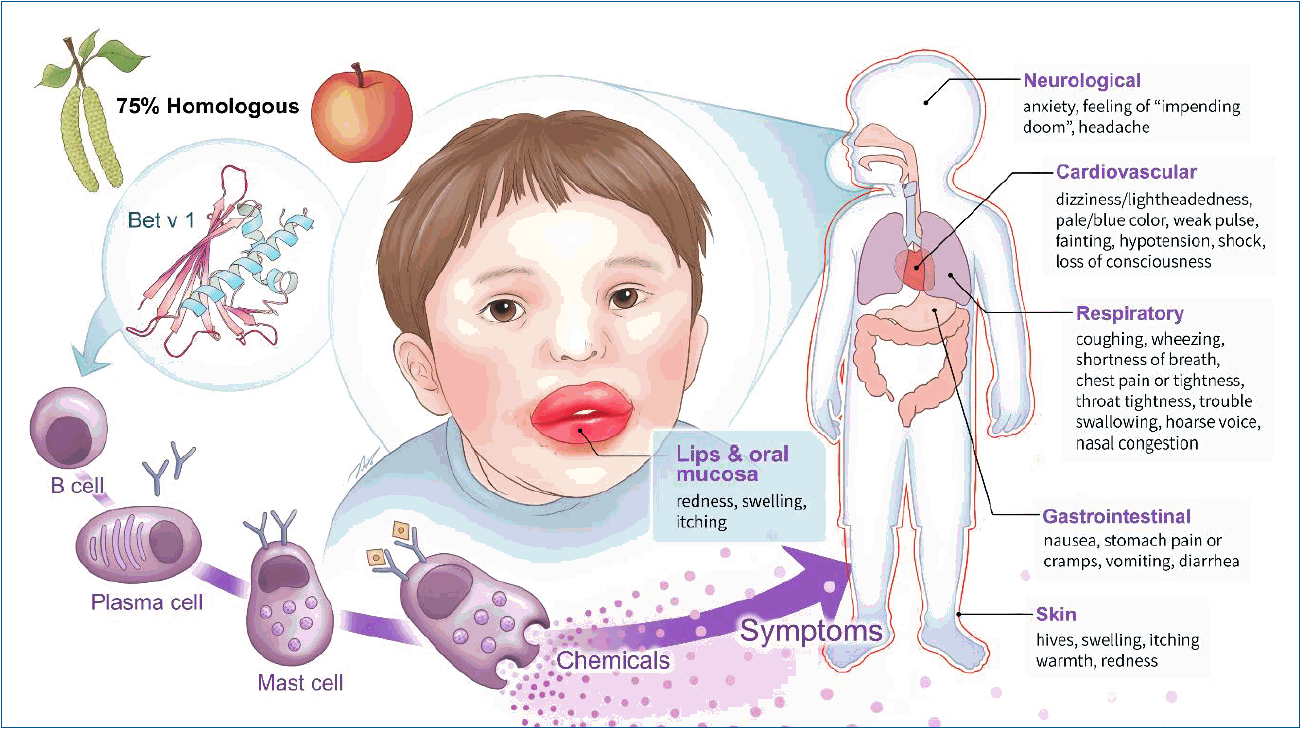
The symptoms of pollen-food allergy syndrome primarily include a rash or itching sensation around the lips; however, it can also cause systemic symptoms such as a whole-body rash, respiratory difficulty, coughing, wheezing, vomiting, abdominal pain, low blood pressure or paleness, headache, and dizziness.
Cross-reactivity between pollen and food
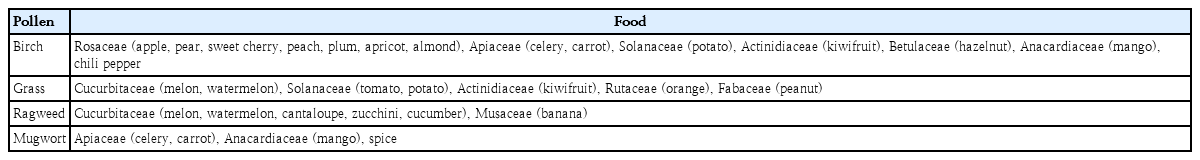
PFAS is a phenomenon that occurs because of cross-reactivity between pollen and food. Cross-reactivity occurs when allergens share epitopes. Pollen-specific IgE can recognize homologous food allergens that share the same epitopes [1,17-20]. Birch pollen, a representative spring pollen, shows cross-reactivity with many food allergens but more frequently with members of the Rosaceae family, such as apple, pear, sweet cherry, peach, plum, apricot, and almond. Apple has such a prominent clinical relevance with birch that the term apple-birch syndrome is in use. It also has cross-reactivity with celery, carrot, potato, kiwifruit, hazelnut, and mango. In a recent domestic study, the food that caused PFAS most frequently in children aged 7 years or older with atopic dermatitis sensitized to birch was apple and the most frequent food was kiwifruit in children aged 2−7 years [15]. Grass, a summer pollen, shows cross-reactivity with melon, water melon, tomato, potato, kiwifruit, orange, and peanut. An autumn pollen, ragweed has cross-reactivity with melon, watermelon, cantaloupe, zucchini, cucumber, and banana, while mugwort has cross-reactivity with celery, carrot, mango, and spices [15,21-25]. The PFAS caused by cross-reactivity between autumn pollen and various foods is called celery-mugwort-spices syndrome and ragweed-melon-banana syndrome (Table 2) [26].
Antigens causing PFAS
Food allergens causing IgE sensitization in IgE-mediated food allergy are divided into 2 classes by the sensitization path way. For class 1 food allergens, IgE sensitization occurs after exposure through the gastrointestinal tract. All foods can belong to this class, but major ones are eggs, milk, peanuts, nuts, fish, soybeans, wheat, and crustaceans, which are the major causes of food allergies in infants and children and often lead to systemic reactions or anaphylaxis. Class 2 food allergens are structurally homologous with plant proteins such as pollen allergens sensitized through the respiratory organs. They cause problems mostly after adolescence and mainly cause mild symptoms or PFAS. However, systemic reactions cannot be excluded and ana phylaxis may occur depending on the allergen and individual [25].
Allergens causing PFAS are largely classified into pathogenesisrelated protein (PR)-10 family, profilin, lipid transfer protein (LTP), and cross-reactive carbohydrate determinants (CCD) (Table 3) [25]. The PR-10 family is the most important and representative allergen causing PFAS. Bet v 1, a major allergen of birch, belongs to this family. Hence, it is also called the “Bet v 1 group.” This protein increases when plants are in a stressful situation and exists in most foods. Mal d 1 of apple, Pru ar 1 of apricot, and Pru av 1 of sweet cherry belong to the Bet v 1 group and cause cross-reactivity between birch pollen and Rosaceae family fruits [2,21,25]. Bet v 1 often causes symptoms limited to the mouth because it easily loses antigenicity upon exposure to heat and digestive enzymes.
In contrast, Api g 1 of celery and Gly m 4 of soybean are allergens that mainly cause systemic symptoms despite belonging to the same PR-10 family. Celery and soybean allergies are reportedly accompanied by systemic reactions, including anaphylaxis, in more than half of sensitized patients. Api g 1, the main allergen of celery, is strong against heat, and Gly m 4, the main allergen of soybean, is vulnerable to fermentation but strong against heat. The characteristics of these allergens seem to cause severe allergic reactions [27-30].
Profilin, an actin-binding protein of 12–15 kDa, is involved in maintaining the cytoskeleton. It exists in a variety of plants and foods. Bet v 2 of birch, Art v 4 of mugwort, and Api g 4 of celery belong to this family [2,21,25]. The cross-reactivity of celerybirch/mugwort-spices, grass pollen-celery-carrots, and tree pollen-hazelnut is caused by profilin [31-33]. Similar to Bet v 1, the antigenicity of profilin is decreased by heat and digestive enzymes [2].
LTP belongs to the PR-14 family. Examples include Art v 3 of mugwort, Pru p 3 of peach, Pru ar 3 of apricot, Pru av 3 of sweet cherry, and Mal d 3 of apple [2,25]. Unlike most other allergens causing PFAS, LTP is stable to heat and digestive enzymes and can cause class 1 or class 2 food allergies [16,34]. The stability of LTP makes it reach the gastrointestinal immune system and enable an allergenic conformation, and it can cause sensitization and systemic symptoms. As a result, LTP can more easily cause severe systemic reactions, such as systemic urticaria, angioedema, dyspnea, and anaphylaxis, than the PR-10 family [35,36]. Severe allergic reactions by LTP are often triggered by physical exercise and nonsteroidal anti-inflammatory medications [37].
CCD, a glycoprotein that contains N-linked glycan and has IgE-binding ability, can cause wide-ranging cross-reactivity with different species. It exists in celery, tomato, potato, peanut, ragweed, and birch. However, it is unclear whether all of these antigens cause clinical symptoms [30,32,38].
Diagnosis
The most important factor in diagnosing PFAS is to collect a detailed history. Skin prick tests (SPTs) and serum-specific IgE antibody measurements show large differences in sensitivity and specificity depending on the type of trigger foods. The commercial food extracts used in SPTs can cause unexpected pro tein denaturation in the antigen manufacturing process and show relatively low sensitivity and specificity in the SPTs of fruits and vegetables. Therefore, a prick-to-prick test is sometimes used, which directly tests the individual using fresh fruits and vegetables. In general, for apple, orange, tomato, carrot, celery, sweet cherry, and peach, the prick-to-prick test shows higher specificity and sensitivity than SPTs using a commercial food extract or serum-specific IgE antibody measurement using a radioallergosorbent test [39,40]. A component-resolved diagnosis using microarray techniques can also be used, but it remains difficult to distinguish between a sensitization and a true allergy [41]. If the findings of a clinical history and allergy test are ambiguous, PFAS can be confirmed through an oral food challenge test [25].
Treatment
The primary treatment of PFAS is to avoid the trigger foods [42]. Food allergies were reportedly lost in approximately one third of some adult patients after the complete restriction of trigger foods for 2 years. However, the general opinion is that food restriction cannot be a treatment to fundamentally eradicate PFAS because pollen, rather than food allergens, causes the cross-reactivity. Heating the trigger foods before eating them can be another solution because most fruits and vegetables causing PFAS are vulnerable to heat, which decreases allergenicity [1,43-45]. However, this requires caution because heating is not effective for heat-stable allergens such as those in the LTP family, Api g 1 of celery, and Gly m 4 of soybean. Furthermore, to the contrary, when peanuts are roasted, allergenicity increases because the IgE-binding capacity of the main allergens of peanuts, Ara h 1 and Ara h 2, increases [1]. Risk factors for systemic symptoms include a history of systemic reactions to one of the related foods, reactions to cooked form of the food, positive reaction in the SPT with a commercial food extract, no sensitization to relevant pollen allergens, and peach allergy [16]. For patients with PFAS and a history of anaphylaxis, a self-injectable epinephrine should be prescribed [31]. The role of immunotherapy as a treatment for PFAS is controversial. One study found that 84% of patients experienced the disappearance or significant reduction of apple allergies after immunotherapy with birch pollen extract. However, 30 months after the cessation of immunotherapy, many patients showed a tendency toward recurrence [45,46]. Some studies have reported improved symptoms of food allergies after pollen immunotherapy, whereas others have failed to prove their effects [47-50].
Conclusion
The prevalence of PFAS in Korean children with pollen allergy was recently reported to be 42.7%. In a study of children sensitized to birch pollens, the prevalence was as high as 36.6% in those aged 2–7 years. Furthermore, a recent multicenter study in Korea reported that the incidence of anaphylaxis was 8.9% in patients with PFAS, which is much higher than that reported in the past. PFAS has not received much attention historically because physicians have considered it a mild transient disease. However, our findings demonstrate that pediatric patients with pollen allergy should be interviewed in detail to screen for PFAS. In cases of doubt, an active examination is required. Treatments for PFAS should be individualized for patients because it can cause a wide range of symptoms from mild allergy to severe anaphylaxis.
Notes
No potential conflict of interest relevant to this article was reported.
See the commentary “Insights into pediatric pollen food allergy syndrome” via https://doi.org/10.3345/cep.2019.01179.