Clinical presentations of critical cardiac defects in the newborn: Decision making and initial management
Article information
Abstract
The risk of mortality and morbidity of patients with congenital heart defects (CHDs) is highest during neonatal period and increases when diagnosis and proper management are delayed. Neonates with critical CHDs may present with severe cyanosis, respiratory distress, shock, or collapse, all of which are also frequent clinical presentations of various respiratory problems or sepsis in the newborn. Early diagnosis and stabilization and timely referral to a tertiary cardiac center are crucial to improve the outcomes in neonates with CHDs. In this review, the clinical presentation of critical and potentially life-threatening CHDs is discussed along with brief case reviews to help understand the hemodynamics of these defects and ensure proper decision-making in critically ill patients.
Introduction
Congenital heart defects (CHDs) are the most common birth defects with an incidence of approximately 6-8 in 1,000 live births, accounting for 6-10% of all infant deaths and 20-40% of all infant deaths from malformations1-4). While most CHDs are mild forms of defects, 15% are potentially life-threatening and one third of these are cyanotic heart disease1). Because neonates with potentially fatal cardiovascular malformations are frequently asymptomatic just after birth, they may be discharged home early without being diagnosed with CHDs and some of them deteriorate or die before the diagnosis is made4). In a recent report1), 25% of neonates with serious heart defects were discharged without being diagnosed after their birth hospitalizations. Therefore, general pediatricians should realize that early recognition and timely interventions to stabilize such patients and transfer of these patients to an appropriate cardiac center are critically important for improving the outcomes in these critically ill patients. In this review, clinical presentations of potentially life-threatening CHDs in neonates were discussed along with brief case reviews to help pediatricians understand the hemodynamics of these defects and to facilitate correct decision-making in these patients.
Critical congenital heart defects in the newborn
1. Transition from fetal circulation to postnatal circulation
The important hemodynamic change after birth is the transition from fetal circulation (oxygenation occurs in the placenta) to postnatal circulation (oxygenation occurs in the lung). In fetal circulation, oxygenated blood from the placenta returns to the right atrium (RA) via the umbilical vein and the ductus venosus; this blood passes through the foramen ovale and is directed to the left ventricle (LV), which ejects this highly oxygenated blood to the highly metabolic heart and brain. Less oxygenated blood from the superior vena cava (SVC), the portal veins, and the coronary sinus and that from the lower body via the inferior vena cava (IVC) are mainly directed to the right ventricle (RV), which ejects it to the descending aorta via the main pulmonary artery and the ductus arteriosus, and eventually to the placenta for oxygenation. Therefore, in this system, the presence of central shunts (ductus arteriosus, ductus venosus and foramen ovale) is very important for effective fetal circulation. After birth, oxygen uptake occurs in the lung bed, which is independently supplied by the RV, and the LV separately supplies the systemic circulation with oxygenated blood. In this serial circulation, these shunts are not necessary and actually will close within the first hours or days of life.
2. Ductal-dependent lesions
Most patients with critical CHDs have a severely hypoplastic or small RV or LV and/or severe obstructive lesions in the valves or great arteries in the right or left heart system. Therefore, patients with these lesions will have a severely compromised systemic or pulmonary circulation, cannot tolerate the transition from fetal circulation to postnatal serial circulation after birth, and will depend on the central shunts, especially patent ductus arteriosus (PDA) for adequate circulation in each system. These lesions which are dependent on blood flow through the PDA for adequate circulation are collectively referred as "ductal-dependent lesions". Patients with ductal dependent lesions will present with severe cyanosis, shock or collapse as the PDA constricts within hours or days after birth.
Lesions with ductal-dependent pulmonary circulation, in which the pulmonary blood flow for oxygenation is supplied by the systemic circulation (the aorta) via the PDA (flow from the aorta to the pulmonary artery) usually present with severe cyanosis. These are usually right-sided obstructive lesions or right-to-left shunt lesions such as severe form of tetralogy of Fallot, pulmonary atresia (PA) with ventricular septal defect (VSD), critical pulmonary valve stenosis (PS) or pulmonary atresia with intact ventricular septum, Ebstein's anomaly, or any complex CHD with severe PS or PA.
Lesions with ductal-dependent systemic circulation, in which the systemic output is supplied by the pulmonary artery system via the PDA (flow from the main pulmonary artery to the aorta) may present with systemic hypotension, shock, or collapse as the PDA constricts or closes after birth. This category of CHDs include various kinds of left-sided obstructive lesions such as the hypoplastic left heart syndrome (HLHS), severe aortic valve stenosis, coarctation of the aorta, interruption of the aortic arch, or any complex lesions with systemic outflow obstruction.
3. Ductal-independent mixing lesions
Some cases of CHDs may present with cyanosis and congestive heart failure or pulmonary edema, that is, increased pulmonary blood flow. The most common anomaly associated with such cases is transposition of the great arteries (TGA), in which the ventriculoarterial connections are discordant (two great arteries are "misplaced" or "transposed" across the interventricular septum); thus, the aorta arises from the morphologic right ventricle and the pulmonary artery arises from the morphologic left ventricle. In this unique situation, the systemic and pulmonary circulations are parallel; therefore, the oxygenated pulmonary venous blood re-enteres the pulmonary circulation via the morphologic left ventricle and the deoxygenated systemic venous blood is directed to the aorta via the morphologic right ventricle. Mixing of blood between these two circulations, which occurs via PDA, ASD or PFO, or VSD, is essential for survival in neonates with TGA.
Truncus arteriosus is a rare congenital heart defect in which one great vessel (truncus) arises from both ventricles and then gives rise to the aorta and the pulmonary arteries. This defect is always associated with a large VSD. The systemic and pulmonary venous blood is mixed in the ventricles and the truncal vessel, thereby leading to cyanosis, and redistributed to the systemic and pulmonary circulation. Thus, because the vascular resistance of pulmonary circulation is much lower than that of systemic circulation, the blood flow to the pulmonary circulation is usually much higher than that to the systemic circulation, thereby leading to severe congestive heart failure.
Total anomalous pulmonary venous connection (return) (TAPVC or TAPVR) is a cyanotic heart defect in which all pulmonary veins fail to connect to the left atrium. All the pulmonary venous blood returns to the right atrium via various connections between the pulmonary veins and the right heart system (innominate vein, superior vena cava, coronary sinus, portal system, or inferior vena cava), thereby resulting in the mixing of blood in the right atrium (presents with cyanosis) and pulmonary overcirculation (presents with pulmonary edema). The systemic circulation is maintained by a right-to-left shunt flow through the PFO or ASD. If there is a significant obstruction in the pulmonary venous drainage (most commonly infracardiac-type of TAPVR), pulmonary edema and severe cyanosis will develop after birth.
Major clinical presentations in neonates with critical CHDs
1. Cyanosis
Cyanosis is a physical sign characterized by bluish discoloration of the skin or mucous membranes due to the presence of excessive amount (not a ratio) of reduced hemoglobin in the capillary bed. Clinically, cyanosis can be noticed when the concentration of reduced hemoglobin is more than 3-5 g/dL.
Various neonatal clinical conditions may present with cyanosis. Some healthy neonates look blue (nonpathologic cyanosis) due to facial plethora, neonatal polycythemia, circum oral venous congestion, or just a dark lip. However, because neonates with pathologic cyanosis are usually critically ill and most of the cyanotic CHDs are potentially life-threatening, early assessment and decision-making, and timely interventions are very important to improve their outcomes.
There are two mechanisms (or types) of pathologic cyanosis. One mechanism involves excessive oxygen uptake (or consumption) by the tissues (peripheral cyanosis). In patients with peripheral cyanosis, the extremities are cyanotic, pale and cool, but the tongue and conjunctiva are pinkish, that is, systemic arterial oxygen saturation is usually normal. Sepsis, cold exposure, shock or low cardiac output state, hypoplycemia, or metabolic disorders are differential diagnoses of peripheral cyanosis. The other mechanism of cyanosis involves decreased oxygen saturation (increased reduced hemoglobin) in the systemic arterial blood (central cyanosis); this form of cyanosis is usually found in cardiovascular, pulmonary, central nervous system or neuromuscular abnormalities, or methemoglobinemia. Patients with central cyanosis show cyanosis on the extremity as well as on the lip, tongue, and conjunctiva. Since peripheral circulation is usually normal, the patient's extremities are warm and capillary filling is rapid. Arterial blood gas analysis will show decreased oxygen tension and saturation in patients with central cyanosis.
The most common noncardiac causes of cyanosis in the newborn are pulmonary disorders such as primary lung disease, airway obstruction and extrinsic compression of the lungs5). Therefore, differentiating the cardiovascular and pulmonary causes of cyanosis in the neonates is an important issue in clinical practice. In the majority of patient, high index of suspicion and a thorough history taking and physical examination with blood pressure measurements at the four extremities, measurements of oxygen saturation with a hyperoxia test, EKG, and chest radiography may differentiate cyanosis of different origins.
Because some cases of neonatal undiagnosed cyanotic CHD may show rapid deterioration or result in the death of the patient, immediate medical interventions are mandatory in critically ill neonates with suspected congenital heart disease even before an accurate cardiac diagnosis is made.
Some useful clues can be employed to differentiate cyanosis of cardiac and pulmonary origins (Table 1). In patients with cardiac cause of cyanosis, respiration is relatively comfortable despite cyanosis, which may worsen on crying. A heart murmur and abnormal cardiac silhouette indicate cardiac defects. While cyanotic CHDs with ductal-dependant pulmonary circulation (right-sided obstructive lesions) usually show normal or decreased pulmonary vascular markings (darker lung field) in chest radiographs, ductal-independent mixing lesions such as TGA, truncus arteriosus, and total anomalous pulmonary venous return (TAPVR) may be frequently associated with pulmonary vascular congestion or edema. An abnormal QRS axis or rhythm disturbances suggest heart defects. Arterial blood CO2 tension is normal or low. Response to 100% O2 supply is blunted because of right-to-left shunts in the cardiac level. A neonate with oxygen saturation less than 85% (O2 tension less than 50 mmHg) in both room air and 100% oxygen is very likely to have a cyanotic CHD with an intracardiac right-to-left shunt, and immediate presumptive therapy with prostaglandin infusion should be initiated in such patients5).
Patients with pulmonary causes of cyanosis usually show apparent tachypnea, distress, and retraction. Cyanosis may improve on crying because of increased ventilation. Rale, crackle, or wheezing may be heard in such patients. In chest radiography, the cardiac shape and size are usually normal, but the lung fields may show abnormal findings such as a ground-glass appearance, pneumonic infiltration, atelectasis, or pneumothorax. In these patients, arterial blood CO2 tension is usually high because of airway problems and alveolar hypoventilation. These patients show a profound response to 100% O2 supply and hyperventilation, although this response is transient in some severe cases. In cyanotic patients with lung problems, arterial blood O2 saturation may easily increase up to 100% (arterial blood O2 tension >150 mmHg) in 100% oxygen supply ("hyperoxia" test), but this value hardly reach 100% in patients with cyanotic CHDs5).
Certain CHDs may present with a "differential cyanosis", in which the preductal part of the body (upper part of the body) is pinkish but the postductal part of the body (lower part of the body) is cyanotic, or vice versa ("reverse differential cyanosis"). The prerequisite for this unique situation is the presence of a right-to-left shunt through the PDA and severe coarctation of the aorta or aortic arch interruption or severe pulmonary hypertension. In patients with severe coarctation of the aorta or interruption of the aortic arch with normally related great arteries, the preductal part of the body is supplied by highly oxygenated pulmonary venous blood via the LA and LV, whereas the postductal part is supplied by deoxygenated systemic venous blood via the RA, RV, main pulmonary artery (MPA) and the PDA. In the newborn with structurally normal heart, a differential cyanosis may be associated with persistent pulmonary hypertension of the newborn. In the cases of TGA with coarctation of the aorta or aortic arch interruption, the upper body is mostly supplied by systemic venous blood via the RA, RV, and ascending aorta, whereas the lower body is supplied by highly oxygenated pulmonary venous blood via the LA, LV, MPA, and then the PDA. For accurate detection of differential cyanosis, oxygen saturation should be measured in both preductal (right finger) and postductal (feet) parts of the body.
2. Respiratory distress and/or pulmonary edema
Severe respiratory distress or pulmonary edema develops in cases of CHDs with excessive and nonrestrictive pulmonary blood flow or lesions associated with pulmonary venous obstruction, which result in tachypnea, chest wall retraction and increased work of breathing.
The common lesions responsible for these symptoms include most left-sided obstructive lesions (present with systemic hypotension or collapse), TGA with restrictive interatrial communication (presents with severe cyanosis), an obstructive type of TAPVR (presents with severe cyanosis) and truncus arteriosus (present with mild cyanosis). Simple left-to-right shunt lesions including VSD, ASD, AVSD, and PDA rarely present with severe pulmonary edema or distress during the neonatal period because of the relatively high pulmonary vascular resistance, which may restrict pulmonary blood flow. Preterm PDA may present with pulmonary edema and respiratory distress.
3. Systemic hypotension, shock, or collapse
In the newborn, various clinical conditions may present with systemic hypotension, shock, or collapse. The most common cardiac causes of these symptoms are the left-sided obstructive lesions, such as HLHS, critical AS, severe coarctation, and interruption of the aortic arch. A typical example is HLHS, in which the left ventricle cannot support the systemic circulation. The RV supports the pulmonary and systemic circulation that is maintained by the flow from the MPA to the aorta through the PDA. The upper part of the body and coronary arteries are supplied by the retrograde flow to the aortic arch and ascending aorta in this situation. Constriction and closure of the PDA result in sudden systemic hypotension and shock. In such situations, the only way to rescue the patient is to open the PDA immediately and perform other supportive managements including fluid therapy, respiratory care (usually involving mechanical ventilation with sedation, paralysis, and controlled ventilation), and inotropic support. The other causes of cardiogenic shock in the newborn are neonatal dilated cardiomyopathy or myocarditis and myocardial dysfunction due to tachyarrhythmias such as atrial flutter or paroxysmal supraventricular tachycardia.
Other significant conditions that may be suspected in the differential diagnosis of neonatal shock are neonatal sepsis or meningitis, hypoglycemia, and inborn errors of metabolism. In addition to careful history-taking and physical examination, chest radiography and electrocardiography are very useful to differentiate CHDs from other causes in emergent situations. One study in the neonates presenting with either bacterial sepsis or meningitis, or left-sided obstructive lesions showed that the presence of cardiomegaly predicted heart defects with 85% sensitivity and 95% specificity with a positive predictive value of 95% for CHDs6). If a neonate presenting with shock is suspected to have a CHD, the initial management should involve the basics of advanced life support along with immediate prostaglandin infusion to maintain the PDA even before an accurate diagnosis is made by a cardiologist5).
Review of some typical cases and discussion
1. Lesions presenting with cyanosis as the main clinical presentation
Case 1. A 3-day-old neonate with cyanosis
The baby was born by normal spontaneous vaginal delivery without perinatal problem at a local obstetric clinic. During a routine neonatal check-up, heart murmur and mild lip cyanosis were detected. The baby appeared healthy and fed well. Its breathing sound was clear. No definite tachypnea or chest retraction was noted. A grade III/VI harsh systolic ejection murmur was heard at the upper left sternal border. Pulse oximetry checked at the right finger and toe showed the same oxygen saturation of around 85%, and after oxygen supply via nasal prong (5 L/min), it registered around 90% at the both sites. The blood pressures in the right arm and right ankle were within the normal limits for the patient's age. EKG showed a QRS axis of 150° and satisfied the voltage criteria for right ventricular hypertrophy. Chest X-ray showed no cardiomegaly or significant lung lesion (Fig. 1A).
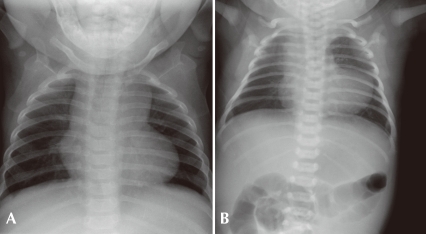
A) This picture shows a typical "boot-shape" cardiac silhouette without cardiomegaly, clear lung fields, and decreased pulmonary vascular markings. B) Chest radiograph of a patient with pulmonary atresia with ventricular septal defect showing elevated cardiac apex without cardiomegaly, narrowed mediastinal shadow (hypoplastic thymus and pulmonary trunk), and normal or slightly decreased pulmonary vascular markings. This patient also had a chromosome 22q11.2 microdeletion syndrome.
This is a typical clinical presentation of a common cyanotic congenital heart disease. The presence of cyanosis and cardiac murmur after birth in this case indicated that the cyanosis had a cardiac origin. The additional findings that indicated CHDs were a blunted response to oxygen supply, abnormal QRS axis and voltage, clear or rather darker lung field, and a typical cardiac silhouette (boot-shape) on chest X-ray (Fig. 1). Echocardiography revealed a typical form of tetralogy of Fallot (TOF), a large malaligned perimembranous VSD, PS (valvular and subvalvar infundibular PS), overriding of the aorta and RV hypertrophy. In TOF, the degree of cyanosis depends on the severity of PS, that is, the pulmonary blood flow reduces with an increase in the severity of PS, thereby causing more severe cyanosis. Normal heart size, the darker lung fields, and slightly indistinct hilar vascular markings on the chest X-ray indicated decreased pulmonary blood flow. If the PS is very severe or the pulmonary artery or valve is atretic (pulmonary atresia (PA) with VSD; an extreme form of TOF, Fig. 1B), patients will present with profound cyanosis when the PDA constricts. Management to maintain the PDA along with adequate hydration should be initiated immediately to maintain pulmonary blood flow. Fluid restriction and dehydration generally aggravate the cyanosis in cyanotic heart diseases, except in the cases of PDA-independent mixing lesions with pulmonary congestion (TGA, truncus arteriosus or TAPVR). The surgical treatment options for a severe form of TOF or PA with VSD are initial palliative procedures such as modified Blalock-Taussig shunt, insertion of a stent into the PDA, or insertion of a stent into the RV outflow track, which is followed by a total repair. In some centers, a total repair during the neonatal period is preferred.
Case 2. A neonate with cyanosis and tachypnea
A neonate was referred from a local obstetric clinic because of cyanosis and tachypnea a few hours after birth. The baby was born after a normal full term vaginal delivery and was meconium-stained at birth. On the physical examination, the baby appeared slightly pale, and showed blue lips and nasal flaring. The respiratory rate was 70/min with chest wall retraction. Chest auscultation revealed a coarse breathing sound with fine crackles in both lung fields. Heart rate was 170/min. No remarkable cardiac murmur was detected. Oxygen saturation checked by pulse oximetry was 90% at the right arm and 75% at the leg. The blood pressures at the right arm and leg were the same and within the normal limits for the patient's age. The baby was intubated and received mechanical ventilation. Arterial blood gas analysis during mechanical ventilation with a FiO2 of 1.0 showed PaO2 180 mmHg, PaCO2 50 mmHg and oxygen saturation 100%. Chest X-ray showed diffuse haziness in both lung fields with air bronchograms, mild cardiomegaly, and large thymic shadow (Fig. 2).
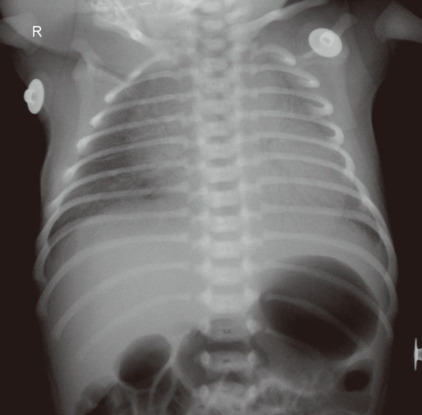
This picture shows a diffuse ground-glass appearance in both lung fields, mild cardiomegaly and a large thymus shadow in the mediastinum.
This case was slightly confusing, but there were some clues to the cause of cyanosis. The baby had a history of meconium staining, and showed cyanosis and severe respiratory symptoms with an abnormal breathing sound. There was no remarkable cardiac murmur or EKG finding. In the chest X-ray (Fig. 2), the heart shadow was slightly large but the main mediastinal shadow was the thymus. A diffuse haziness in both lung fields with an air bronchogram rather than increased vascular markings suggested that primary pulmonary problems were the cause of cyanosis and respiratory distress. Another finding important in differential diagnosis is differential cyanosis, i.e., higher O2 saturation in the right arm than in the leg, which indicates a right-to-left shunt through the PDA. Differential diagnoses in this situation are PPHN and severe coarctation of the aorta or arch interruption. The patient showed a profound response to 100% oxygen supplementation. Echocardiography showed a structurally normal heart with RV dilatation, moderate TR with a velocity of 4 m/sec, RA dilatation, bi-directional shunt (mainly right-to-left shunt during ventricular systole) through the PDA, and right-to-left shunt via PFO. The final diagnosis was PPHN caused by pneumonia and possibly by meconium aspiration. Fortunately, this baby showed rapid improvement after supportive management. Pulmonary vasodilators such as inhaled nitric oxide (iNO), sildenafil, bosentan, or iloprost are good options for severe cases. However, if the condition is intractable to medical management and caused by reversible lung problems, an extracorporeal membrane oxygenation (ECMO) will rescue the patient7).
Case 3. A neonate with severe cyanosis after birth
This baby presented with severe cyanosis just after delivery at a local clinic and was transferred to a tertiary care center with an oxygen supply via a nasal prong. The reported oxygen saturation monitored by pulse oximetry during transfer was around 50-60%. On admission, the baby was mildly tachypneic and mildly lethargic. Whole body cyanosis was noticed, and it did not improve after O2 supplementation (SaO2 around 40%). After intubation and mechanical ventilation with a FiO2 1.0, arterial blood gas analysis showed pH 7.34, PaO2 30 mmHg (O2 saturation 66%), and PaCO2 28 mmHg. Chest examination revealed grade II/VI systolic murmur at the lower sternal border without definite rale or wheezing in the lung fields. The liver was palpable at a 1-finger-breadth. EKG showed a QRS axis of 120°, a tall p wave and right ventricular hypertrophy. Chest radiography showed cardiomegaly but clear lung fields and decreased pulmonary vascular markings (Fig. 3A).
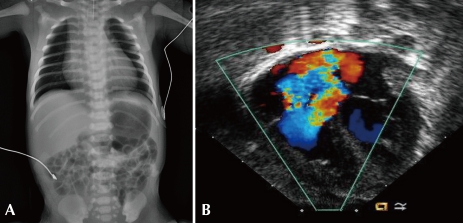
A) Chest radiograph of a neonate with severe cyanosis shows cardiomegaly, but clear lung fields and decreased pulmonary vascular markings. B) Echocardiography with color Doppler mapping shows severe tricuspid regurgitation in a patient with functional pulmonary atresia.
This was a rare case of severe cyanotic CHDs intractable to oxygen therapy and mechanical ventilation alone. Initial physical examination showed clear breathing sound and cardiac murmur, thereby implying the cardiac origin of cyanosis. Chest X-ray showed no evidence of lung problem, but it did show clearer or darker lung fields, decreased pulmonary vascular markings, and cardiomegaly. These findings indicated a lesion with ductal-dependant pulmonary circulation, which necessitated an immediate empirical therapy with prostaglandin infusion to rescue this baby even before echocardiagraphic diagnosis was done (O2 saturation was around 40% and decreasing). Generally, in cases of CHDs, the heart size increases along with an increase in pulmonary blood flow. If there is a cardiomegaly in CHDs with suspected severe PS or PA (conditions with decreased pulmonary blood flow), the possible mechanisms of cardiomegaly would be myocardial dysfunction, severe atrioventricular valve regurgitation (usually involving tricuspid valve), and/or enlargement of the atria. The common cardiac defects of such cases are Ebstein's anomaly of the tricuspid valve, congenital TV dysplasia, and PA with intact ventricular septum (PA IVS) with severe TR. Echocardiography in this patient revealed severe TR (Fig. 3B) due to flail anterior and posterior leaflets, moderate dilatation of the RV, RA enlargement with a right-to-left shunt through the PFO, pulmonary atresia and small PDA. Particularly in this patient, the morphology of the atretic pulmonary valve appeared normal, but the leaflets did not open during systole. There was no antegrade flow through the pulmonary valve during systole, but trivial regurgitation was observed during diastole, thereby implying "functional pulmonary atresia". Functional pulmonary atresia, a relatively uncommon clinical condition, occurs in neonates with severe TR associated with Ebstein's anomaly, tricuspid valve dysplasia, or transient myocardial dysfunction with normal intracardiac anatomy8-10). In patients with increased perinatal pulmonary vascular resistance and severe TR, the RV cannot generate adequate pressure and forward flow to open the pulmonary valve. Therefore, administration of pulmonary vasodilators to lower the pulmonary vascular resistance along with early closure of the PDA, which further elevates the pulmonary artery pressure, may rescue a neonate in a critical clinical state10). In the cases of anatomic pulmonary atresia or critical pulmonary stenosis, balloon valvotomy (valvuloplasty) or surgical treatment is required.
2. Lesions presenting with cyanosis and pulmonary congestion or edema
Case 4. A 6-day-old neonate with severe cyanosis and tachypnea
This term baby was born without any apparent perinatal problems and was discharged home 2 days after birth. At home, the baby was reported to be doing well for one or two days after discharge, but it appeared blue and showed tachypnea and poor feeding. The baby was brought to the local clinic at which it was born, and it was transferred emergently to a tertiary care center. On admission, the baby looked pale and cyanotic and showed tachypnea. Oxygen saturation values at the right fingers and toes were around 60% initially and 75% after O2 supplementation. The respiratory rate was 70/min with chest wall retraction. Chest auscultation revealed fine crackles in the lung fields but did not reveal any remarkable cardiac murmur. The blood pressures at the right arm and the right ankle were 60/45 mmHg. EKG showed no remarkable findings, and chest radiograph showed cardiomegaly and diffuse haziness and increased pulmonary vascular markings (Fig. 4A).
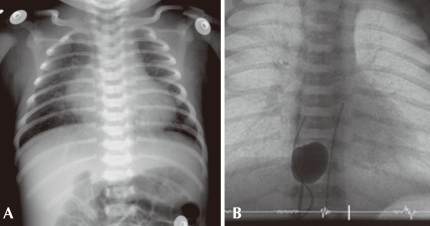
A) This picture shows cardiomegaly with increased pulmonary vascular markings. The mediastinal shadow is narrow and the cardiac silhouette looks like an "egg-on-side". B) A fluoroscopic image of balloon atrial septostomy in a neonate with transposition of the great arteries.
This patient showed profound cyanosis and respiratory difficulty that did not respond to 100% oxygen supply, thereby implying the cardiac origin of the cyanosis. The chest X-ray (Fig. 4A) showed cardiomegaly and pulmonary congestion indicating increased pulmonary blood flow. Because the severe cyanosis was observed with pulmonary over-circulation (no PS) in this patient, this case can be considered to be quite different from the previous cases of right-sided obstructive lesions (case 1 or case 3), which presented with decreased pulmonary blood flow and intracardiac right-to left shunt.
The most common diagnosis for this case could be a simple TGA (with no VSD and PS), in which parallel rather than serial systemic and pulmonary circulation causes severe cyanosis (the result of inadequate intercirculatory mixing) and pulmonary congestion. A similar situation can occur in cases of TAPVR or truncus arteriosus, which are ductal-independent mixing lesions. In obstructive type TAPVR with severe cyanosis, the pulmonary venous congestion or edema is striking and the heart size is usually normal. In truncus arteriosus, pulmonary congestion with cardiomegaly is evident, but cyanosis is usually mild because of adequate mixing of the blood via the large VSD.
In this case, after initial empirical therapy with prostaglandin infusion and diuretics, echocardiography confirmed the diagnosis of TGA with a constricting PDA and restrictive mixing of blood through the PFO. Because of persistent severe cyanosis and pulmonary congestion, an emergency balloon atrial septostomy was done on the day of admission (Fig. 4B), and an arterial switch operation (the Jatene procedure) was performed two days later.
Case 5. A neonate presenting with severe cyanosis and respiratory distress after birth
This baby was born at a gestational age of 35 weeks and had birth weight of 2.5 kg. At birth, it showed cyanosis and mild tachypnea and was admitted to an intensive care unit for close observation and oxygen supplementation. At 3-4 h after birth, the patient's cyanosis and tachypnea aggravated, and it underwent intubation and mechanical ventilation. Oxygen saturation values before and after mechanical ventilation (FiO2, 1.0) were 65% and 85%, respectively. Chest auscultation revealed no remarkable cardiac murmur. Fine crackles were heard in both lung fields. EKG was unremarkable. Chest X-ray (Fig. 5) showed severe and diffuse haziness with obliteration of the right cardiac silhouette and a left-sided cardiac apex. Respiratory distress syndrome (RDS) was initially suspected, and a surfactant was administered. However, the clinical conditions did not improve after the surfactant treatment and the ventilator support.
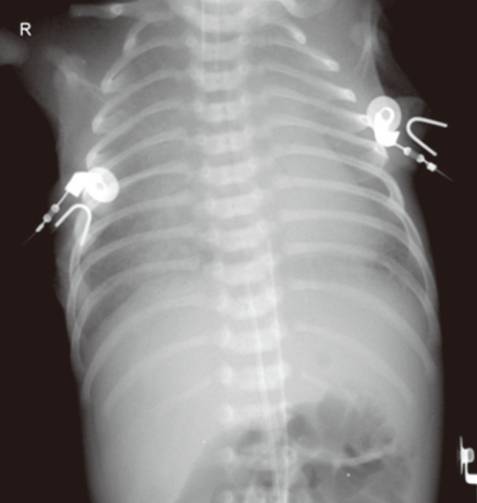
Chest radiograph of a preterm neonate with severe cyanosis and respiratory distress shows a diffuse and severe haziness nearly obliterating the cardiac silhouette. The final diagnosis was an obstructive type of total anomalous pulmonary venous return.
This preterm baby presented with progressively worsening severe cyanosis and respiratory difficulty a few hours after birth, and was suspected to have RDS. The remarkable finding in the chest radiograph was a severe and diffuse haziness in both lung fields that nearly obliterated the cardiac silhouette (Fig. 5). It is quite reasonable to suspect RDS in this preterm neonate, although other causes such as bacterial pneumonia should be considered. However, in cases with severe cyanosis and respiratory distress that do not respond to oxygen supplementation and positive mechanical ventilation, and/or surfactant administration, TAPVR with severe pulmonary venous obstruction should be ruled out by immediate echocardiographic examination, because TAPVR with severe pulmonary venous obstruction will require immediate surgical repair. This case was diagnosed as an infracardiac type of TAPVR (in which common pulmonary venous channel drains into the portal system) with severe obstruction, and the patient was sent to the operating room for surgical repair.
3. Lesion presenting with systemic hypotension, shock or collapse
Case 6. A neonate presenting with sudden shock and acute renal failure
A 9-day-old neonate was transferred to the neonatal intensive care unit for the evaluation of lethargy and respiratory distress. The full-term baby was born by cesarean section at a local obstetric clinic and was discharged home two days after birth. The baby was reported to have done "quite well" at home until the 6th day after birth, when her mother noticed that her breathing was very fast with chest retraction and that she appeared pale and lethargic. On admission, the baby looked pale and lethargic. The skin on the abdomen and legs was mildly cyanotic, icteric, and mottled. Skin turgor was decreased. The blood pressures were 70/45 mmHg (right arm) and 50/30 mmHg (leg). Oxygen saturations were 95% (right finger) and 85% (toes). Respiration rate was 70/min and heart rate was 160/min. Body temperature was 37℃. Chest auscultation revealed grade II-III/VI ejection-type cardiac murmur at the upper left sternal border and no remarkable rale or wheezing in the lung fields. EKG showed a sinus rhythm and biventricular hypertrophy. Chest radiography (Fig. 6) showed cardiomegaly and increased pulmonary vascular markings. Arterial blood gas analysis showed pH 7.30, pCO2 27mmHg, pO2 68 mmHg, and HCO3 15 mmol/L. The blood urea nitrogen (BUN) and creatinine (Cr) were 20.3 mg/dL and 1.24 mg/dL, respectively. Serum glutamic-oxaloacetic transaminase (GOT) and glutamic-pyruvic transaminase (GPT) were 746 and 154 U/L, respectively. The serum creatine phosphokinase (CPK) and lactate dehydrogenase (LDH) were 1,720 and 6,410 U/L, respectively. No urine output was observed for the first 2 hours after admission.
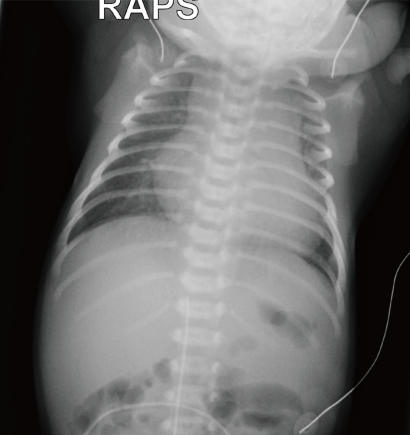
Chest radiograph of a neonate presenting with severe respiratory distress and shock shows severe cardiomegaly, hepatomegaly and increased pulmonary vascular markings.
This patient's main clinical manifestation was a sudden deterioration of systemic circulation, which was followed by acute renal failure and respiratory distress. However, cyanosis was not remarkable in this patient. The characteristic findings on physical examinations were differential cyanosis and different blood pressures in the preductal (right upper extremity) and postductal (leg) area. The higher blood pressure in the preductal part of the body indicated lesions with aortic arch obstruction or interruption, although similar blood pressures in both parts of the body cannot rule out aortic arch obstruction or interruption in the presence of a large PDA. The differential cyanosis (higher O2 saturation in the right arm) indicates the right-to-left shunt through the PDA. Regardless of the anatomic diagnosis in detail, these patients should be resuscitated immediately by using advanced life support and empirical therapy, including mechanical ventilation with sedation and paralysis, infusion of prostaglandin, adequate fluid therapy and diuretic administration, and inotropic support. This patient was diagnosed as an interruption of the aortic arch (type A), a constricting PDA, and a large subarterial VSD. However, this complicated CHD had not been recognized at birth and the patient had been discharged home. Not a small proportion of neonates with critical CHD may appear normal or have subtle signs; these signs may escape the clinician's notice in a routine neonatal examination1, 4, 11, 12).
The most common potentially fatal defects missed at birth discharge are left-sided obstructive lesions such as HLHS and coarctation of the aorta4, 11, 12). In the majority of patients showing left-sided obstructive lesions just after birth, the systemic circulation is maintained by blood flow through the large PDA; pulmonary overflow is not profound because of elevated pulmonary vascular resistance; and cyanosis is usually mild, especially in the upper part of the body. A decrease in pulmonary vascular resistance and the PDA size may result in sudden systemic hypotension or shock and severe pulmonary congestion. Therefore, in routine clinical practice, clinicians should be alert to subtle signs of heart failure, such as tachypnea, tachycardia, unexplained cough or irritability, poor and interrupted feeding, excessive sweating, or pallor.
4. Other complex lesions with single ventricle physiology or heterotaxy syndrome
Other complex cardiac defects with single ventricle physiology or heterotaxy syndromes may show various combinations of obstruction in the pulmonary or systemic circulation (which can be lesions associated with either ductal-dependent pulmonary or systemic circulation) and intracardiac anatomy. Clinical manifestations will vary according to the associated lesions and may include severe cyanosis or severe congestive heart failure, or both. Clinical decision-making in such cases is basically the same as previously described and discussed. In addition to symptoms, signs, and physical findings, chest radiography is very useful to identify complex lesions. The position of the abdominal organs, the shape, position, and size of the heart, and pulmonary vascularity should be studied carefully. Transverse liver, midline stomach gas shadow, and abnormal position of the heart on the chest radiograph indicate a heterotaxy syndrome, and relatively small heart silhouette and decreased pulmonary vascularity indicate significant PS (Fig. 7). Usual position of the abnormal organs with dextrocardia, cardiomegaly and increased pulmonary vascular markings suggest a complex CHD without PS, that may present with severe congestive heart failure rather than severe cyanosis (Fig. 8).
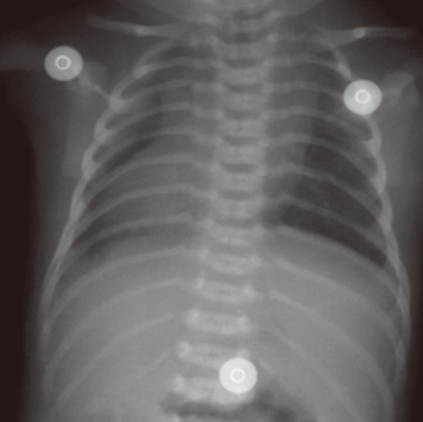
Chest radiograph of a patient presenting with cyanosis shows a transverse liver, midline stomach shadow, and dextrocardia, which suggest a complex heart defect, probably a heterotaxia syndrome. Clear lung fields with a normal cardiac size indicate a significant PS and decreased pulmonary blood flow in the clinical situation.
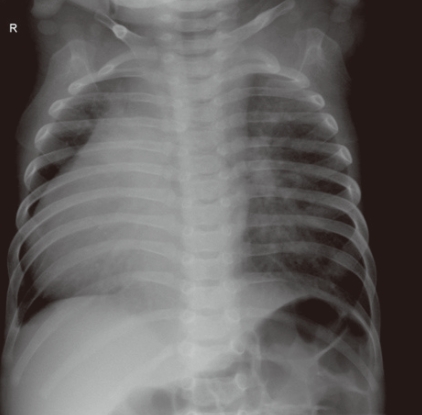
This patient has a usual position of the abdominal organs (right-sided liver and left-sided stomach) and dextrocardia, thereby suggesting a complex lesion. Cardiomegaly and increased pulmonary vascular markings suggest pulmonary overcirculation, which may lead to congestive heart failure rather than severe cyanosis.
Conclusions
Neonates with Critical CHDs may be unrecognized at birth, discharged home, and deteriorate or die before accurate cardiac diagnosis and management. Clinicians should be vigilant for subtle signs and symptoms of significant heart defects. All neonates presenting with severe cyanosis, respiratory distress, shock, or collapse should be suspected to have potentially life-threatening cardiac defects; lesions with ductal-dependant pulmonary or systemic circulation and ductal-independent but critical lesions, including TGA and TAPVR. Differentiation of CHDs from pulmonary problems (lesions with cyanosis) or sepsis (lesions presenting systemic shock) is of utmost importance, and may be possible in the majority of patients by performing careful history-taking, physical examinations with four-extremity blood pressures and pre- and post-ductal oxygen saturations, hyperoxia test, EKG, and chest radiography. Although an accurate diagnosis by a cardiologist is extremely preferable, immediate empirical therapies with prostaglandin infusion, inotropic support, or adequate fluid therapy with dieresis should not be delayed in patients suspected to have critical CHDs.
