Understanding of type 1 diabetes mellitus: what we know and where we go
Article information
Abstract
The incidence of type 1 diabetes mellitus (T1DM) in children and adolescents is increasing worldwide. Combined effects of genetic and environmental factors cause T1DM, which make it difficult to predict whether an individual will inherit the disease. Due to the level of self-care necessary in T1DM maintenance, it is crucial for pediatric settings to support achieving optimal glucose control, especially when adolescents are beginning to take more responsibility for their own health. Innovative insulin delivery systems, such as continuous subcutaneous insulin infusion (CSII), and noninvasive glucose monitoring systems, such as continuous glucose monitoring (CGM), allow patients with T1DM to achieve a normal and flexible lifestyle. However, there are still challenges in achieving optimal glucose control despite advanced technology in T1DM administration. In this article, disease prediction and current management of T1DM are reviewed with special emphasis on biomarkers of pancreatic β-cell stress, CSII, glucose monitoring, and several other adjunctive therapies.
Introduction
Type 1 diabetes mellitus (T1DM) is a disease characterized by decreased insulin secretion or complete insulin deficiency due to autoimmune beta-cell dysfunction, and it occurs mostly in children and adolescent populations [1]. The risk of complications in T1DM can be managed through intensive glucose control, which sometimes leads to undesirable adverse effects (AEs), including increased risk of weight gain, hypoglycemia, higher insulin doses, and frequent injections [2]. Although insulin medication and glucose monitoring have advanced, patients with T1DM experience difficulties in reaching their target glucose control goals. As a result, studies have been conducted on clinical evidence of adjunctive therapies to insulin therapy in T1DM. Along with this, identification of promising β-cell specific biomarkers is becoming a reality, owing to leading-edge technology. In this article, the current understanding of disease prediction and management of T1DM are reviewed with special emphasis on the following: biomarkers of pancreatic β-cell stress, potential role of continuous subcutaneous insulin infusion (CSII), continuous glucose monitoring (CGM), and several adjunctive therapy.
Incidence of T1DM in Korea
The frequency of T1DM varies depending upon geography, age, sex, family history, and ethnicity. In Europe, Australia, and the Middle East, rates of T1DM are up by 2%–5% per year [3-5]. In a retrospective cohort study in southern part of Korea from 2001 to 2010, the average approximate incidence rate of T1DM was 2.01/100,000 [6]. The reported incidence of T1DM in Korean children and adolescents from 2012 to 2014 was 3.19/100,000 [7].
Genetic factors for T1DM
To date, more than twenty regions of the genome recognized as predisposing factors for T1DM have been recognized; however, only two (human leukocyte antigen [HLA] region, insulin gene region) of them have shown strong evidence in association with the disease [8,9]. HLA genes are well known for their polymorphisms and great variability, such that no specific mutation has been identified as a direct cause of T1DM [10]. Approximately 40% of the genetic risk associated with T1DM is related to the HLA region class II, especially HLA-DR and HLA-DQ, where haplotypes with the greatest association are DRB1 * 0401 or * 0405 and DQB1 * 0301 (DR4-DQ8) [11]. However, less than 20% of cases are related to major histocompatibility complex class I mutations, in which haplotypes HLAB * 3906 or HLA-A * 2402 set susceptibility towards T1DM [10].
Environmental risk factors for T1DM
Epidemiologic studies suggest that environmental factors play a leading role in the development of T1DM in activating the disease [12]. These factors include cow milk protein [13], vitamin D, viral infections, and limited exposure to microorganisms during childhood [14].
1. Study of vitamin D
Vitamin D can shift the balance of the body’s T-cell response toward downregulation of the T-helper-1 immune response [15]. In particular, vitamin D and T1DM vitamin D receptor (VDR) have been considered to play a role in the pathogenesis of T1DM. In 2015, Cheon et al [16]. conducted a study about the contribution of VDR polymorphisms to T1DM susceptibility in a genetically homogenous population in Korea. It was suggested that T and b TaqI and BsmI alleles might be protective against T1DM in Korean subjects. A meta-analysis performed by Qin et al. [17] revealed that the VDR BsmI B allele, bb genotype was closely connected with T1DM risk in Asians, while the bb genotype was linked to T1DM risk in overall populations. There has been skepticism about the effect of vitamin D supplementation as a preventative measure of T1DM. A Norwegian study reported that higher serum 25-hydroxyvitamin D during late pregnancy was related to lower risk of T1DM in the offspring [18], but a Finnish study revealed that there was no association between serum concentrations of 25-hydroxyvitamin D in the first trimester of pregnancy and the risk of T1DM in babies [19]. A retrospective meta-analysis research revealed that infants given vitamin D had lower risks than those without supplementation (odds ratio, 0.71) [20]. The Diabetes Autoimmunity Study in the Young investigated 25-hydroxyvitamin D concentrations in plasma during infancy and throughout childhood and identified no link to islet autoimmunity or advancement to T1DM [21]. However, there is limited supporting evidence from prospective birth cohort studies despite interest in vitamin D supplementation to prevent islet autoimmunity and T1DM.
2. Study of cow’s milk
Most prospective birth cohort studies have not proven association between early exposure to cows’ milk and either islet autoimmunity or T1DM [22-24]. In a double-blind, randomized trial, among 230 infants at risk of T1DM, those given a casein hydrolysate formula showed relatively lower risk of islet autoimmunity than those receiving cow's milk-based formula during the first 6 to 8 months [25]. However, this effect on islet autoimmunity was not confirmed in the larger phase 3 study [26]. Cow's milk ingestion in childhood has been related to both increased risk of islet autoimmunity [27,28] and T1DM [29,30]. Higher intake of cow's milk might boost advancement to T1DM among children with islet autoimmunity [13], which could be mediated by a portion of certain fatty acids in cow's milk and meats [31]. This result could promote a new modality of dietary interventions to stave off T1DM when confirmed by further studies.
3. Study of intestinal microbiota
Some environmental factors of T1DM are interconnected with the human microbiome. Gut microbes affect glucose and lipid metabolism, immunity, and systemic inflammation outside of the intestines [32-34]. T1DM risk might be modulated by commensal microbiota [32,35], but research has been limited. Some have reported lower microbial diversity in children with islet autoimmunity prior to diabetes progression, compared with a healthy control group [35,36]. Larger studies with carefully selected controls are needed using next-generation sequencing of the microbiome at multiple time-points.
4. Study of infections
Recent findings have shown that enteroviral VP1 protein immunoreactivity in the β cells of children with T1DM was more frequently detected than in controls [37,38]. In an in vitro study, it was proposed that continuous enteroviral infection of human pancreatic ductal cells might lessen their ability to transdifferentiate into β cells, thus causing β-cell mass reduction owing to autoimmunity [39].
Biomarkers of pancreatic β-cell stress and death in T1DM
Studies have been conducted to identify circulating biomarkers that dictate islet β-cell stress and death, which could be used to recognize individuals at risk of developing T1DM before the onset of β-cell destruction [40]. In practice, finding β-cell biomarkers has been challenging because β-cells represent a small fraction (≤1%) of the total pancreatic mass [41]. However, separation and identification of β-cell specific/selective biomarkers are becoming available to researchers through advanced sequencing technologies and methodologies [40]. The Belgian Diabetes Registry showed that autoantibody-positive first-degree relatives with random circulating proinsulin/C-peptide (PI/C) ratios above the 66th percentile had an increased risk of T1DM development in 5 years [42]. Recently, it was reported that fasting PI/C ratios within an international cohort of autoantibody-positive family members of individuals with T1DM were significantly higher up to 12 months before the onset of T1DM [43]. Several micro-RNAs (miRNAs) have been also suggested as biomarkers in patients with T1DM. In-depth sequencing of human islets and isolated β-cells has identified several miRNAs, including miRNA-25, with relative enrichment in β-cells [44]. One of the most abundant miRNAs in β-cells, miRNA-375, inhibited glucose-stimulated insulin secretion, and it was increased in serum after allogeneic and autologous islet transplantation [45]. Further, there have been other potential miRNAs elevated in the serum of patients with recent-onset T1DM [44]. One notably potential approach for biomarker advancement has been the identification of differentially methylated DNA fragments [40]. Due to the nearly exclusive manner of gene expression of multiple genes in the islet β-cell, it can translate to potential regulation by selective DNA methylation [40]. In islets and β-cells, human and mouse genes have been known to be hypomethylated at selective CpG sites in the coding and promoter regions [46], and human INS expression is reversely connected with methylation at many of these sites in human islets (Table 1) [47].
CGM in T1DM
It is evident that use of CGM can dramatically improve the quality of glycemic control in T1DM in comparison to self-monitoring of blood glucose (SMBG) [48]. The benefit is especially notable in high-risk individuals with frequent or severe hypoglycemia, often associated with hypoglycemia unawareness [48]. GM can be used effectively with either multiple daily injections (MDI) or with CSII [49-51].
1. Approval for nonadjunctive use
Currently, CGM is approved by the FDA in the outpatient setting as an ancillary instrument, which allows for an increase in information obtained from standard home blood glucose monitoring devices and aids in the recognition of hypoglycemic and hyperglycemic events [52]. The initial approval was given to only one model, the Dexcom G5 sensor [48]. Many CGM users have already found that the CGM provides sufficient information, accuracy, and reliability for insulin dosage adjustments based on the CGM alone [48,51,53].
2. Efficacy of CGM in conjunction with MDI or CSII
Most of the early studies with CGM were conducted on people with T1DM who were also using a CSII. There were questions as to whether similar benefits would be seen in people using basal-bolus therapy with MDI. There is now considerable evidence that improvements in the quality of glycemic control are essentially identical in MDI and CSII users [49-51]. The effectiveness of CGM for people with T1DM using MDI as well as CSII was clearly described in recent randomized clinical trials showing that changes in mean glucose were equivalent for users of MDI and CGM [49,50,54].
3. CGM use in high-risk patients for hypoglycemia
A recent clinical trial assessed the performance of CGM for patients at high risk to experience hypoglycemia. Van Beers et al. [51] conducted a crossover randomized study and demonstrated a dramatic increase in percentage of time in the target range, which was identical for MDI or CSII users, with an accompanying marked reduction in the hypoglycemia frequency.
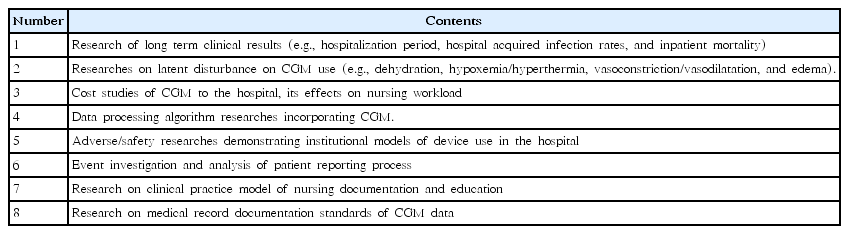
4. CGM studies in hospitalized patients
CGM usage in the inpatient setting and ICU remains a work in progress [55,56]. There has been considerable interest in using CGM in the hospitalized patients for glycemic control, in particular for patients continuing use of outpatient therapies, for control of insulin infusions, and for use in the intensive care unit [48]. Continuous improvements in the accuracy, robustness, and usability of CGM sensors offer a promising outlook for their role in the inpatient setting [48]. Thabit et al. [57] have recently reported substantial improvements in glucose control in a randomized parallel-arm study of 40 inpatients with type 2 DM using a closed-loop control without premeal boluses. Specifically, the proportion of time in the target range increased from 38.1% in the control group to 59.8%, a 21.8% change. In practice, there are safety concerns regarding data analysis. Occasionally, inexperienced experts might make hasty or inappropriate decisions about doses because of the enormity of data output. Further studies are recommended to provide sufficient information about the possible benefits of CGM for hospitalized patients (Table 2). Since CGMs are often used along with insulin pumps, guidelines should be established for both technologies in hospitalized patients.
Use and efficacy of CSII in T1DM
Currently, pediatric patients with T1DM are frequently treated with CSII. A meta-analysis conducted by Kaiserman et al. [58] included 26 studies of more than 2,500 pediatric and adolescent patients with T1DM. Within the randomized controlled trials (RCTs) comparing insulin lispro CSII with MDI, glycosylated hemoglobin (HbA1c) levels achieved with insulin lispro were similar to or better than observations with the MDI treatment arm. In the RCTs, insulin lispro CSII and MDI therapy showed comparable incidences of diabetic ketoacidosis and severe hypoglycemia. The Agency for Healthcare Research and Quality systematic review of RCTs concluded that both insulin delivery modalities (CSII and MDI) display similar effectiveness on glycemic control and severe hypoglycemia in pediatric patients with T1DM [59]. In children and adults with T1DM, CSII improves quality of life greater than MDI, albeit with low strength of evidence. In supplementary findings, it was shown that the use of sensor-augmented insulin pumps (real time-CSII+CGM) was superior to MDI/SMBG in lowering HbA1c in patients with T1DM [59]. Recently, Ruiz-de-Adana et al. [60] performed a randomized study including 45 patients with T1DM (mean HbA1c, 8.6%±1.8%). Significant improvements were demonstrated in the HbA1c (7.9%±0.7% vs. 7.0%±0.6%, P<0.001) and quality of life in the CSII group after 6 months. HbA1c levels of the CSII group were lower than the MDI/glargine group (CSII 7.0%±0.6% vs. MDI/G 7.6%±0.9%, P<0.03).
Adjunctive therapies to insulin therapy in T1DM
Although insulin technology and its administration have shown remarkable advances, individuals with T1DM have continued difficulties in maintaining target glucose levels and sometimes suffer AE, such as severe hypoglycemia and weight gain [61]. Therefore, the evaluation of adjunctive therapies in individuals with T1DM is required.
1. Metformin as T1DM treatment
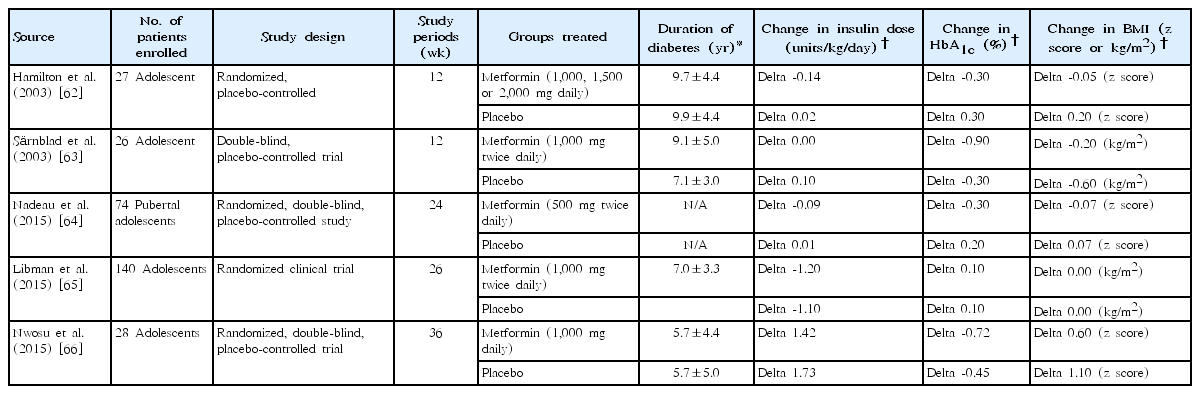
Metformin lowers hepatic glucose production, reduces glucose absorption in the intestines, and fosters glucose uptake and insulin sensitivity in peripheral tissues, resulting in a reduction of fasting and postprandial glucose [61]. Several clinical studies have been conducted for metformin therapy in T1DM pediatric patients (Table 3). Hamilton et al. [62] examined pediatric patients (n=27) with T1DM who required insulin doses >1 unit/kg/day. HbA1c was lower by up to 0.6% in the metformin group than in the placebo group. Fasting plasma glucose (FPG) was significantly reduced with metformin (-21.6±36 mg/dL vs. 1.8±45 mg/dL, P=0.004). Insulin doses were notably lower in the metformin group (-0.14±0.1 units/kg/day vs. 0.02±0.2 units/kg/day, P<0.01). It was observed that body mass index (BMI) was lower in the metformin group, but it was not significant. In another clinical trial, 30 adolescent patients were randomly given metformin or placebo for three months. HbA1c level significantly decreased (9.6% to 8.7%) in the metformin group, but it did not change in the placebo group. Individuals having lower insulin sensitivity showed greatest benefit from metformin therapy [63]. A randomized, double-blinded placebo-controlled study examined adolescent patients (n=74) with T1DM for 6 months [64]. They were randomized to either metformin 500 mg twice daily or placebo. Overweight or obese patients with metformin showed a trend toward lower HbA1c. Daily insulin dose (DID) in units and units/kg was significantly reduced by metformin. Further, metformin dramatically decreased BMI z scores. To assess metformin added to insulin therapy in overweight or obese patients with T1DM, 2 RCTs were conducted. In the first trial of enrolled patients (n=140, aged 12–20 years) [65], HbA1c dropped significantly around 3 months, but it was not maintained around 6 months. Mean total DID per kg was lower in the metformin group than the placebo group (-0.1 [-0.2 to -0.0], P<0.001). The metformin and placebo groups showed BMI reductions of at least 10% from baseline (24% vs. 7%, P=0.01). The second trial included patients (n=28, aged 10–20 years) with T1DM [66], and there was no significant difference in HbA1c and FPG levels between the metformin and placebo group for 9 months. The change in total DID was 0.31 units/kg between the metformin and placebo group.
2. DPP-4 inhibitors as T1DM treatment
Dipeptidyl peptidase-4 (DPP-4) inhibitors, including sitagliptin and vildagliptin, are known to increase GLP-1 and decrease glucagon levels after a meal without interrupting counterregulatory response during hypoglycemia [67-69]. In small study, it was observed that sitagliptin therapy for 4 weeks significantly decreased HbA1c levels by 0.2%–0.3% [70]; however, longer study demonstrated no significant reductions [71]. Changes in insulin requirements also range from no change [67,68] to minimal reductions of 0.05–0.13 units/kg/day; however, larger reductions were seen in postprandial insulin use [70,71]. According to a meta-analysis, DPP-4 inhibitors were not beneficial for patients with T1DM because of the limited glucose-lowering effect and the risk of severe hypoglycemia [72]. Considering the inadequate evidence for decreases in HbA1c levels and reduction of insulin dose, the application of DPP-4 inhibitors in the clinic setting is restricted at present.
3. SGLT2 (sodium-glucose cotransporter 2 inhibition) inhibitors as T1DM treatment
SGLT2 inhibitors may contribute to weight reduction in overweight/obese patients with T1DM and decreased glucose levels by blocking SGLT2 in the renal proximal tubule, interrupting glucose reabsorption [73,74]. However, evidence on the beneficial effect of SGLT2 inhibitors as adjunct to insulin treatment in T1DM remains insufficient. A randomized, double-blind study investigated patients (n=62) with T1DM concerning safety [75]. The results of efficacy revealed no significant difference between dapagliflozin and placebo in terms of mean glucose level, except reductions in FPG and TDID. Canagliflozin was examined in patients (n=351) with T1DM by a randomized, double-blind trial (phase 2 study) [76]. After 18 weeks, the HbA1c level was decreased by ≥0.4%; however, body weight change was not observed in patients. Further, an average rate of hypoglycemia was not different among groups.
Conclusions
Our current understanding about the cause and administration of T1DM is still far from complete, despite technological advances in T1DM. To improve TIDM predictions, new biomarkers are needed that can redefine risk groups and offer insights into the mechanism of disease onset. For the prevention of devastating complications and better management of T1DM, improvements in adjunct therapies are still needed. Future research efforts should be focused on improving disease prediction and the insulin delivery system, overcoming device limitations, and providing additional data to facilitate optimal clinical management.
Notes
No potential conflict of interest relevant to this article was reported.