Eosinophilic gastroenteritis in an 18-year-old male with prolonged nephrotic syndrome
Article information
Abstract
Eosinophilic gastroenteritis is a rare disease characterized by prominent eosinophilic tissue infiltration of the gastrointestinal tract. Here, we report a case of eosinophilic gastroenteritis in an 18-year-old patient with prolonged nephrotic syndrome who presented with abdominal pain and peripheral hypereosinophilia. During the previous 2 years, he had visited local Emergency Department several times because of epigastric pain and nausea. He had been treated with steroid-dependent nephrotic syndrome since 3 years of age. Tests ruled out allergic and parasitic disease etiologies. Gastroduodenoscopy with biopsy revealed marked eosinophilic infiltration in the duodenum. Renal biopsy findings indicated minimal change disease spectrum without eosinophilic infiltration. The oral deflazacort dosage was increased, and the patient was discharged after abdominal pain resolved. To our knowledge, this is the first report of eosinophilic gastroenteritis in a patient with minimal change disease.
Introduction
Nephrotic syndrome (NS) in children is typically idiopathic, but may also occur secondary to other diseases including eosinophilic disorders1234). Eosinophilic gastroenteritis (EG) is an eosinophilic disease characterized by eosinophilic inflammation of the gastrointestinal (GI) tract56). There have been few previous reports of these 2 distinct diseases coexisting in the same patient7). Here we describe a rare case of EG presenting as abdominal pain with peripheral eosinophilia in prolonged NS.
Case report
An 18-year-old male was admitted with epigastric pain and nausea. During the previous 2 years he visited a local Emergency Department several times with a similar clinical presentation. He had been treated for steroid-dependent NS since he was three years of age, and was taking 6 mg (0.1 mg/kg) of oral deflazacort every other day when admitted. There was no history of allergic disease such as food allergies, asthma or atopic dermatitis.
On admission, the results of laboratory tests included leukocyte count, 18,0×103/µL with 28.3% eosinophils (absolute eosinophil count, 5,108/µL); hemoglobin, 15.5 mg/dL; platelets, 582×103/µL; blood urea nitrogen, 9.9 mg/dL; creatinine, 0.42 mg/dL; total protein, 4.2 g/dL; albumin, 2.4 g/dL; total cholesterol, 362 mg/dL; aspartate aminotransferase (AST), 18 IU/L; alanine aminotransferase (ALT), 16 IU/L; amylase, 234 IU/L; and urine protein, ±; and urinalysis showed no other abnormal findings. Abdomen and pelvis computed tomogarphic scans were performed due to elevated amylase, and revealed wall thickening from the distal esophagus to the proximal jejunum and distal jejunal loops without evidence of pancreatitis (Fig. 1). When he was referred to our hospital after discharge from the local hospital, the patient's physical examination demonstrated no specific findings. Additional laboratory values were as follows: leukocyte count, 14,8×103/µL with 20.7% eosinophils of (absolute eosinophil count, 3,063/µL); hemoglobin, 14.6 mg/dL; platelets, 501×103/µL; peripheral blood smear, mild leukocytosis with eosinophilia; amylase, 68 IU/L; total IgE, 85.0 IU/mL; anti-dsDNA antibody, (–); stool exam, (–); anti-amoeba antibody (Ab) IgG, (–); Toxocariasis Ab, (–); urine protein, 4+; and spot urine protein/creatinine, 3.1.
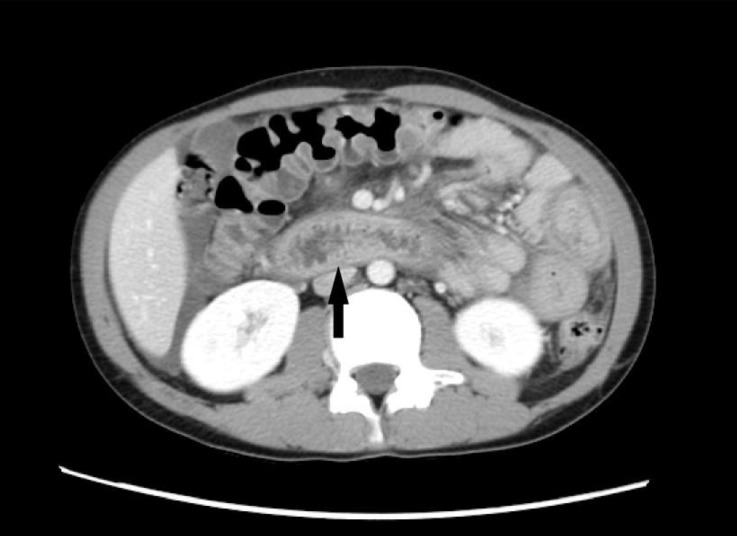
A computed tomographic scan of the abdomen showing the layered wall thickening and mucosal fold thickening of the duodenum (arrow).
The patient had grossly edematous findings without erosion or ulcer in duodenum on esophagogastroduodenoscopy. Biopsy specimens of the duodenal mucosa showed eosinophilic infiltration in the lamina propria up to 35/high-power field (Fig. 2). Renal biopsy was performed to evaluate the relationship between hypereosinophilic syndrome (HES) and renal disease. The biopsy findings were (1) within the minimal change disease (MCD) spectrum with IgM deposits (IgM nephropathy), and (2) mild tubular atrophy with interstitial fibrosis. There was no infiltration of inflammatory cells, even eosinophils. Chest radiography and cardiac sonography were unremarkable. A diagnosis of EG and relapsed NS was made. On the 6th hospital day, the patient complained of epigastric pain again and the quantity of oral deflazacort was increased to 54 mg (1.2 mg/kg) per day, after which his symptoms improved. On the 11th hospital day, the patient was discharged with decreased proteinuria (spot urine protein/creatinine, 2.0) and decreased eosinophilia (absolute eosinophil count, 72/µL). On follow-up, mild to moderate peripheral eosinophilia was a continuing issue, but the patient did not complained of further abdominal pain for 2 years. But recently he was admitted again for repetitive severe epigastric pain. There was severe peripheral eosinophilia (absolute eosinophil count, 5,054/µL), without proteinuria. The results of both abdomen computed tomography and duodenal biopsies were similar with previous results. Biopsy specimens of the duodenal mucosa showed eosinophilic infiltration up to 30/high-power field. His symptom was improved after treating with 50 mg of intravenous methylprednisolone IV for 3 days and he was discharged with decreased eosinophilia, on the 8th hospital day (absolute eosinophil count, 318/µL).
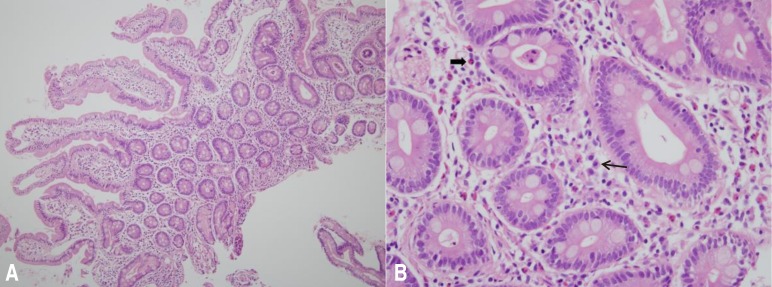
(A) No epithelial dysplasia or structural abnormalities are visible (H&E, ×100). (B) Some inflammatory cells, including lymphocytes, plasma cells (thin arrow), and eosinophils (broad arrow), are visible in the lamina propria. The number of eosinophils increased up to 35/high-power field without intraepithelial infiltration (H&E, ×400).
Discussion
The patient reported here had peripheral eosinophilia with abdominal pain as a form of EG and MCD presenting as NS. Serum eosinophilia and abdominal pain improved after administration of increased oral deflazacort. Heavy proteinuria also decreased in response to the same treatment. On second admission, the patient only developed EG and was treated with methylprednisolone.
Although the etiology of EG is unknown, a history of allergies is reported in one-third to one-half of all patients with EG, suggesting an allergic basis6). One interesting case report describes the occurrence of NS in a patient with EG and a food allergy. In this patient, a direct causal association between atopic EG and NS was suggested by renal biopsy showing immune complex glomerulonephritis with bovine serum albumin7). Our patient had no history of allergy and normal serum IgE. To our knowledge, this is the first case report of child or adolescent with nonatopic EG and NS.
EG may also occur secondary to systemic diseases such as connective tissue disease or HES5). HES is characterized by marked (>1,500×106/L) and persistent (>6 months) eosinophilia and multiorgan involvement without a clear etiology. The most frequently involved organs are the cardiovascular, pulmonary, hematopoietic and nervous systems4). HES also involves the GI tract in 25% of cases. It may be difficult to differentiate between HES with intestinal involvement and EG5).
EG is defined based on the following 3 criteria: the presence of GI symptoms, biopsy indicating eosinophilic infiltration of one or more areas of the GI tract from the esophagus to the colon, or characteristic radiological findings with peripheral eosinophilia, and no evidence of parasitic or extraintestinal disease8). Mucosal eosinophilic gastroenteritis, the most common type, manifests as abdominal pain, diarrhea, protein-losing enteropathy and/orvomiting568), while the peripheral eosinophilic count appears normal in 23% to 40% of EG patients6). In our case, diagnosis was based on patient history (recurrent abdominal pain with nausea), infiltration of eosinophils on duodenal biopsy and peripheral eosinophilia. The patient had GI symptoms, but no extraintestinal signs such as skin nodules, lymphadenopathy or hepatosplenomegaly on physical examination, and no evidence of other organ involvement. Primary EG is a more appropriate diagnosis for our patient than GI involvement with HES. However, patients with EG may, in rare cases, progress to HES, so long-term follow-up and careful exam for new signs of HES are necessary5).
Renal involvement is rarely observed in HES4910). The mechanisms implicated in renal involvement are eosinophil cytotoxicity, mass effect due to eosinophilic infiltration and thromboembolic events secondary to cardiac damage10). In our case, NS anteceded the emergence of eosinophilia by several years. Thus, renal disease may be less likely to develop secondary to HES in our patient.
The first line of treatment for NS is corticosteroids1). Corticosteroids improve proteinuria by regulating abnormal immune reactions11). Corticosteroids are also the most effective anti-inflammatory drugs used in the treatment of EG568). Their beneficial effects result from (1) the suppression of the synthesis and effects of eosinophil survival factors, (2) the direct induction of eosinophil apoptosis and (3) the stimulation of engulfment by professional phagocytic cells12). Despite their different pathological mechanisms, the treatment of both EG and NS is the same, and thus there is a possibility that diagnosis of EG was delayed in our case.
In summary, we report the first case of nonatopic EG with NS. However, the association of these concomitant presentations is not clear. As many as 45% of children with NS will have a frequently relapsing course, and some will be steroid-dependent13). This case emphasizes that, in children, treatment with prolonged steroid therapy has the potential to mask other diseases, even those that may be fatal. Thus, pediatricians should be cautious when examining child or adolescent treated with steroids for long periods.
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.