Air pollution and childhood obesity
Article information
Abstract
Childhood obesity is a global health concern. Air pollution is also a crucial health threat, especially in developing countries. Over the past decade, a number of epidemiologic and animal studies have suggested a possible role of pre- or postnatal exposure to air pollutants on childhood obesity. Although no clear mechanism has been elucidated, physical inactivity, oxidative stress, and epigenetic modifications have been suggested as possible mechanisms by which obesity develops due to air pollution. In this review, we summarize and review previous epidemiologic studies linking air pollution and childhood obesity and discuss the possible mechanisms underlying air pollution-induced obesity based on in vivo and in vitro evidence.
Key message
Questions: What are the possible effects of air pollution on the occurrence of childhood obesity and what are the mechanisms?
Finding: Epidemiologic studies suggest that air pollutants might act as obesogens in the pediatric population, and their possible mechanisms include oxidative stress, physical inactivity, and epigenetic modulation.
Meaning: This paper reviews updated information on air pollution, one of the modifiable environmental factors in childhood obesity.
Introduction
The global prevalence of childhood obesity has increased almost 8–10 times over the last 30–40 years [1,2]. Childhood obesity can lead to various comorbidities, including type 2 diabetes, hypertension, nonalcoholic fatty liver disease, cardiovascular disease, and even cancer in later life [3]. A rapid increase in the prevalence of obesity has occurred with the markedly increased production of industrial chemicals, suggesting potential causative links [4]. In particular, as more than 90% of children worldwide live in an environment with air pollution levels above the World Health Organization guideline, the link between air pollution and childhood obesity is drawing increasing attention [5]. A number of studies on the effects of air pollutants on childhood obesity were reported in the 2010s. Here we review epidemiologic studies on the association between air pollution and childhood obesity and speculate on the underlying mechanisms.
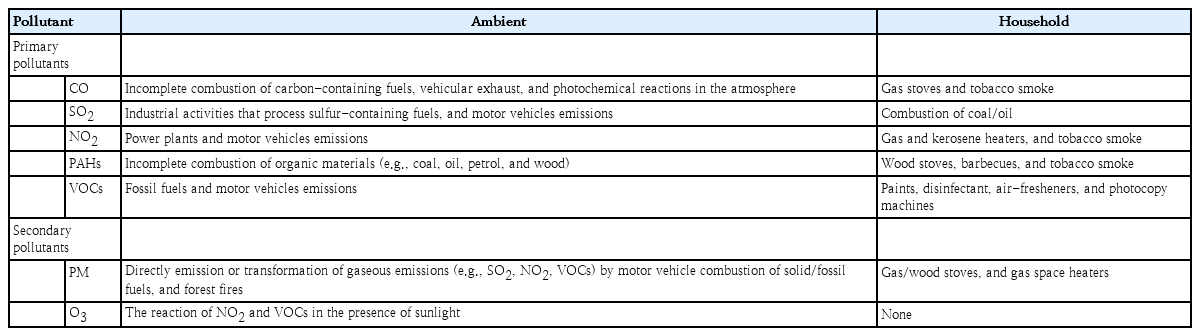
Air pollutants and major sources of exposure
Ambient air pollution is mainly caused by the combustion of fossil fuels, waste incineration, industrial/agricultural processes, and natural processes including thunderstorms and volcanic eruptions [6]. Household air pollution is primarily generated by the incomplete combustion of fossil fuels during cooking, heating, and lighting. Other household air pollutants include tobacco smoke, mold spores, building materials, and volatile organic compounds (VOCs) [6]. The air pollution sources vary among regions according to industrialization degree. In urban areas, the combustion of fossil fuels for energy production is the primary source of air pollution, while in rural areas, the main sources of air pollution are pollutants generated in the household and from incineration for heating, cooking, and waste disposal [7].
Major air pollutants include particulate matter (PM), ozone (O3), carbon monoxide (CO), sulfur dioxide (SO2), nitrogen dioxide (NO2), and polycyclic aromatic hydrocarbons (PAHs). Primary air pollutants are emitted from direct sources (e.g., factories, construction sites, fires, cars, and roads) and secondary pollutants are formed by chemical reactions with other substances in the air [8]. Possible sources of air pollutants are presented in Table 1.
Among air pollutants, PM poses the greatest health concern since it is a complex heterogeneous mixture of all kinds of air pollutants (e.g., nitrates, sulfates, elemental and organic carbon, VOCs, and PAHs), biological compounds (e.g., endotoxin, mold, pollen), and metals (e.g., iron, nickel, copper, and zinc) [9]. There is increasing evidence that the health impact of PM is dependent on its chemical composition [10].
Depending on the aerodynamic diameter of the particles, PM is classified as coarse (PM10), with a diameter of <10 microns, or fine (PM2.5), with a diameter of <2.5 microns [8]. Fine particles generally penetrate the lower respiratory tract more easily, while coarse particles tend to lodge in the upper respiratory tract [8]. Most epidemiologic studies have consistently reported that PM2.5 is the most harmful fraction [11].
Association of air pollution and childhood obesity in epidemiologic studies
1. Prenatal exposure to air pollutants
Human epidemiologic studies on the relationship between prenatal exposure to air pollutants and childhood obesity are listed in Table 2. Longitudinal studies from the U.S. Project Viva cohort consistently reported that traffic density and roadway proximity during pregnancy or at delivery are associated with obesity parameters including body mass index (BMI) and fat mass in children aged 6 months to 10 years [12-14]. This study group also suggested a possible impact of prenatal air pollution exposure on reduced birth weight. However, other studies on the association between individual traffic-related air pollutants and childhood obesity have not verified this association. Exposure to nitrogen oxides (NOx) and elemental carbon was not associated with childhood obesity in cohort studies from Sweden, the USA, and Hong Kong [15-17]. SO2 exposure was even negatively related to BMI in adolescent boys [17].
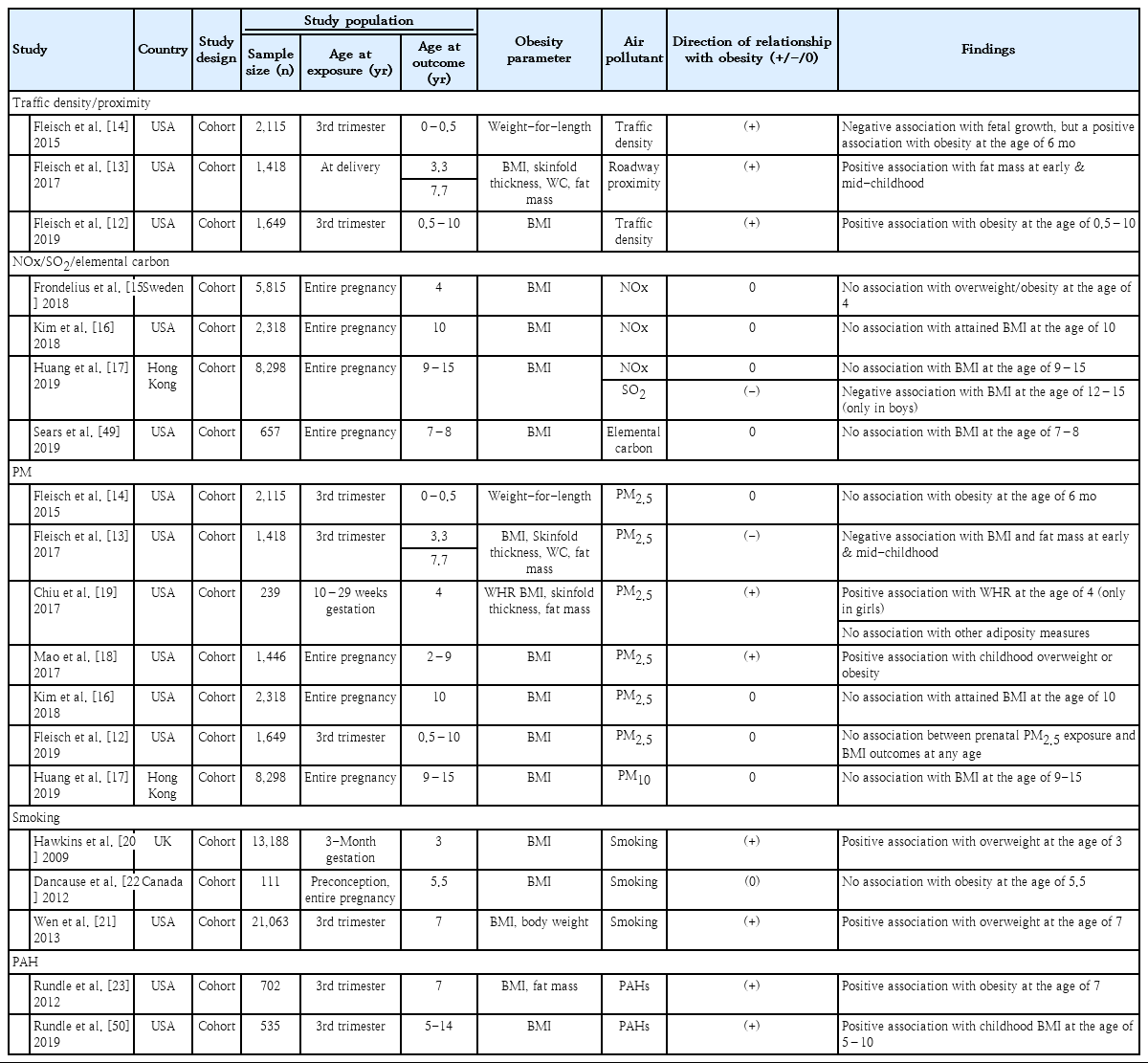
Association between prenatal exposure to air pollution and childhood obesity in human epidemiologic studies
A relatively large number of studies have examined the effects of exposure to PM versus other air pollutants on childhood obesity. Most cohort studies from the USA and Hong Kong reported a null impact of PM2.5 and PM10 exposure during pregnancy on obesity parameters in children and adolescents [12-14,16,17]. Only 2 cohort studies from Boston, Massachusetts, reported a weak association between PM during pregnancy and obesity parameters including BMI at 2–9 years of age and waist-to-hip ratio at 4 years of age [18,19].
Prenatal exposure to tobacco smoke, a representative source of household air pollution, is reportedly related to an increased risk of overweight at ages 3 and 7 years in large-scale national cohort studies from the USA and the UK [20,21]. A small-scale study from Canada reported a null effect of tobacco smoke on BMI in 5-year-old children [22]. PAHs, which are known to be highly correlated to tobacco smoke exposure, were also associated with childhood BMI in 2 cohort studies from New York. In these studies, exposure to PAHs measured by personal air monitoring during pregnancy was positively correlated with BMI in children aged 5–14 years in African-American and Hispanic children [23,24].
2. Postnatal exposure of air pollutants
The impacts of postnatal air pollutant exposure on childhood obesity identified through human epidemiologic studies are presented in Table 3. As with prenatal exposure studies, a positive correlation between residential traffic density/roadway proximity in childhood and BMI at 4–8 years was demonstrated by 2 large cohort studies conducted in Southern California [25,26]. However, a school-based cross-sectional study reported that the positive association between the presence of arterial roads around school and the obesity rates in elementary schoolers was not statistically significant after the adjusting for crime rates and economic levels around the schools [27]. However, other studies on the association between individual traffic-related air pollutants and childhood obesity have not confirmed this association.

Association between postnatal exposure to air pollution and childhood obesity in human epidemiologic studies
Of note, most studies of the association between NOx, a major traffic-related air pollutant, and childhood obesity reported a statistically significant positive correlation. In 3 cohort studies performed in the USA, teenage exposure to NOx concentrations was positively correlated with BMI gain until 18 years of age, while 1-year-old exposure to NOx concentrations was positively associated with BMI gain until 10 years of age [16,28,29]. Recent studies published in the Netherlands and Spain have also reported that residential NO2 concentrations are associated with a higher risk of overweight in childhood and adolescence [30,31]. However, in an Italian cohort study, residential NO2 concentrations measured at birth and age 4 were not associated with obesity at 4 and 8 years of age [32].
Study results on the link between postnatal PM exposure and childhood obesity are inconsistent. In 2 cohort studies from the USA, PM2.5 concentrations in infancy and at 8–15 years of age were positively associated with childhood obesity and BMI at 18 years of age [18,29]. Another cross-sectional study from Spain supported this relationship in children aged 7–10 years [31]. Meanwhile, 3 other cohort studies from the USA, Italy, and the Netherlands reported a null association between PM2.5/PM10 exposure and childhood obesity [16,27,32]. In contrast, a negative association between PM10 exposure in infancy and subsequent poor weight gain during toddlerhood was reported by a cohort study from Korea [33].
Like prenatal smoking exposure, childhood tobacco smoke exposure is positively correlated with increased adolescent BMI [34]. In particular, NOx exposure reportedly has a synergistic effect with tobacco smoke on increasing obesity risk [34]. The relationship between postnatal PAH exposure and obesity development was similar to that between prenatal exposure and obesity development. A cross-sectional study in the USA reported a relatively strong positive correlation between urinary PAH levels and obesity parameters in children aged 6–11 years [35].
Plausible mechanisms by which air pollution affects childhood obesity
1. Physical inactivity
Air pollution increases the likelihood of obesity by inducing sedentary behaviors. Exposure to air pollution can cause cardiorespiratory symptoms such as coughing, shortness of breath, and high blood pressure, impeding outdoor activity by impairing athletic performance [36,37]. Air pollution alarms through various media also influence people’s decisions regarding physical activity [38].
2. Oxidative stress and systematic inflammation
One of the most important mechanisms of interest is the systemic inflammatory reactions that occur through the stimulation of oxidative stress processes. Several animal and human studies demonstrated an increase in proinflammatory cytokines (e.g., interleukin-6 and tumor necrosis factor-α) in the systemic circulation following inhalation exposure to diesel exhaust particles or PM [39-42]. Oxidative stress and mitochondrial damage in adipose tissue caused by air pollutant exposure increase the differentiation of white adipocytes, which store extra energy in the form of triglycerides, and increase the differentiation of brown adipocytes, which release energy as heat [43,44]. This change adversely affects the energy balance in adipose tissue, predisposing the person to obesity and metabolic abnormalities. Further, in utero exposure to air pollutants such as diesel exhausts increases the likelihood of obesity in offspring by causing fetal brain inflammation and a subsequent increase in appetite [45].
3. Hypothalamic-pituitary-adrenal axis
Chronic psychological stress and the subsequent activation of the hypothalamic-pituitary-adrenal (HPA) axis is a well-known risk factor for obesity development and metabolic dysfunction. Recent studies have demonstrated that adrenocorticotropic hormone and glucocorticoid concentrations increase after the inhalation of ozone or PM in animal models. Therefore, chronic activation of the stress response system is also emerging as a possible mechanism of obesity following air pollution exposure [46].
4. Epigenetic modulation
Epigenetic modulation might be a plausible mechanism, especially in cases of prenatal exposure to air pollution. Prenatal long-term exposure to air pollutants reported exerting epigenetic effects including alterations of DNA methylation, microRNAs, and noncoding RNAs and regulation of chromatin [47]. These changes may cause the derangement of the mitochondrial machinery, which is closely related to the control of energy metabolism and inflammation. For instance, a recent study showed that in utero exposure to PAHs induced offspring obesity by hypomethylation of peroxisome proliferator–activated receptor-gamma and activation of various genes associated with adipogenesis in the offspring’s adipose tissue [48].
Conclusions
Previous in vitro and in vivo studies indicated that air pollutants might act as obesogens by inducing physical inactivity and epigenetic modulation and promoting oxidative stress and HPA axis. The effects of air pollution on childhood obesity seem to vary according to pollutant type and components, exposed region and area, exposure measurement methods, and exposure duration. Further, exposure timing, observation duration, sex, and ethnicity may be important variables in the study of the effects of air pollution on obesity. Although longitudinal human studies on the possible effects of air pollution on the development of obesity are increasing, most focus on the effects of individual air pollutants, not the mixed effects of various air pollutants. Future large-scale and long-term follow-up studies considering all these factors are required to determine the effects of air pollution on childhood obesity.
Notes
No potential conflict of interest relevant to this article was reported.