Final height of Korean patients with early treated congenital hypothyroidism
Article information
Abstract
Purpose
Congenital hypothyroidism (CH) is the most common endocrine disorder in children. Thyroid hormone deprivation results not only in mental retardation but also growth retardation. This study investigates the final height (FH) in Korean patients with CH detected by newborn screening and examines factors that may affect the FH.
Methods
The medical records of Korean CH patients (n=45) were reviewed. The FH was examined and target height (TH) was calculated based on mid-parental height. The FH z score (FHZ) and TH z score (THZ) were computed using the 2007 Korean National Growth Chart. The FHZ and THZ were compared with a Student t test. The impact of the etiology of CH (athyreosis, dyshormonogenesis, ectopic thyoid, hypoplastic thyroid), initial serum thyroid stimulating hormone (TSH) level, initial free thyroxine (T4) level, and time of therapy initiation based on FH was assessed.
Results
The mean FHZ was 0.10±1.01 for male patients and -0.11±1.09 for female patients. There were no significant differences between FHZ and THZ for both female (P=0.356) and male patients (P=0.237). No significant relationship was found between FH and the etiology of CH, initial TSH level, initial free T4 level, and the time of therapy initiation.
Conclusion
Early intervention and satisfactory management do not appear to impede growth in Korean patients with CH. Thus, early detection and proper management of patients with CH detected by newborn screening program are necessary.
Introduction
Congenital hypothyroidism (CH) is the most common endocrine disorder in children. Thyroid hormone is important for growth and intellectual development of children and thyroid hormone deprivation results in deleterious effects on bone growth [1]. Infants with CH who are not treated with levothyroxine (L-T4) have disordered and delayed epiphyseal development and grow poorly [2,3].
Newborn screening for CH using dried blood spot sampling that started in the 1970s in most of the developed Countries and in the early 1990s in Korea enabled early diagnosis and treatment of CH [4,5]. Many studies have shown that early treatment of CH leads to normalization of cognitive development [6-8]. The linear growth of patient with CH has been examined in several studies [1,9-13], but there are few studies about the final growth of patient with CH [14-17]. Furthermore there is no report regarding the growth of Korean patients with CH.
The factors that affect the final height (FH) of CH patient are also controversial. Salerno et al. [14] reported that the FH of Italian patients with CH were normal, and the FH was not correlated with the severity of CH at diagnosis, L-T4 dosage at the beginning of treatment, or etiology of the defect. Similarly, Adachi et al. [15] also documented that the FH of Japanese patient with CH was normal and there was no relationships between the FH and the etiology of CH, severity of CH (Thyrotropin [TSH] level at the start of treatment and current L-T4 dose), time of therapy initiation and adequacy of treatment during the first year of life. However, Bain and Toublanc [16] reported the main factors for a taller FH than TH were the age at the start of L-T4 treatment and the quality of treatment. They reported that the patients with taller FH than TH had a significantly younger mean age at the start of treatment than the patients with smaller FH than TH (24.8±13.5 vs. 42.8±47.3, P=0.004). Heidari et al. [18] also emphasized the importance of the first therapeutic L-T4 dosage, age at onset of treatment and age at the first free T4 normalization for anthropometric development based on a longitudinal study of 760 neonates born in 2002–2009, with CH diagnosed by neonatal screening in Iran.
Thus, this study investigated the FH in Korean patients with CH detected by newborn screening, and examined the factors that may affect the height outcome.
Materials and methods
1. Patients and methods
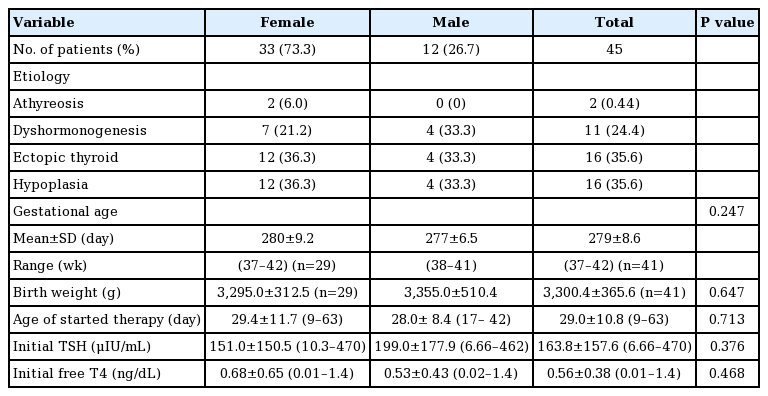
This study reviewed the medical records of Korean CH patients who were followed up at Soonchunhyang University Seoul Hospital (Korea) from January 1994 to December 2015. Total 219 patients (151 females, 68 males) had reached their FH by the time of the study. Of these patients, 174 patients (118 females, 56 males) were excluded due to lack of clinical information at the beginning of the L-T4 treatment. Therefore, 45 patients (33 females, 12 males) were enrolled in this study. The patients were considered to have reached their FH when growth rate was less than 1 cm/yr.
We checked the initial serum TSH, free T4 levels at diagnosis, the age of starting L-T4 treatment and the etiology of CH on the medical records. The etiology of CH was determined by a 99mTc-pertechnetate thyroid scan. All patients, except 4 women with unidentifable gestational age and birth weight, were born full term after an uncomplicated pregnancy. The patients kept on continuous L-T4 replacement therapy and visited hospital every 3 to 6 months to check serum TSH and free T4. The mean age of starting L-T4 treatment was 29.0±10.8 days. The etiologies of the patients were 2 athyreosis (2 females), 11 dyshormonogenesis (7 females, 4 males), 16 ectopic thyroid (12 females, 4 males), and 16 hypoplastic thyroid (12 females, 4 males). All the patients with CH due to dyshormonogenesis tried to discontinue L-T4 but kept on continuous L-T4 replacement therapy because their serum TSH level was rised above 20 μIU/mL. The clinical profiles of patients with CH are summarized in Table 1.
For each patient, the FH was examined and the target height (TH) was calculated by midparental height [19]. The 2007 Korean National Growth Chart was used to derive the FH z scores (FHZ) and the TH z scores (THZ) [20]. The impact of the etiology of CH, initial TSH, initial free T4 and time of therapy initiation on the FH was assessed.
The Institutional Review Board (IRB) of Soonchunhyang University Hospital approved this study (approval number: SCHUH 2017-08-008-001). Obtaining of written informed consent was exempted from IRB.
2. Statistical analysis
Data were expressed as the mean±standard deviation and the statistical analysis was performed with SPSS ver. 17.5 (SPSS Inc., Chicago, IL, USA). The FHZ and THZ were compared using Student t test. We divided the patient to 4 groups by the etiology of CH (athyreosis, dyshormonogenesis, ectopic thyroid, hypoplastic thyroid) to evaluate the impact of etiology of CH on the FH. One-way analysis of variance was used to compare the 4 groups based on etiology. The regression analysis with linear model was used to evaluate the relationship between FH and initial serum TSH and free T4 level. Finally, we divided the patients to 2 groups: group A, the patients who started the treatment for CH within 30 days of birth (n=25); group B, the patients who started the treatment after 30 days of birth (n=22) and compared the groups A and B by single Student t test to evaluate the influence of L-T4 treatment initiation time on the FH. All the differences with P<0.05 were considered statistically significant.
Results
1. The FH in patients with CH
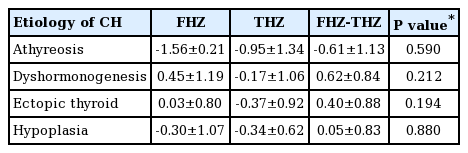
The FH of female patients was 159.8±5.5 cm and the FHZ was -0.11±1.09. The FH of male patients was 174.0±5.7 cm and the FHZ was 0.10±1.01. The calculated differences between the FHZ and the THZ (FHZ-THZ) were 0.23±0.88 in female patients and 0.44±0.91 in male patients. There were no significant differences between FHZ and THZ for both female patients (P=0.356) and male patients (P=0.237). There were no differences between FHZ and THZ for each group divided by etiology of CH and no significant relationship was found between the etiology of CH and the FHZ-THZ (Table 2).
2. The factors affecting to FH
The correlation between the severity of hypothyroidism and FH was examined. We compared the FHZ and FHZ-THZ with the initial serum TSH and initial free T4 with regression analysis. As shown in Figs. 1 and 2, there was no correlation between both FHZ and FHZ-THZ with initial serum TSH and initial free T4.
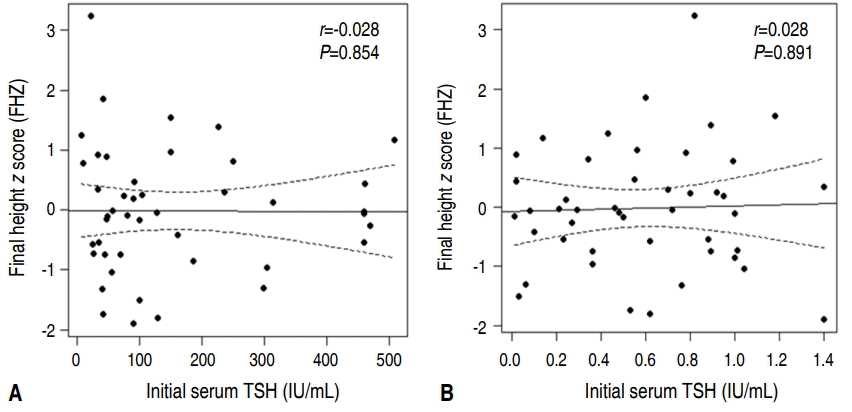
Regression of the final height z score (FHZ) based on the initial serum thyroid stimulating hormone (TSH) level (A) and initial serum free thyroxine (T4) level (B) at initiation of levothyroixine (L-T4) treatment. There were no correlations between FHZ and initial serum TSH or between FHZ and free T4 level.
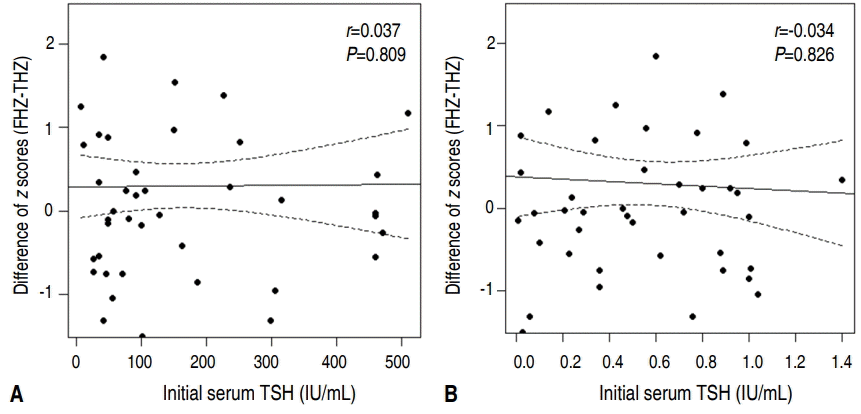
Regression of the difference in z scores (FHZ-THZ) based on the initial serum thyroid stimulating hormone (TSH) level (A) and initial serum free thyroxine (T4) level (B) at initiation of treatment. There were no correlations between FHZ-THZ and initial serum TSH or between FHZ-THZ and free T4 level.
Finally, we compared the FHZ-THZ of the group A and the FHZ-THZ of the group B using Student t test. There was no difference between the FHZ of the group A and that of the group B (P=0.6471) (Fig. 3).
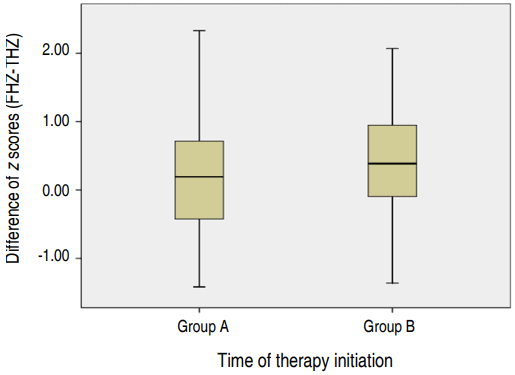
Difference in z scores (FHZ-THZ) according to the time of therapy initiation. Group A patients started treatment for CH within 30 days of birth. Group B patients started treatment more than 30 days after birth. There was no difference between the FHZ of group A and that of group B (*P=0.6471). FHZ, final height z score; THZ, target height z score; CH, congenital hypothyroidism.
Discussion
The newborn screening programs for CH started in the 1970s enabled early detection and treatment of CH. Newborn screening programs for CH have been highly successful in improving both cognitive and growth outcome. Several studies reported that early diagnosis and treatment of CH prevents poor growth due to hypothyroidism [9-10,13]. Grant [10] reported that height, weight and body mass index of 361 patients with CH aged 3–4 years old were normal. Moschini et al. [13] observed 42 patients who had been diagnosed with CH by newborn screening program and reported the height, weight and head circumference of the patients were in 50th–75th percentile at 6 years of age, if the treatment was initiated within 33 days after birth.
Salerno et al. [14] studied 55 Italian patients with CH. The patients were diagnosed with CH at 25±5 days of age by the newborn screening program. The height of the patients at 17±5 years old was normal (standard deviation score, 0.1±1.1) and the FH was not affected by the birth date, the age at diagnosis and the starting LT4 dose. Adachi et al. [15] reported the FH of 27 Japanese patients with CH who started L-T4 treatment at the age of 23.9±10.8 days and the FH in CH patients detected by neonatal screening. They reported that the FH of patients with CH were equivalent to TH and as long as early intervention and satisfactory management are ensured, severe CH does not appear to worsen the outcome. Likewise, in support of these previous studies, the current study of Korean patients with CH had no difference between their FH and TH, and also had no difference between the FH and height of Korean reference population.
However, Delvecchio et al. [17] reported that the FH of 215 Italian patients with CH were taller than their TH which was attributed to an improvement in the FH of Italian people over the last 20 years. Bain and Toublanc [16] also reported that patients with CH reached a taller FH than their TH. They concluded that the initial severity of the CH was not a prognostic factor for the FH, whereas the age at start of treatment was significant. They reported that the patients who exceeded their TH had an earlier start of treatment versus children who failed to reach their TH (24.8±13.5 days vs. 42.0±47.3 days, P=0.004). The discrepancy between these previous studies and our findings highlight the need for further research to conclusively identify the prognostic factors for the FH of patients with CH.
In 2012, Seth et al. [21] reported about characteristics of delayed diagnosed patients with CH. They studied 94 Indian patients with hypothyroidism who diagnosed beyond 5 years of age and 34 of the patients (36.1%) were CH. In their study, all the patients with CH who diagnosed beyond 5 years of age had growth failure and they emphasized the importance of early detection and treatment of patients with CH by newborn screening program.
In this study, we could not find information about the puberty onset and the other hormone serum levels that can influence the FH. Further research including these points is needed to evaluate factors affecting the growth of patients with CH.
In conclusion, as long as early intervention and satisfactory management are performed, the CH does not appear to worsen the growth of Korean patients with CH. And the etiology of CH, initial TSH, initial free T4 level and the time of therapy initiation do not affect the FH in CH patients. Thus, it is important to detect early and provide proper management to patients with CH by newborn screening program.
Notes
No potential conflict of interest relevant to this article was reported.